ImPACT Quick Test
Computerized Cognitive Assessment Aid For Concussion
ImPACT Applications, Inc.
The following data is part of a premarket notification filed by Impact Applications, Inc. with the FDA for Impact Quick Test.
Pre-market Notification Details
| Device ID | K170551 |
| 510k Number | K170551 |
| Device Name: | ImPACT Quick Test |
| Classification | Computerized Cognitive Assessment Aid For Concussion |
| Applicant | ImPACT Applications, Inc. 9665 Granite Ridge Drive, Suite 550 San Diego, CA 92123 |
| Contact | Michael Zagorski |
| Correspondent | Michael Zagorski ImPACT Applications, Inc. 9665 Granite Ridge Drive, Suite 550 San Diego, CA 92123 |
| Product Code | POM |
| CFR Regulation Number | 882.1471 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2017-02-24 |
| Decision Date | 2017-06-21 |
| Summary: | summary |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 00864127000325 | K170551 | 000 |
| 00864127000356 | K170551 | 000 |
Trademark Results [ImPACT Quick Test]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
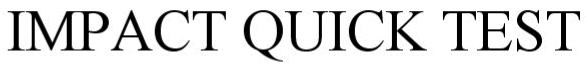 IMPACT QUICK TEST 87977941 5537423 Live/Registered |
ImPACT Applications, Inc. 2016-08-04 |
 IMPACT QUICK TEST 87328913 5746036 Live/Registered |
ImPACT Applications, Inc. 2017-02-08 |
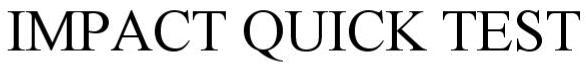 IMPACT QUICK TEST 87127120 5530843 Live/Registered |
ImPACT Applications, Inc. 2016-08-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.