ImPACT
Computerized Cognitive Assessment Aid For Concussion
IMPACT APPLICATIONS, INC
The following data is part of a premarket notification filed by Impact Applications, Inc with the FDA for Impact.
Pre-market Notification Details
| Device ID | K170209 |
| 510k Number | K170209 |
| Device Name: | ImPACT |
| Classification | Computerized Cognitive Assessment Aid For Concussion |
| Applicant | IMPACT APPLICATIONS, INC 9665 GRANITE RIDGE DRIVE SUITE 550 San Diego, CA 92123 |
| Contact | Michael Zagorski |
| Correspondent | Michael Zagorski IMPACT APPLICATIONS, INC 9665 GRANITE RIDGE DRIVE SUITE 550 San Diego, CA 92123 |
| Product Code | POM |
| CFR Regulation Number | 882.1471 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Special |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2017-01-24 |
| Decision Date | 2017-02-23 |
| Summary: | summary |
Trademark Results [ImPACT]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 IMPACT 98601500 not registered Live/Pending |
Marli J. Washington 2024-06-14 |
 IMPACT 98528216 not registered Live/Pending |
L'Chef, LLC 2024-04-30 |
 IMPACT 98528203 not registered Live/Pending |
L'Chef, LLC 2024-04-30 |
 IMPACT 98502001 not registered Live/Pending |
Hornady Manufacturing Company 2024-04-16 |
 IMPACT 98384931 not registered Live/Pending |
HCMP, Inc. 2024-01-31 |
 IMPACT 98328305 not registered Live/Pending |
The Impact Fund 2023-12-22 |
 IMPACT 98328269 not registered Live/Pending |
Indian American Impact 2023-12-22 |
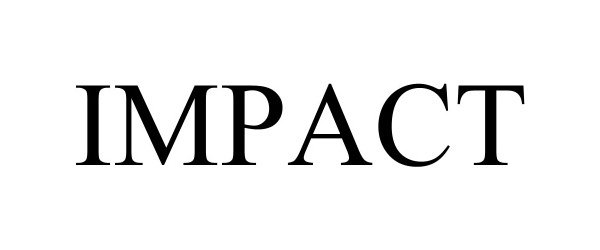 IMPACT 98299820 not registered Live/Pending |
NOBULL, LLC 2023-12-05 |
 IMPACT 98285346 not registered Live/Pending |
Lazarex Cancer Foundation 2023-11-26 |
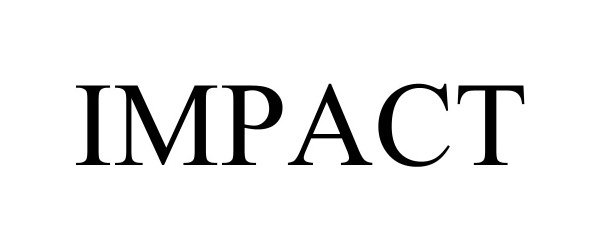 IMPACT 98277080 not registered Live/Pending |
Montage Hotels & Resorts, LLC 2023-11-19 |
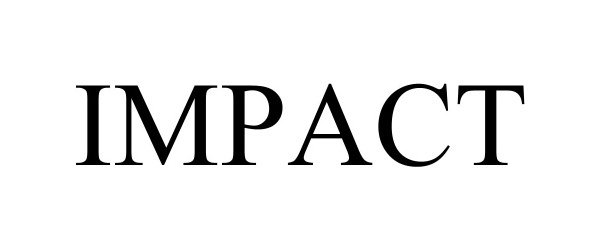 IMPACT 98189133 not registered Live/Pending |
Alveo Technologies Inc. 2023-09-20 |
 IMPACT 98090236 not registered Live/Pending |
MedTech Global Products LLC 2023-07-18 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.