I-ED COIL
Device, Neurovascular Embolization
Kaneka Pharma America LLC
The following data is part of a premarket notification filed by Kaneka Pharma America Llc with the FDA for I-ed Coil.
Pre-market Notification Details
| Device ID | K192068 |
| 510k Number | K192068 |
| Device Name: | I-ED COIL |
| Classification | Device, Neurovascular Embolization |
| Applicant | Kaneka Pharma America LLC 546 Fifth Avenue 21st Floor New York, NY 10036 |
| Contact | Kazuhiko Inoue |
| Correspondent | Takeaki Miyata Kaneka Corporation 1-12-32 Akasaka Minato-ku, JP 107-6028 |
| Product Code | HCG |
| CFR Regulation Number | 882.5950 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2019-08-01 |
| Decision Date | 2020-04-25 |
NIH GUDID Devices
Trademark Results [I-ED COIL]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
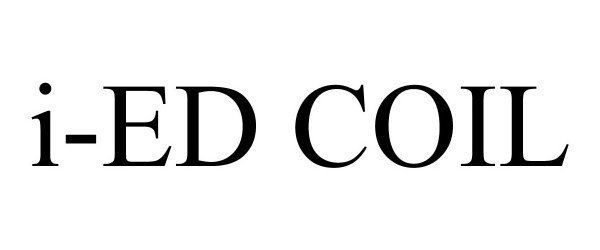 I-ED COIL 90192094 not registered Live/Pending |
KANEKA CORPORATION 2020-09-18 |
 I-ED COIL 88471539 not registered Live/Pending |
KANEKA CORPORATION 2019-06-13 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.