TEMPFLEX
Crown And Bridge, Temporary, Resin
KERR CORPORATION
The following data is part of a premarket notification filed by Kerr Corporation with the FDA for Tempflex.
Pre-market Notification Details
| Device ID | K110747 |
| 510k Number | K110747 |
| Device Name: | TEMPFLEX |
| Classification | Crown And Bridge, Temporary, Resin |
| Applicant | KERR CORPORATION 1717 WEST COLLINS AVENUE Orange, CA 92867 |
| Contact | Wendy Garman |
| Correspondent | Wendy Garman KERR CORPORATION 1717 WEST COLLINS AVENUE Orange, CA 92867 |
| Product Code | EBG |
| CFR Regulation Number | 872.3770 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2011-03-17 |
| Decision Date | 2011-06-14 |
| Summary: | summary |
Trademark Results [TEMPFLEX]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
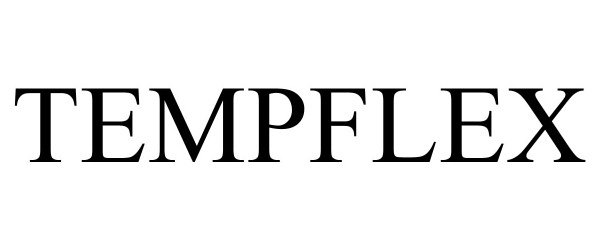 TEMPFLEX 97876241 not registered Live/Pending |
Techtronic Cordless GP 2023-04-06 |
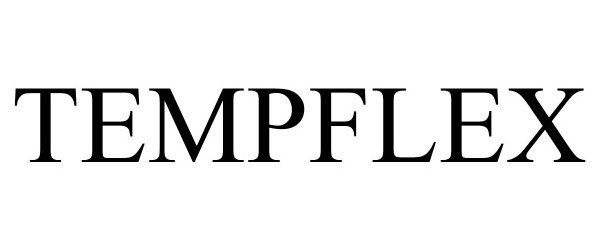 TEMPFLEX 90445509 not registered Live/Pending |
ANHUI PARKER NEW MATERIAL CO.,LTD 2021-01-01 |
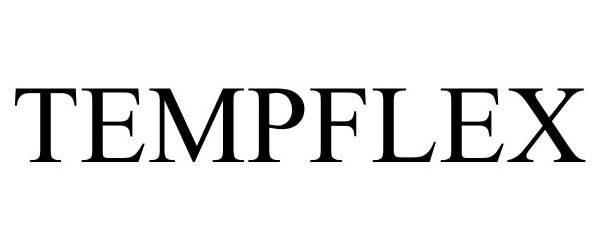 TEMPFLEX 77479853 3557026 Dead/Cancelled |
Environmental Lubricants Manufacturing,Inc. 2008-05-21 |
 TEMPFLEX 73007946 0996780 Dead/Expired |
DAYCO CORPORATION 1973-12-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.