IRAD
System, Image Processing, Radiological
Claritas HealthTech Pte Ltd
The following data is part of a premarket notification filed by Claritas Healthtech Pte Ltd with the FDA for Irad.
Pre-market Notification Details
| Device ID | K212470 |
| 510k Number | K212470 |
| Device Name: | IRAD |
| Classification | System, Image Processing, Radiological |
| Applicant | Claritas HealthTech Pte Ltd 20A Tanjong Pagar Road Singapore, SG 088443 |
| Contact | Devika Dutt |
| Correspondent | Devika Dutt Claritas HealthTech Pte Ltd 20A Tanjong Pagar Road Singapore, SG 088443 |
| Product Code | LLZ |
| CFR Regulation Number | 892.2050 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2021-08-06 |
| Decision Date | 2021-10-20 |
Trademark Results [IRAD]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 IRAD 90375160 not registered Live/Pending |
NuTech Energy Alliance, Ltd. 2020-12-11 |
 IRAD 87926534 5759137 Live/Registered |
Independent Radiology Partners, LLC 2018-05-17 |
 IRAD 85097612 3940491 Dead/Cancelled |
Guilherme Cardoso de Cardoso 2010-08-01 |
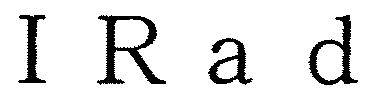 IRAD 79052582 3595083 Live/Registered |
Tokyo Electron Limited 2008-03-21 |
 IRAD 78636012 3326210 Live/Registered |
IRAD LLC INTERNET RADIO 2005-05-24 |
 IRAD 76017957 not registered Dead/Abandoned |
Qureshey, Safi 2000-04-05 |
 IRAD 73655982 1475455 Dead/Cancelled |
INTERNATIONAL RESEARCH AND DEVELOPMENT CORPORATION 1987-04-20 |
 IRAD 73655981 1467747 Dead/Cancelled |
INTERNATIONAL RESEARCH AND DEVELOPMENT CORPORATION 1987-04-20 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.