BODYSCAN
Camera, Scintillation (gamma)
SIEMENS GAMMASONICS, INC.
The following data is part of a premarket notification filed by Siemens Gammasonics, Inc. with the FDA for Bodyscan.
Pre-market Notification Details
| Device ID | K870818 |
| 510k Number | K870818 |
| Device Name: | BODYSCAN |
| Classification | Camera, Scintillation (gamma) |
| Applicant | SIEMENS GAMMASONICS, INC. 2000 NUCLEAR DR. Des Plaines, IL 60018 |
| Contact | Joseph P Stowell |
| Correspondent | Joseph P Stowell SIEMENS GAMMASONICS, INC. 2000 NUCLEAR DR. Des Plaines, IL 60018 |
| Product Code | IYX |
| CFR Regulation Number | 892.1100 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 1987-03-03 |
| Decision Date | 1987-03-16 |
Trademark Results [BODYSCAN]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
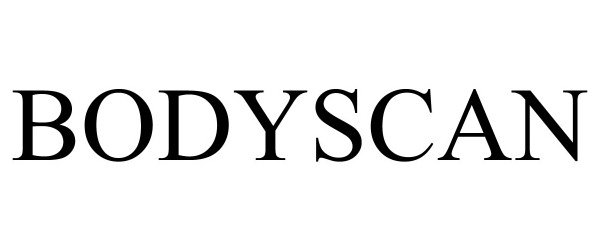 BODYSCAN 90738861 not registered Live/Pending |
MerchSource, LLC 2021-05-27 |
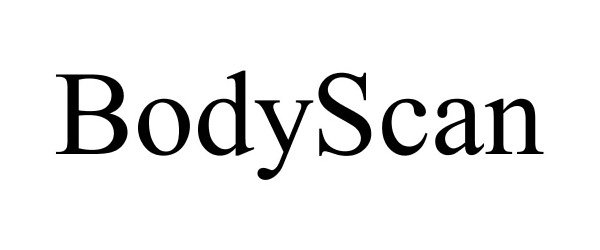 BODYSCAN 85905042 not registered Dead/Abandoned |
TechMed 3D Inc. 2013-04-15 |
 BODYSCAN 78095555 not registered Dead/Abandoned |
BodyScan Holdings, Inc. 2001-11-28 |
 BODYSCAN 78028369 not registered Dead/Abandoned |
Multi-Dimensional Imaging, Inc. 2000-09-29 |
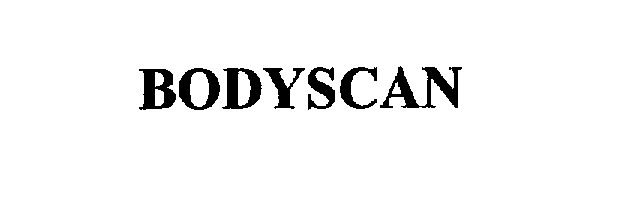 BODYSCAN 75868080 2423209 Dead/Cancelled |
SonnenBraune, Inc. 1999-12-10 |
 BODYSCAN 75531476 not registered Dead/Abandoned |
Phazx Systems Inc. 1998-08-05 |
 BODYSCAN 75486881 not registered Dead/Abandoned |
BRAUN AKTIENGESELLSCHAFT 1998-05-19 |
 BODYSCAN 75451962 not registered Dead/Abandoned |
Sanan, Denis 1998-03-17 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.