InCore® Lapidus Sterile Kits
Screw, Fixation, Bone
Nextremity Solutions, Inc.
The following data is part of a premarket notification filed by Nextremity Solutions, Inc. with the FDA for Incore® Lapidus Sterile Kits.
Pre-market Notification Details
| Device ID | K190231 |
| 510k Number | K190231 |
| Device Name: | InCore® Lapidus Sterile Kits |
| Classification | Screw, Fixation, Bone |
| Applicant | Nextremity Solutions, Inc. 210 North Buffalo Street Warsaw, IN 46580 |
| Contact | Greg Denham |
| Correspondent | Greg Denham Nextremity Solutions, Inc. 210 North Buffalo Street Warsaw, IN 46580 |
| Product Code | HWC |
| CFR Regulation Number | 888.3040 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2019-02-06 |
| Decision Date | 2019-05-24 |
Trademark Results [InCore]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
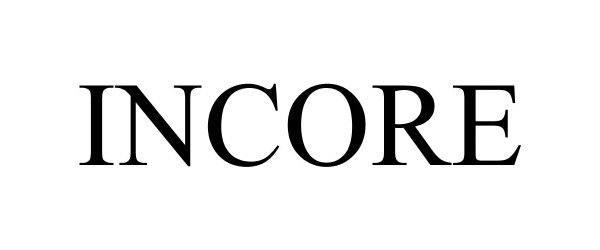 INCORE 87677798 5634277 Live/Registered |
NEXTREMITY SOLUTIONS, INC. 2017-11-09 |
 INCORE 86934148 5067606 Live/Registered |
LINKCOM Manufacturing Co., Ltd. 2016-03-09 |
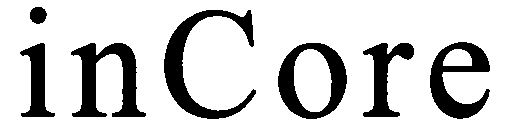 INCORE 79027932 3639604 Live/Registered |
NAP ENTERPRISE CO., LTD. 2006-07-21 |
 INCORE 76335151 not registered Dead/Abandoned |
CISCO TECHNOLOGY, INC. 2001-11-06 |
 INCORE 74693258 2186164 Dead/Cancelled |
ISG TECHNOLOGIES INC. 1995-06-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.