SUTURE LOCK
Retention Device, Suture
SMITH & NEPHEW ENDOSCOPY, INC.
The following data is part of a premarket notification filed by Smith & Nephew Endoscopy, Inc. with the FDA for Suture Lock.
Pre-market Notification Details
| Device ID | K964935 |
| 510k Number | K964935 |
| Device Name: | SUTURE LOCK |
| Classification | Retention Device, Suture |
| Applicant | SMITH & NEPHEW ENDOSCOPY, INC. 130 FORBES BLVD. Mansfield, MA 02048 |
| Contact | Susan A Finneran |
| Correspondent | Susan A Finneran SMITH & NEPHEW ENDOSCOPY, INC. 130 FORBES BLVD. Mansfield, MA 02048 |
| Product Code | KGS |
| CFR Regulation Number | 878.4930 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 1996-12-10 |
| Decision Date | 1997-02-25 |
| Summary: | summary |
Trademark Results [SUTURE LOCK]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SUTURE LOCK 98021258 not registered Live/Pending |
Universal Tech Corporation 2023-05-31 |
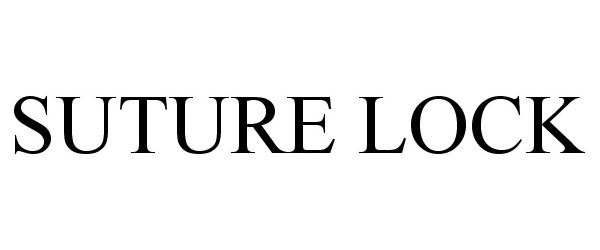 SUTURE LOCK 86637156 not registered Dead/Abandoned |
JULVIA TECHNOLOGIES INC. 2015-05-21 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.