CO2 PNEUMO-DISSECTOR
Laparoscope, Gynecologic (and Accessories)
COOK UROLOGICAL, INC.
The following data is part of a premarket notification filed by Cook Urological, Inc. with the FDA for Co2 Pneumo-dissector.
Pre-market Notification Details
| Device ID | K972647 |
| 510k Number | K972647 |
| Device Name: | CO2 PNEUMO-DISSECTOR |
| Classification | Laparoscope, Gynecologic (and Accessories) |
| Applicant | COOK UROLOGICAL, INC. 1100 WEST MORGAN ST. P.O. BOX 227 Spencer, IN 47460 |
| Contact | Tammy Bacon |
| Correspondent | Tammy Bacon COOK UROLOGICAL, INC. 1100 WEST MORGAN ST. P.O. BOX 227 Spencer, IN 47460 |
| Product Code | HET |
| CFR Regulation Number | 884.1720 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 1997-07-15 |
| Decision Date | 1998-04-10 |
| Summary: | summary |
Trademark Results [CO2 PNEUMO-DISSECTOR]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
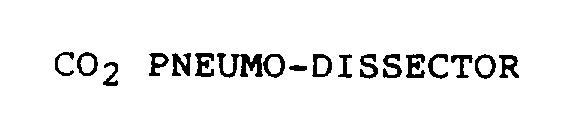 CO2 PNEUMO-DISSECTOR 75211779 not registered Dead/Abandoned |
Vance Products Incorporated 1996-12-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.