Dental Pain Eraser
Device, Electrical Dental Anesthesia
Synapse Dental
The following data is part of a premarket notification filed by Synapse Dental with the FDA for Dental Pain Eraser.
Pre-market Notification Details
| Device ID | K193570 |
| 510k Number | K193570 |
| Device Name: | Dental Pain Eraser |
| Classification | Device, Electrical Dental Anesthesia |
| Applicant | Synapse Dental 67 Phenix Avenue Cranston, RI 02920 |
| Contact | Cosmo Haralambidis |
| Correspondent | Valerie Defiesta-ng Experien Group, LLC 224 Airport Parkway, Suite 250 San Jose, CA 95110 |
| Product Code | LWM |
| CFR Regulation Number | 510(k) Premarket Notification [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Special |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2019-12-23 |
| Decision Date | 2020-01-17 |
Trademark Results [Dental Pain Eraser]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
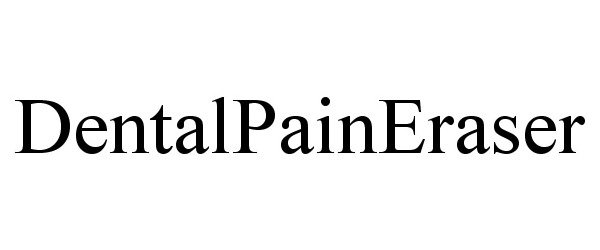 DENTAL PAIN ERASER 87587652 5885907 Live/Registered |
Haralambidis, Cosmo 2017-08-29 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.