SMART PLUG
Powered Laser Surgical Instrument
HGM, INC.
The following data is part of a premarket notification filed by Hgm, Inc. with the FDA for Smart Plug.
Pre-market Notification Details
| Device ID | K950120 |
| 510k Number | K950120 |
| Device Name: | SMART PLUG |
| Classification | Powered Laser Surgical Instrument |
| Applicant | HGM, INC. 3959 WEST 1820 SOUTH Salt Lake City, UT 84104 |
| Contact | C L Christman |
| Correspondent | C L Christman HGM, INC. 3959 WEST 1820 SOUTH Salt Lake City, UT 84104 |
| Product Code | GEX |
| CFR Regulation Number | 878.4810 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 1995-01-12 |
| Decision Date | 1995-08-11 |
Trademark Results [SMART PLUG]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SMART PLUG 97759072 not registered Live/Pending |
H.S. CRAFT MANUFACTURING CO. 2023-01-18 |
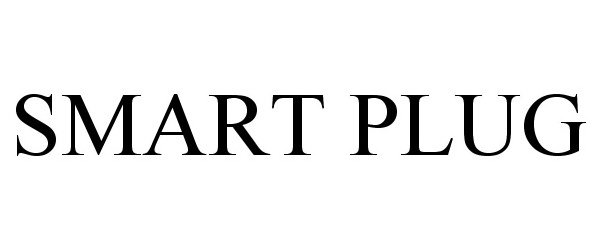 SMART PLUG 87313123 5289821 Live/Registered |
Verscene Inc. 2017-01-25 |
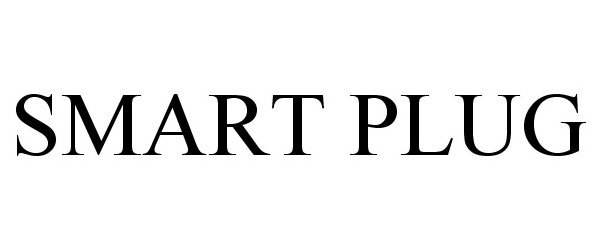 SMART PLUG 85848856 4490468 Live/Registered |
MCALISTER TECHNOLOGIES, LLC 2013-02-13 |
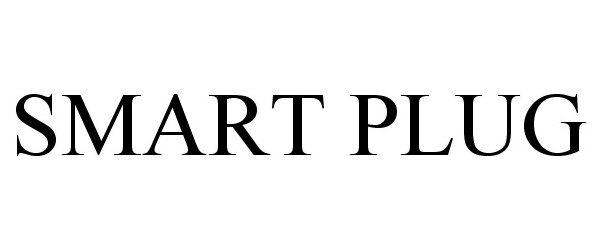 SMART PLUG 78749184 3473791 Dead/Cancelled |
GPK Products Inc. 2005-11-08 |
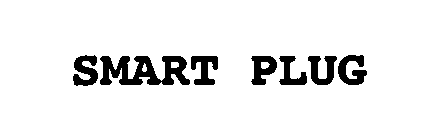 SMART PLUG 76466566 3170203 Live/Registered |
MCALISTER TECHNOLOGIES, LLC 2002-11-13 |
 SMART PLUG 76387943 not registered Dead/Abandoned |
Smart Company Creations, Inc. 2002-03-27 |
 SMART PLUG 75685069 not registered Dead/Abandoned |
Lutz, Tony 1999-05-07 |
 SMART PLUG 75654884 not registered Dead/Abandoned |
Jitendra Ambalal Patel 1999-03-14 |
 SMART PLUG 75558076 not registered Dead/Abandoned |
CRAVE BUSTERS, LLC 1998-09-24 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.