CARDIOSKIN
Transmitters And Receivers, Electrocardiograph, Telephone
Bioserenity SAS
The following data is part of a premarket notification filed by Bioserenity Sas with the FDA for Cardioskin.
Pre-market Notification Details
| Device ID | K173248 |
| 510k Number | K173248 |
| Device Name: | CARDIOSKIN |
| Classification | Transmitters And Receivers, Electrocardiograph, Telephone |
| Applicant | Bioserenity SAS 47 Boulevard De L'Hopital Paris, FR 75013 |
| Contact | Quang Tran |
| Correspondent | Esin Yesilalan Voisin Consulting Inc. Life Sciences 222 Third Street Suite 3121 Cambridge, MA 02142 |
| Product Code | DXH |
| CFR Regulation Number | 870.2920 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2017-10-10 |
| Decision Date | 2018-08-11 |
| Summary: | summary |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 03615220001156 | K173248 | 000 |
| 03615220001033 | K173248 | 000 |
| 03615220001040 | K173248 | 000 |
| 03615220001057 | K173248 | 000 |
| 03615220001064 | K173248 | 000 |
| 03615220001071 | K173248 | 000 |
| 03615220001088 | K173248 | 000 |
| 03615220001095 | K173248 | 000 |
| 03615220001101 | K173248 | 000 |
| 03615220001118 | K173248 | 000 |
| 03615220001125 | K173248 | 000 |
| 03615220001132 | K173248 | 000 |
| 03615220001149 | K173248 | 000 |
| 03615220001026 | K173248 | 000 |
Trademark Results [CARDIOSKIN]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
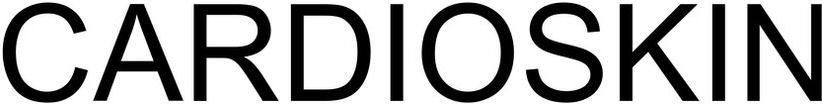 CARDIOSKIN 79199348 5455572 Live/Registered |
BIOFARMA 2016-08-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.