SimpleSENSE
Transmitters And Receivers, Electrocardiograph, Telephone
Nanowear Inc.
The following data is part of a premarket notification filed by Nanowear Inc. with the FDA for Simplesense.
Pre-market Notification Details
| Device ID | K201669 |
| 510k Number | K201669 |
| Device Name: | SimpleSENSE |
| Classification | Transmitters And Receivers, Electrocardiograph, Telephone |
| Applicant | Nanowear Inc. 53 Boerum Place, Suite 3F Brooklyn, NY 11201 |
| Contact | Venkatesh Varadan |
| Correspondent | Melissa Walker Graematter, Inc. 1324 Clarkson Clayton Ctr, #332 St Louis, MO 63011 |
| Product Code | DXH |
| Subsequent Product Code | BZQ |
| Subsequent Product Code | DPS |
| Subsequent Product Code | DQD |
| Subsequent Product Code | DSB |
| CFR Regulation Number | 870.2920 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2020-06-19 |
| Decision Date | 2020-11-06 |
Trademark Results [SimpleSENSE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
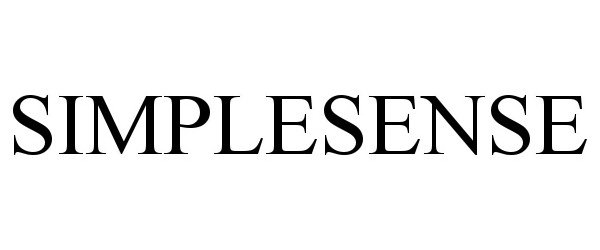 SIMPLESENSE 90863147 not registered Live/Pending |
Nanowear, Inc. 2021-08-03 |
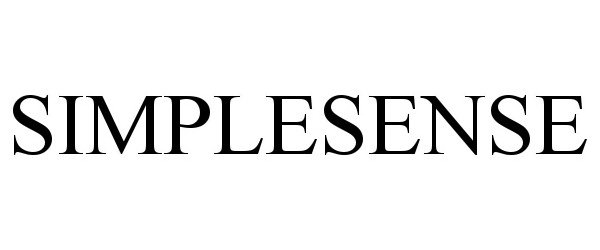 SIMPLESENSE 90805663 not registered Live/Pending |
SimpleSense Inc. 2021-07-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.