CLICKER
Injector, Fluid, Non-electrically Powered
BIOJECT, INC.
The following data is part of a premarket notification filed by Bioject, Inc. with the FDA for Clicker.
Pre-market Notification Details
| Device ID | K994384 |
| 510k Number | K994384 |
| Device Name: | CLICKER |
| Classification | Injector, Fluid, Non-electrically Powered |
| Applicant | BIOJECT, INC. 7620 S.W. BRIDGEPORT RD. Portland, OR 97224 |
| Contact | Nancy Gertlar |
| Correspondent | Nancy Gertlar BIOJECT, INC. 7620 S.W. BRIDGEPORT RD. Portland, OR 97224 |
| Product Code | KZE |
| CFR Regulation Number | 880.5430 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 1999-12-28 |
| Decision Date | 2000-06-22 |
| Summary: | summary |
Trademark Results [CLICKER]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
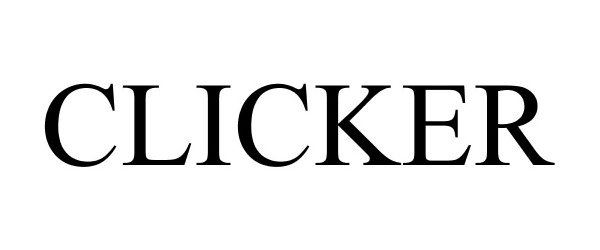 CLICKER 86850783 5251832 Live/Registered |
K2 SPORTS, LLC 2015-12-16 |
 CLICKER 86220200 not registered Dead/Abandoned |
Global Tobacco LLC 2014-03-13 |
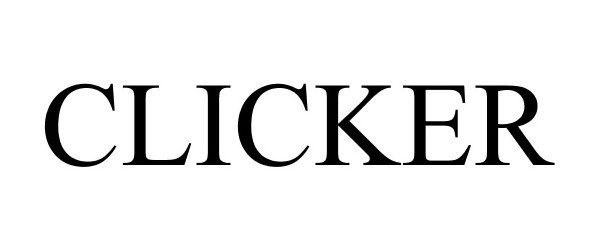 CLICKER 86150339 not registered Dead/Abandoned |
No-Spill Systems, Inc. 2013-12-21 |
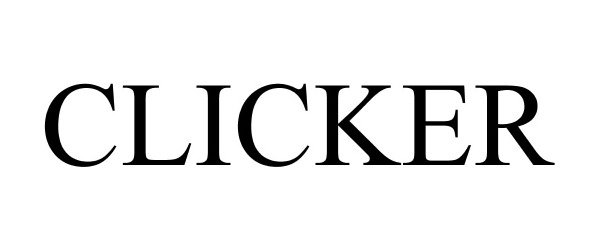 CLICKER 86082153 not registered Dead/Abandoned |
Global Tobacco LLC 2013-10-03 |
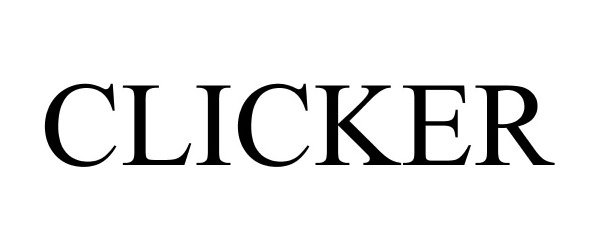 CLICKER 85698219 not registered Dead/Abandoned |
National Cycle, Inc. 2012-08-08 |
 CLICKER 85413406 not registered Dead/Abandoned |
K-2 Corporation 2011-09-01 |
 CLICKER 79182674 not registered Dead/Abandoned |
MIKROELEKTRONIKA DOO BEOGRAD (ZEMUN) 2015-12-02 |
 CLICKER 79016850 not registered Dead/Abandoned |
Laerdal Medical AS 2005-05-18 |
 CLICKER 78846810 not registered Dead/Abandoned |
Calderon, Isaac 2006-03-27 |
 CLICKER 77968989 not registered Dead/Abandoned |
The Clicker Company, LLC 2010-03-25 |
 CLICKER 77564807 3608582 Live/Registered |
THOMPSON, Greg 2008-09-08 |
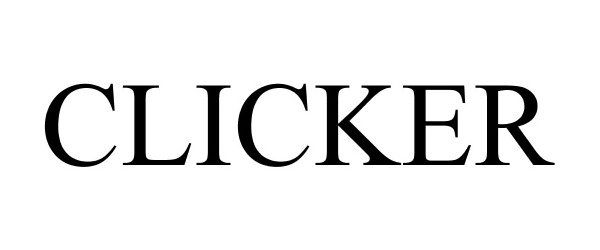 CLICKER 77299625 not registered Dead/Abandoned |
Dignam, David 2007-10-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.