Flex Fit
Resin, Denture, Relining, Repairing, Rebasing
DK Mungyo Corporation
The following data is part of a premarket notification filed by Dk Mungyo Corporation with the FDA for Flex Fit.
Pre-market Notification Details
| Device ID | K190790 |
| 510k Number | K190790 |
| Device Name: | Flex Fit |
| Classification | Resin, Denture, Relining, Repairing, Rebasing |
| Applicant | DK Mungyo Corporation 248, Anha-ro, Hallim-myeon Gimhae-si, KR 50852 |
| Contact | Kang Nae In |
| Correspondent | Priscilla Chung LK Consulting Group USA, Inc. 1150 Roosevelt, STE 200 Irvine, CA 92620 |
| Product Code | EBI |
| CFR Regulation Number | 872.3760 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2019-03-27 |
| Decision Date | 2020-07-31 |
Trademark Results [Flex Fit]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FLEX FIT 98307857 not registered Live/Pending |
deko, LLC 2023-12-11 |
 FLEX FIT 97460497 not registered Live/Pending |
Hero, Stephen 2022-06-16 |
 FLEX FIT 97389419 not registered Live/Pending |
deko, LLC 2022-04-29 |
 FLEX FIT 90381661 not registered Live/Pending |
Magnetek, Inc. 2020-12-14 |
 FLEX FIT 87524836 5628820 Live/Registered |
Eastman Outdoors, LLC 2017-07-12 |
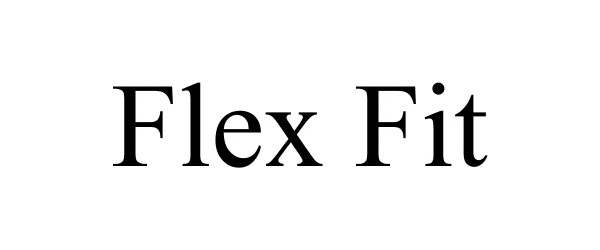 FLEX FIT 78613426 not registered Dead/Abandoned |
Eastern Pacific Apparel, Inc. 2005-04-21 |
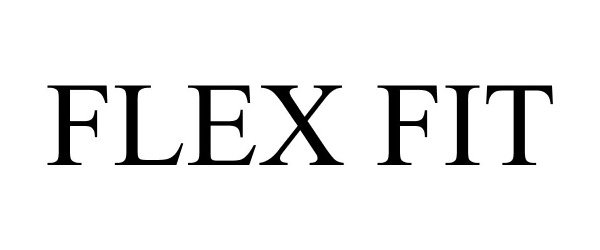 FLEX FIT 77649946 3764992 Live/Registered |
Poly-America, L.P. 2009-01-15 |
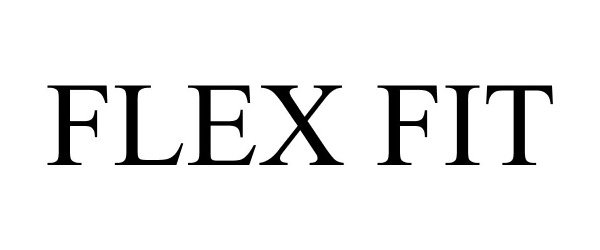 FLEX FIT 77334551 not registered Dead/Abandoned |
Shooting Star International 2007-11-20 |
 FLEX FIT 76239757 not registered Dead/Abandoned |
Stuller Settings, Inc. 2001-04-13 |
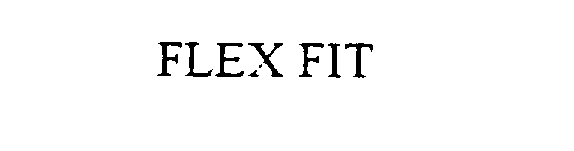 FLEX FIT 75799243 not registered Dead/Abandoned |
Sportscope, Inc. 1999-09-15 |
 FLEX FIT 75507161 not registered Dead/Abandoned |
Bach, Barry 1998-06-23 |
 FLEX FIT 75315174 not registered Dead/Abandoned |
Emhart Inc. 1997-06-26 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.