ENDO-SPONGE
Accessories, Cleaning Brushes, For Endoscope
ENDOVATIONS
The following data is part of a premarket notification filed by Endovations with the FDA for Endo-sponge.
Pre-market Notification Details
| Device ID | K892218 |
| 510k Number | K892218 |
| Device Name: | ENDO-SPONGE |
| Classification | Accessories, Cleaning Brushes, For Endoscope |
| Applicant | ENDOVATIONS ROSSMOYNE INDUSTRIAL PARK 5020 RITTER ROAD, SUITE 211 Mechanicsburg, PA 17055 |
| Contact | Susan P Bjorkman |
| Correspondent | Susan P Bjorkman ENDOVATIONS ROSSMOYNE INDUSTRIAL PARK 5020 RITTER ROAD, SUITE 211 Mechanicsburg, PA 17055 |
| Product Code | MNL |
| CFR Regulation Number | 876.1500 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 1989-04-03 |
| Decision Date | 1989-05-31 |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 10350770928118 | K892218 | 000 |
| 10680651928112 | K892218 | 000 |
Trademark Results [ENDO-SPONGE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
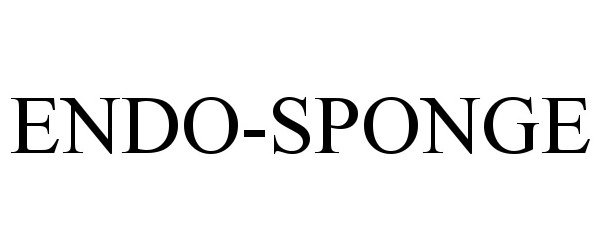 ENDO-SPONGE 90861086 not registered Live/Pending |
Aesculap AG 2021-08-02 |
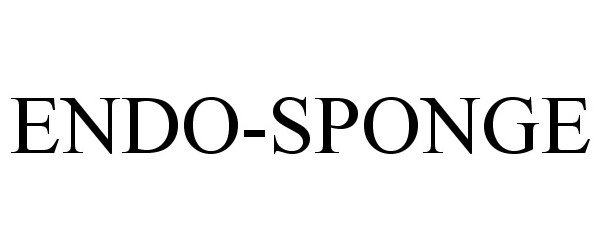 ENDO-SPONGE 86477426 4789447 Live/Registered |
Aesculap AG 2014-12-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.