BonOs® Inject Bone Cement; NEO Pedicle Screw System™
Bone Cement, Posterior Screw Augmentation
Neo Medical SA
The following data is part of a premarket notification filed by Neo Medical Sa with the FDA for Bonos® Inject Bone Cement; Neo Pedicle Screw System™.
Pre-market Notification Details
| Device ID | K222256 |
| 510k Number | K222256 |
| Device Name: | BonOs® Inject Bone Cement; NEO Pedicle Screw System™ |
| Classification | Bone Cement, Posterior Screw Augmentation |
| Applicant | Neo Medical SA Route De Lausanne 157 A Villette (Lavaux), CH 1096 |
| Contact | Jonas Larsson |
| Correspondent | Sandra Soniec meditec Consulting GmbH Obermoosstrasse 23 Boll, CH 3067 |
| Product Code | PML |
| Subsequent Product Code | NDN |
| Subsequent Product Code | NKB |
| CFR Regulation Number | 888.3027 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Special |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2022-07-27 |
| Decision Date | 2022-08-26 |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 07640177821225 | K222256 | 000 |
| 07640177821218 | K222256 | 000 |
| 07640177821195 | K222256 | 000 |
| 07640177821058 | K222256 | 000 |
| 07640177821041 | K222256 | 000 |
| 07640177821034 | K222256 | 000 |
| 07640177821027 | K222256 | 000 |
| 07640177820839 | K222256 | 000 |
| 07640177820822 | K222256 | 000 |
Trademark Results [BonOs]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
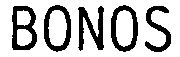 BONOS 79030690 3392067 Live/Registered |
Osartis GmbH 2006-08-28 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.