METHYLPHENIDATE HYDROCHLORIDE (LA)- methylphenidate hydrochloride capsule, extended release
Methylphenidate Hydrochloride by
Drug Labeling and Warnings
Methylphenidate Hydrochloride by is a Prescription medication manufactured, distributed, or labeled by Teva Pharmaceuticals USA, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use METHYLPHENIDATE HYDROCHLORIDE EXTENDED-RELEASE CAPSULES LA safely and effectively. See full prescribing information for METHYLPHENIDATE HYDROCHLORIDE EXTENDED-RELEASE CAPSULES LA.
METHYLPHENIDATE HYDROCHLORIDE extended-release capsules for oral use, CII
Initial U.S. Approval: 1955WARNING: ABUSE AND DEPENDENCE
See full prescribing information for complete boxed warning.
- CNS stimulants, including methylphenidate hydrochloride extended-release capsules, other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence (5.1, 9.2, 9.3).
- Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy (5.1, 9.2).
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
Methylphenidate hydrochloride extended-release capsules are a central nervous system (CNS) stimulant indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in pediatric patients 6 to 12 years of age. (1)
DOSAGE AND ADMINISTRATION
- Administer orally once daily in the morning. (2.2)
- Capsules may be swallowed whole, or opened and the entire contents sprinkled on applesauce. (2.2)
- Should not be crushed, chewed, or divided. (2.2)
- Patients new to methylphenidate: Start at 20 mg daily, titrating the dose weekly in 10-mg increments. Doses above 60 mg daily are not recommended. (2.3)
- For patients currently using methylphenidate hydrochloride tablets or methylphenidate hydrochloride extended-release tablets: Dosage is based on current dose regimen. (2.4)
- If switching from other methylphenidate products, discontinue treatment and titrate with methylphenidate hydrochloride extended-release capsules. (2.4)
DOSAGE FORMS AND STRENGTHS
Extended-release capsules: 10 mg, 20 mg, 30 mg, and 40 mg (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Serious Cardiovascular Events: Sudden death has been reported in association with CNS-stimulant treatment at usual doses in pediatric patients with structural cardiac abnormalities or other serious heart problems. In adults, sudden death, stroke, and myocardial infarction have been reported. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm arrhythmias, or coronary artery disease. (5.2)
- Blood Pressure and Heart Rate Increases: Monitor blood pressure and pulse. Consider the benefits and risk in patients for whom an increase in blood pressure or heart rate would be problematic. (5.3)
- Psychiatric Adverse Reactions: Use of stimulants may cause psychotic or manic symptoms in patients with no prior history or exacerbation of symptoms in patients with preexisting psychiatric illness. Evaluate for preexisting psychotic or bipolar disorder prior to methylphenidate hydrochloride extended-release capsules use. (5.4)
- Priapism: Cases of painful and prolonged penile erections, and priapism have been reported with methylphenidate products. Immediate medical attention should be sought if signs or symptoms of prolonged penile erections or priapism are observed. (5.5)
- Peripheral Vasculopathy, including Raynaud’s Phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants. (5.6)
- Long-Term Suppression of Growth: Monitor height and weight at appropriate intervals in pediatric patients. (5.7)
ADVERSE REACTIONS
Most common adverse reactions (greater than 5% during incidence) were headache, insomnia, upper abdominal pain, decreased appetite, and anorexia. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Teva Pharmaceuticals USA, Inc. at 1-888-838-2872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 12/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: ABUSE AND DEPENDENCE
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Pretreatment Screening
2.2 General Dosing Information
2.3 Patients Currently Using Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets
2.4 Switching from other Methylphenidate Products
2.5 Dose Reduction and Discontinuation
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Potential for Abuse and Dependence
5.2 Serious Cardiovascular Reactions
5.3 Blood Pressure and Heart Rate Increases
5.4 Psychiatric Adverse Reactions
5.5 Priapism
5.6 Peripheral Vasculopathy, Including Raynaud’s Phenomenon
5.7 Long-Term Suppression of Growth
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Clinically Important Interactions with Methylphenidate Hydrochloride Extended-Release Capsules
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3 Dependence
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
14 CLINICAL STUDIES
14.1 Children and Adolescents
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: ABUSE AND DEPENDENCE
CNS stimulants, including methylphenidate hydrochloride extended-release capsules, other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)].
-
1 INDICATIONS AND USAGE
Methylphenidate hydrochloride extended-release capsules are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD), in pediatric patients 6 to 12 years of age [see Clinical Studies (14)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Pretreatment Screening
Prior to initiating treatment with central nervous system (CNS) stimulants, including methylphenidate hydrochloride extended-release capsules, assess for the presence of cardiac disease (i.e., perform a careful history including family history of sudden death or ventricular arrhythmia, and physical examination) [see Warnings and Precautions (5.2)].
Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy. Maintain careful prescription records, educate patients about abuse, monitor for signs of abuse and overdose, and periodically reevaluate the need for methylphenidate hydrochloride extended-release capsules use [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9)].
2.2 General Dosing Information
The recommended starting dose for methylphenidate hydrochloride extended-release capsules is 20 mg once daily. Increase dosage gradually, in increments of 10 mg weekly. Daily dosage above 60 mg is not recommended. When a lower initial dose is appropriate, patients may begin treatment with 10 mg.
Administer methylphenidate hydrochloride extended-release capsules orally once daily in the morning. Methylphenidate hydrochloride extended-release capsules may be swallowed as whole capsules or may be administered by sprinkling the capsule contents on a small amount of applesauce (see specific instructions below). Methylphenidate hydrochloride extended-release capsules and/or their contents should not be crushed, chewed, or divided.
The capsules may be carefully opened and the beads sprinkled over a spoonful of applesauce. The applesauce should not be warm because it could affect the modified release properties of this formulation. The mixture of drug and applesauce should be consumed immediately in its entirety. The drug and applesauce mixture should not be stored for future use.
Pharmacological treatment of ADHD may be needed for extended periods. Periodically reevaluate the long-term use of methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets, and adjust dosage as needed.
2.3 Patients Currently Using Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets
The recommended dose of methylphenidate hydrochloride extended-release capsules for patients currently taking methylphenidate hydrochloride tablets twice daily or methylphenidate hydrochloride extended-release (SR) is provided below.
Table 1: Recommended Dose Conversion from Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets
Previous Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets Dose Recommended Methylphenidate Hydrochloride Extended-Release Capsules Dose 5 mg methylphenidate hydrochloride tablets twice daily 10 mg once daily 10 mg methylphenidate hydrochloride tablets twice daily or 20 mg methylphenidate hydrochloride extended-release tablets 20 mg once daily 15 mg methylphenidate hydrochloride tablets twice daily 30 mg once daily 20 mg methylphenidate hydrochloride tablets twice daily or 40 mg methylphenidate hydrochloride extended-release tablets 40 mg once daily 30 mg methylphenidate hydrochloride tablets twice daily or 60 mg methylphenidate hydrochloride extended-release tablets 60 mg once daily 2.4 Switching from other Methylphenidate Products
If switching from other methylphenidate products, discontinue that treatment, and titrate with methylphenidate hydrochloride extended-release capsules using the titration schedule.
Do not substitute for other methylphenidate products on a milligram-per-milligram basis, because different methylphenidate base compositions and differing pharmacokinetic profiles [see Description (11), Clinical Pharmacology (12.3)].
Clinical judgment should be used when selecting the starting dose. Daily dosage above 60 mg is not recommended.
2.5 Dose Reduction and Discontinuation
If paradoxical worsening of symptoms or other adverse reactions occur, reduce the dosage, or, if necessary, discontinue methylphenidate hydrochloride extended-release capsules. If improvement is not observed after appropriate dosage adjustment over a one-month period, the drug should be discontinued.
-
3 DOSAGE FORMS AND STRENGTHS
10 mg extended-release capsules light brown opaque cap and white opaque body, imprinted TEVA on the cap and 5345 on the body
20 mg extended-release capsules white opaque cap and white opaque body, imprinted TEVA on the cap and 5346 on the body
30 mg extended-release capsules ivory opaque cap and ivory opaque body, imprinted TEVA on the cap and 5347 on the body
40 mg extended-release capsules light brown opaque cap and light brown opaque body, imprinted TEVA on the cap and 5348 on the body
-
4 CONTRAINDICATIONS
- Hypersensitivity to methylphenidate or other components of methylphenidate hydrochloride extended-release capsules. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate [see Adverse Reactions (6.1)].
- Concomitant treatment with monoamine oxidase inhibitors (MAOIs), or within 14 days following discontinuation of treatment with an MAOI, because of the risk of hypertensive crises [see Drug Interactions (7.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Potential for Abuse and Dependence
CNS stimulants, including methylphenidate hydrochloride extended-release capsules, other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Boxed Warning, Drug Abuse and Dependence (9.2, 9.3)].
5.2 Serious Cardiovascular Reactions
Sudden death, stroke and myocardial infarction have been reported in adults with CNS-stimulant treatment at recommended doses. Sudden death has been reported in pediatric patients with structural cardiac abnormalities and other serious heart problems taking CNS stimulants at recommended doses for ADHD. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, and other serious heart problems. Further evaluate patients who develop exertional chest pain, unexplained syncope, or arrhythmias during methylphenidate hydrochloride extended-release capsules treatment.
5.3 Blood Pressure and Heart Rate Increases
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm). Individuals may have larger increases. Monitor all patients for hypertension and tachycardia.
5.4 Psychiatric Adverse Reactions
Exacerbation of Preexisting Psychosis
CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a preexisting psychotic disorder.
Induction of a Manic Episode in Patients with Bipolar Disorder
CNS stimulants may induce a manic or mixed mood episode in patients. Prior to initiating treatment, screen patients for risk factors for developing a manic episode (e.g., comorbid or history of depressive symptoms or a family history of suicide, bipolar disorder, or depression).
New Psychotic or Manic Symptoms
CNS stimulants, at recommended doses, may cause psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) in patients without a prior history of psychotic illness or mania. If such symptoms occur, consider discontinuing methylphenidate hydrochloride extended-release capsules. In a pooled analysis of multiple short-term, placebo-controlled studies of CNS stimulants, psychotic or manic symptoms occurred in approximately 0.1% of CNS stimulant-treated patients, compared to 0 in placebo-treated patients.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate products in both pediatric and adult patients. Priapism was not reported with drug initiation but developed after some time on the drug, often subsequent to an increase in dose. Priapism has also appeared during a period of drug withdrawal (drug holidays or during discontinuation). Patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
5.6 Peripheral Vasculopathy, Including Raynaud’s Phenomenon
CNS stimulants, including methylphenidate hydrochloride extended-release capsules, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in postmarketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
5.7 Long-Term Suppression of Growth
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated patients over 36 months (to the ages of 10 to 13 years), suggests that consistently medicated pediatric patients (i.e., treatment for 7 days per week throughout the year) have a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this period of development.
Closely monitor growth (weight and height) in pediatric patients treated with CNS stimulants, including methylphenidate hydrochloride extended-release capsules. Patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
-
6 ADVERSE REACTIONS
The following are discussed in more detail in other sections of the labeling:
- Abuse and Dependence [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)]
- Known hypersensitivity to methylphenidate or other ingredients of methylphenidate hydrochloride extended-release capsules [see Contraindications (4)]
- Hypertensive crisis when used concomitantly with monoamine oxidase inhibitors [see Contraindications (4), Drug Interactions (7.1)]
- Serious cardiovascular reactions [see Warnings and Precautions (5.2)]
- Blood pressure and heart rate increases [see Warnings and Precautions (5.3)]
- Psychiatric adverse reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral vasculopathy, including Raynaud’s phenomenon [see Warnings and Precautions (5.6)]
- Long-term suppression of growth [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The clinical program for methylphenidate hydrochloride extended-release capsules consisted of 6 studies: 2 controlled clinical studies conducted in children with ADHD aged 6 to 12 years and 4 clinical pharmacology studies conducted in healthy adult volunteers. These studies included a total of 256 subjects; 195 children with ADHD and 61 healthy adult volunteers. The subjects received methylphenidate hydrochloride extended-release capsules in doses of 10 to 40 mg per day. Safety of methylphenidate hydrochloride extended-release capsules was assessed by evaluating frequency and nature of adverse events, routine laboratory tests, vital signs, and body weight. A placebo-controlled, double-blind, parallel-group study was conducted to evaluate the efficacy and safety of methylphenidate hydrochloride extended-release capsules in children with ADHD aged 6 to 12 years. All subjects received methylphenidate hydrochloride extended-release capsules for up to 4 weeks, and had their dose optimally adjusted, prior to entering the double-blind phase of the trial. In the 2-week double-blind treatment phase of this study, patients received either placebo or methylphenidate hydrochloride extended-release capsules at their individually-titrated dose (range 10 mg to 40 mg).
Adverse reactions with an incidence greater than 5% during the initial 4-week single-blind methylphenidate hydrochloride extended-release capsule titration period of this study were headache, insomnia, upper abdominal pain, appetite decreased, and anorexia.
Adverse reactions with an incidence greater than 2% among methylphenidate hydrochloride extended-release capsule-treated subjects, during the 2-week double-blind phase of the clinical study, are shown in Table 2.
Table 2: Adverse Reactions in Greater Than 2% Methylphenidate Hydrochloride Extended-Release Capsule-Treated Subjects in the 2-Week Double-Blind Phase
Preferred Term Methylphenidate Hydrochloride Extended-Release Capsules
N = 65
N (%)Placebo
N = 71
N (%)Anorexia 2 (3.1) 0 (0.0) Insomnia 2 (3.1) 0 (0.0) Adverse Events Associated with Discontinuation of Treatment
In the 2-week double-blind treatment phase of a placebo-controlled parallel-group study in children with ADHD, one methylphenidate hydrochloride extended-release capsule-treated subject (1/65, 1.5%) discontinued due to an adverse event (depressed mood).
In the single-blind titration period of this study, subjects received methylphenidate hydrochloride extended-release capsules for up to 4 weeks. During this period a total of 6 subjects (6/161, 3.7%) discontinued due to adverse events. The adverse events leading to discontinuation were anger (2 patients), hypomania, anxiety, depressed mood, fatigue, migraine and lethargy.
6.2 Postmarketing Experience
The following adverse reactions have been identified during the post approval use of methylphenidate products. Because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
Adverse Reactions Reported with Methylphenidate Hydrochloride Tablets, Methylphenidate Hydrochloride Extended-Release Tablets, and Methylphenidate Hydrochloride Extended-Release Capsules
Infections and Infestations: nasopharyngitis
Blood and the Lymphatic System Disorders: leukopenia, thrombocytopenia, anemia
Immune System Disorders: hypersensitivity reactions, including angioedema and anaphylaxis
Metabolism and Nutrition Disorders: decreased appetite, reduced weight gain, and suppression of growth during prolonged use in children
Psychiatric Disorders: insomnia, anxiety, restlessness, agitation, psychosis (sometimes with visual and tactile hallucinations), depressed mood
Nervous System Disorders: headache, dizziness, tremor, dyskinesia including choreoathetoid movements, drowsiness, convulsions, cerebrovascular disorders (including vasculitis, cerebral hemorrhages and cerebrovascular accidents), serotonin syndrome in combination with serotonergic drugs
Eye Disorders: blurred vision, difficulties in visual accommodation
Cardiac Disorders: tachycardia, palpitations, increased blood pressure, arrhythmias, angina pectoris
Respiratory, Thoracic and Mediastinal Disorders: cough
Gastrointestinal Disorders: dry mouth, nausea, vomiting, abdominal pain, dyspepsia
Hepatobiliary Disorders: abnormal liver function, ranging from transaminase elevation to severe hepatic injury
Skin and Subcutaneous Tissue Disorders: hyperhidrosis, pruritus, urticaria, exfoliative dermatitis, scalp hair loss, erythema multiforme rash, thrombocytopenic purpura
Musculoskeletal and Connective Tissue Disorders: arthralgia, muscle cramps, rhabdomyolysis
Investigations: weight loss (adult ADHD patients)
Adverse Reactions Reported with Other Methylphenidate-Containing Products
The list below shows adverse reactions not listed with methylphenidate hydrochloride tablets, methylphenidate hydrochloride extended-release tablets, or methylphenidate hydrochloride extended-release capsules formulations that have been reported with other methylphenidate-containing products.
Blood and Lymphatic Disorders: pancytopenia
Immune System Disorders: hypersensitivity reactions such as auricular swelling, bullous conditions, eruptions, exanthemas
Psychiatric Disorders: affect lability, mania, disorientation, libido changes
Nervous System disorders: migraine
Eye Disorders: diplopia, mydriasis
Cardiac Disorders: sudden cardiac death, myocardial infarction, bradycardia, extrasystole
Vascular Disorders: peripheral coldness, Raynaud's phenomenon
Respiratory, Thoracic and Mediastinal Disorders: pharyngolaryngeal pain, dyspnea
Gastrointestinal Disorders: diarrhea, constipation
Skin and Subcutaneous Tissue Disorders: angioneurotic edema, erythema, fixed drug eruption
Musculoskeletal, Connective Tissue and Bone Disorders: myalgia, muscle twitching
Renal and Urinary Disorders: hematuria
Reproductive System and Breast Disorders: gynecomastia
General Disorders: fatigue, hyperpyrexia
Urogenital Disorders: priapism
-
7 DRUG INTERACTIONS
7.1 Clinically Important Interactions with Methylphenidate Hydrochloride Extended-Release Capsules
Table 3 presents clinically important drug interactions with methylphenidate hydrochloride extended-release capsules.
Table 3: Drugs Having Clinically Important Drug Interactions with Methylphenidate Hydrochloride Extended-Release Capsules
Monoamine Oxidase Inhibitors (MAOI)
Clinical Impact
Concomitant use of MAOIs and CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure [see Contraindications (4)].
Intervention
Concomitant use of methylphenidate hydrochloride extended-release capsules with MAOIs or within 14 days after discontinuing MAOI treatment is contraindicated.
Examples
selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue
Antihypertensive Drugs
Clinical Impact
Methylphenidate hydrochloride extended-release capsules may decrease the effectiveness of drugs used to treat hypertension [see Warnings and Precautions (5.3)].
Intervention
Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed.
Examples
Potassium-sparing and thiazide diuretics, calcium channel blockers, ACE inhibitors, ARBs, beta blockers, centrally acting alpha-2 receptor agonists.
Halogenated Anesthetics
Clinical Impact
Concomitant use of halogenated anesthetics and methylphenidate hydrochloride extended-release capsules may increase the risk of sudden blood pressure and heart rate increase during surgery.
Intervention
Avoid use of methylphenidate hydrochloride extended-release capsules in patients being treated with anesthetics on the day of surgery.
Examples
halothane, isoflurane, enflurane, desflurane, sevoflurane
-
8 USE IN SPECIFIC POPULATIONS
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ADHD medications, including methylphenidate hydrochloride extended-release capsules during pregnancy. Healthcare providers are encouraged to register patients by calling the National Pregnancy registry for ADHD medications at 1-866-961-2388 or visit https://womensmentalhealth.org/adhd-medications/.8.1 Pregnancy
Risk Summary
Published studies and postmarketing reports on methylphenidate use during pregnancy have not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. There may be risks to the fetus associated with the use of CNS stimulants use during pregnancy (see Clinical Considerations). No effects on morphological development were observed in embryo-fetal development studies with oral administration of methylphenidate to pregnant rats and rabbits during organogenesis at doses up to 10 and 15 times, respectively, the maximum recommended human dose (MRHD) of 60 mg/day given to adolescents on a mg/m2 basis. However, spina bifida was observed in rabbits at a dose 52 times the MRHD given to adolescents. A decrease in pup body weight was observed in a pre- and post-natal development study with oral administration of methylphenidate to rats throughout pregnancy and lactation at doses 6 times the MRHD given to adolescents (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
CNS stimulants such as methylphenidate hydrochloride extended-release capsules, can cause vasoconstriction and thereby decrease placental perfusion. No fetal and/or neonatal adverse reactions have been reported with the use of therapeutic doses of methylphenidate during pregnancy; however, premature delivery and low birth weight infants have been reported in amphetamine-dependent mothers.
Data
Animal Data
In embryo-fetal development studies conducted in rats and rabbits, methylphenidate was administered orally at doses of up to 75 and 200 mg/kg/day, respectively, during the period of organogenesis. Malformations (increased incidence of fetal spina bifida) were observed in rabbits at the highest dose, which is approximately 52 times the MRHD of 60 mg/day given to adolescents on a mg/m2 basis. The no effect level for embryo-fetal development in rabbits was 60 mg/kg/day (15 times the MRHD given to adolescents on a mg/m2 basis). There was no evidence of morphological development effects in rats, although increased incidences of fetal skeletal variations were seen at the highest dose level (10 times the MRHD of 60 mg/day given to adolescents on a mg/m2 basis), which was also maternally toxic. The no effect level for embryo-fetal development in rats was 25 mg/kg/day (3 times the MRHD on a mg/m2 basis). When methylphenidate was administered to rats throughout pregnancy and lactation at doses of up to 45 mg/kg/day, offspring body weight gain was decreased at the highest dose (6 times the MRHD of 60 mg/day given to adolescents on a mg/m2 basis), but no other effects on postnatal development were observed. The no effect level for pre- and postnatal development in rats was 15 mg/kg/day (~2 times the MRHD given to adolescents on a mg/m2 basis).
8.2 Lactation
Risk Summary
Limited published literature, based on milk sampling from seven mothers reports that methylphenidate is present in human milk, which resulted in infant doses of 0.16% to 0.7% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 1.1 and 2.7. There are no reports of adverse effects on the breastfed infant and no effects on milk production. Long-term neurodevelopmental effects on infants from stimulant exposure are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for methylphenidate hydrochloride extended-release capsules and any potential adverse effects on the breastfed infant from methylphenidate hydrochloride extended-release capsules or from the underlying maternal condition.
Clinical Considerations
Monitor breastfeeding infants for adverse reactions, such as agitation, insomnia, anorexia, and reduced weight gain.
8.4 Pediatric Use
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules for the treatment of ADHD have been established in pediatric patients 6 to 12 years.
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules in pediatric patients less than 6 years have not been established. The long- term efficacy of methylphenidate in pediatric patients has not been established.
Long-Term Suppression of Growth
Growth should be monitored during treatment with stimulants, including methylphenidate hydrochloride extended-release capsules. Pediatric patients who are not growing or gaining weight as expected may need to have their treatment interrupted [see Warnings and Precautions (5.7)].
Juvenile Animal Toxicity Data
Rats treated with methylphenidate early in the postnatal period through sexual maturation demonstrated a decrease in spontaneous locomotor activity in adulthood. A deficit in acquisition of a specific learning task was observed in females only. The doses at which these findings were observed are at least 4 times the MRHD of 60 mg/day given to children on a mg/m2 basis.
In a study conducted in young rats, methylphenidate was administered orally at doses of up to 100 mg/kg/day for 9 weeks, starting early in the postnatal period (postnatal Day 7) and continuing through sexual maturity (postnatal Week 10). When these animals were tested as adults (postnatal Weeks 13 to 14), decreased spontaneous locomotor activity was observed in males and females previously treated with 50 mg/kg/day (approximately 4 times the MRHD of 60 mg/day given to children on a mg/m2 basis) or greater, and a deficit in the acquisition of a specific learning task was seen in females exposed to the highest dose (8 times the MRHD given to children on a mg/m2 basis). The no effect level for juvenile neurobehavioral development in rats was 5 mg/kg/day (approximately 0.5 times the MRHD given to children on a mg/m2 basis). The clinical significance of the long-term behavioral effects observed in rats is unknown.
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Methylphenidate hydrochloride extended-release capsules contain methylphenidate hydrochloride, a Schedule II controlled substance.
9.2 Abuse
CNS stimulants, including methylphenidate hydrochloride extended-release capsules, have a high potential for abuse. Abuse is characterized by impaired control over drug use despite harm, and craving.
Signs and symptoms of CNS stimulant abuse include increased heart rate, respiratory rate, blood pressure, and/or sweating, dilated pupils, hyperactivity, restlessness, insomnia, decreased appetite, loss of coordination, tremors, flushed skin, vomiting, and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed. Abusers of CNS stimulants may chew, snort, inject, or use other unapproved routes of administration which may result in overdose and death [see Overdosage (10)].
To reduce the abuse of CNS stimulants including methylphenidate hydrochloride extended-release capsules, assess the risk of abuse prior to prescribing. After prescribing, keep careful prescription records, educate patients and their families about abuse and on proper storage and disposal of CNS stimulants [see How Supplied/Storage and Handling (16)], monitor for signs of abuse while on therapy, and reevaluate the need for methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets use.
9.3 Dependence
Tolerance
Tolerance (a state of adaptation in which exposure to a drug results in a reduction of the drug’s desired and/or undesired effects over time) can occur during chronic therapy with CNS stimulants, including methylphenidate hydrochloride extended-release capsules.
Dependence
Physical dependence (which is manifested by a withdrawal syndrome produced by abrupt cessation, rapid dose reduction, or administration of an antagonist) may occur in patients treated with CNS stimulants including methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets. Withdrawal symptoms after abrupt cessation following prolonged high-dosage administration of CNS stimulants include dysphoric mood; fatigue; vivid, unpleasant dreams; insomnia or hypersomnia; increased appetite; and psychomotor retardation or agitation.
-
10 OVERDOSAGE
Human Experience
Signs and symptoms of acute overdosage, resulting principally from overstimulation of the central nervous system and from excessive sympathomimetic effects, may include the following: nausea, vomiting, diarrhea, restlessness, anxiety, agitation, tremors, hyperreflexia, muscle twitching, convulsions (which may be followed by coma), euphoria, confusion, hallucinations, delirium, sweating, flushing, headache, hyperpyrexia, tachycardia, palpitations, cardiac arrhythmias, hypertension, hypotension, tachypnea, mydriasis, dryness of mucous membranes, and rhabdomyolysis.
Overdose Management
Consult with a Certified Poison Control Center (1-800-222-1222) for the latest recommendations.
-
11 DESCRIPTION
Methylphenidate Hydrochloride Extended-Release Capsules contain methylphenidate hydrochloride, USP, a CNS stimulant.
Methylphenidate Hydrochloride Extended-Release Capsules are an extended-release formulation of methylphenidate for oral administration with a bi-modal release profile. Each Methylphenidate Hydrochloride Extended-Release Capsule contains pellets, with half the dose as immediate-release and half as delayed-release, thus providing an immediate release of methylphenidate and a second delayed release of methylphenidate.
The active substance in Methylphenidate Hydrochloride Extended-Release Capsules is methyl α-phenyl-2-piperidineacetate hydrochloride. Its structural formula is as follows:
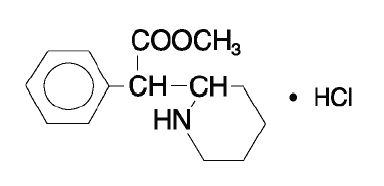
M.W. 269.77
Methylphenidate hydrochloride, USP is a white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone.
Each capsule, for oral administration, has the following inactive ingredients: ammonio methacrylate copolymer, black iron oxide, D&C Yellow #10 Aluminum Lake, FD&C Blue #1 Aluminum Lake, FD&C Blue #2 Aluminum Lake, FD&C Red #40 Aluminum Lake, gelatin, hypromellose, methacrylic acid copolymer, polyethylene glycol, propylene glycol, shellac glaze (modified), sugar spheres, talc, titanium dioxide and triethyl citrate. The 10 mg and 40 mg capsule shell contains FD&C blue no. 1, FD&C red no. 40 and FD&C yellow no. 6. The 30 mg capsule shell contains D&C red no. 33 and D&C yellow no. 10.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Methylphenidate hydrochloride is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d- and l-threo enantiomers. The d-threo enantiomer is more pharmacologically active than the l-threo enantiomer. Methylphenidate blocks the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
Cardiac Electrophysiology
A formal QT study has not been conducted in patients taking methylphenidate hydrochloride extended-release capsules.
The effect of dexmethylphenidate, the pharmacologically active d-enantiomer of methylphenidate hydrochloride, on the QT interval was evaluated in a double-blind, placebo- and open-label active (moxifloxacin)-controlled study following single doses of dexmethylphenidate XR 40 mg (maximum recommended adult total daily dosage) in 75 healthy volunteers. ECGs were collected up to 12 hours postdose. Frederica’s method for heart rate correction was employed to derive the corrected QT interval (QTcF). The maximum mean prolongation of QTcF intervals was less than 5 ms, and the upper limit of the 90% confidence interval (CI) was below 10 ms for all time matched comparisons versus placebo. This was below the threshold of clinical concern and there was no evident-exposure response relationship.
12.3 Pharmacokinetics
Methylphenidate hydrochloride extended-release capsules produce a bi-modal plasma concentration-time profile (i.e., 2 distinct peaks approximately 4 hours apart) when administered orally to children diagnosed with ADHD and healthy adults.
No accumulation of methylphenidate is expected following multiple once daily oral dosing with methylphenidate hydrochloride extended-release capsules, however, there is a slight upward trend in the methylphenidate area under the curve (AUC) and peak plasma concentrations (Cmax1 and Cmax2) after oral administration of methylphenidate hydrochloride extended-release 20 mg and 40 mg capsules to adults.
Absorption
The absolute oral bioavailability of methylphenidate in children was 22 ± 8% for d-methylphenidate and 5 ± 3% for l- methylphenidate. The relative bioavailability of methylphenidate hydrochloride extended-release capsules given once daily is comparable to the same total dose of methylphenidate hydrochloride tablets given in 2 doses 4 hours apart in both children and adults.
The initial rate of absorption for methylphenidate hydrochloride extended-release capsules are similar to that of methylphenidate hydrochloride tablets as shown by the similar rate parameters between the 2 formulations, i.e., initial lag time (Tlag), first peak concentration (Cmax1), and time to the first peak (Tmax1), which is reached in 1 to 3 hours. The mean time to the interpeak minimum (Tminip), and time to the second peak (Tmax2) are also similar for methylphenidate hydrochloride extended-release capsules given once daily and methylphenidate hydrochloride tablets given in 2 doses 4 hours apart (see Figure 1 and Table 1), although the ranges observed are greater for methylphenidate hydrochloride extended-release capsules.
Methylphenidate hydrochloride extended-release capsules given once daily exhibits a lower second peak concentration (Cmax2), higher interpeak minimum concentrations (Cminip), and less peak and trough fluctuations than methylphenidate hydrochloride tablets given in 2 doses given 4 hours apart. This is due to an earlier onset and more prolonged absorption from the delayed-release beads (see Figure 1 and Table 4).
Figure 1: Mean Plasma Concentration Time-Profile of Methylphenidate After a Single Dose of Methylphenidate Hydrochloride Extended-Release Capsules 40 mg and Methylphenidate Hydrochloride Tablets 20 mg Given in Two Doses 4 Hours Apart
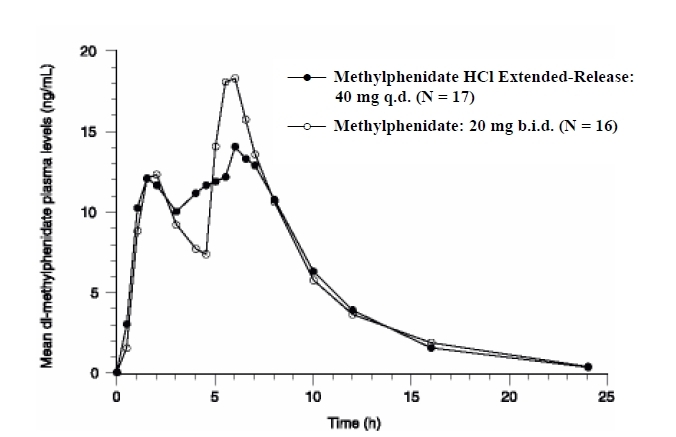
Table 4: Mean ± SD and Range of Pharmacokinetic Parameters of Methylphenidate After a Single Dose of Methylphenidate Hydrochloride Extended-Release Capsules and Methylphenidate Hydrochloride Tablets Given in Two Doses 4 Hours Apart
Population Children Adult Males Formulation
Dose
N
Methylphenidate Hydrochloride
Tablets
10 mg & 10 mg
21
Methylphenidate
Hydrochloride
Extended-Release Capsules
20 mg
18
Methylphenidate Hydrochloride
Tablets
10 mg & 10 mg
9
Methylphenidate
Hydrochloride
Extended-Release Capsules
20 mg
8Tlag (h) 0.24 ± 0.44
0 to 10.28 ± 0.46
0 to 11.0 ± 0.5
0.7 to 1.30.7 ± 0.2
0.3 to 1.0Tmax1 (h) 1.8 ± 0.6
1 to 32.0 ± 0.8
1 to 31.9 ± 0.4
1.3 to 2.72.0 ± 0.9
1.3 to 4.0Cmax1 (ng/mL) 10.2 ± 4.2
4.2 to 20.210.3 ± 5.1
5.5 to 26.64.3 ± 2.3
1.8 to 7.55.3 ± 0.9
3.8 to 6.9Tminip (h) 4.0 ± 0.2
4 to 54.5 ± 1.2
2 to 63.8 ± 0.4
3.3 to 4.33.6 ± 0.6
2.7 to 4.3Cminip (ng/mL) 5.8 ± 2.7
3.1 to 14.46.1 ± 4.1
2.9 to 21.01.2 ± 1.4
0.0 to 3.73.0 ± 0.8
1.7 to 4.0Tmax2 (h) 5.6 ± 0.7
5 to 86.6 ± 1.5
5 to 115.9 ± 0.5
5.0 to 6.55.5 ± 0.8
4.3 to 6.5Cmax2 (ng/mL) 15.3 ± 7.0
6.2 to 32.810.2 ± 5.9
4.5 to 31.15.3 ± 1.4
3.6 to 7.26.2 ± 1.6
3.9 to 8.3AUC(0-∞)
(ng/mL x h-1)102.4 ± 54.6
40.5 to 261.686.6 ± 64.0a
43.3 to 301.4437.8 ± 21.9
14.3 to 85.345.8 ± 10.0
34.0 to 61.6t1/2 (h) 2.5 ± 0.8
1.8 to 5.32.4 ± 0.7a
1.5 to 4.03.5 ± 1.9
1.3 to 7.73.3 ± 0.4
3.0 to 4.2aN = 15.
Effect of Food
Administration times relative to meals and meal composition may need to be individually titrated.
When methylphenidate hydrochloride extended-release capsules were administered with a high fat breakfast to adults, methylphenidate hydrochloride extended-release capsules had a longer lag time until absorption began and variable delays in the time until the first peak concentration, the time until the interpeak minimum, and the time until the second peak. The first peak concentration and the extent of absorption were unchanged after food relative to the fasting state, although the second peak was approximately 25% lower. The effect of a high fat lunch was not examined.
There were no differences in the pharmacokinetics of methylphenidate hydrochloride extended-release capsules when administered with applesauce, compared to administration in the fasting condition. There is no evidence of dose dumping in the presence or absence of food.
Effect of Alcohol
An in vitro study was conducted to explore the effect of alcohol on the release characteristics of methylphenidate from the methylphenidate hydrochloride extended-release capsules 40 mg capsule dosage form. At an alcohol concentration of 40% there was a 98% release of methylphenidate in the first hour. The results with the 40 mg capsule are considered to be representative of the other available capsule strengths.
Distribution
Binding to plasma proteins is low (10% to 33%). The volume of distribution was 2.65 ± 1.11 L/kg for d- methylphenidate and 1.80 ± 0.91 L/kg for l-methylphenidate.
Elimination
The systemic clearance is 0.40 ± 0.12 L/h/kg for d-methylphenidate and 0.73 ± 0.28 L/h/kg for l-methylphenidate. In studies with methylphenidate hydrochloride extended-release capsules and methylphenidate hydrochloride tablets in adults, methylphenidate from methylphenidate hydrochloride tablets is eliminated from plasma with an average half-life of about 3.5 hours, (range 1.3 to 7.7 hours). In children the average half-life is about 2.5 hours, with a range of about 1.5 to 5.0 hours. The rapid half-life in both children and adults may result in un-measurable concentrations between the morning and mid-day doses with methylphenidate hydrochloride tablets. No accumulation of methylphenidate is expected following multiple once a day oral dosing with methylphenidate hydrochloride extended-release capsules. The half-life of ritalinic acid is about 3 to 4 hours.
Metabolism
The absolute oral bioavailability of methylphenidate in children was 22 ± 8% for d-methylphenidate and 5 ± 3% for l- methylphenidate, suggesting pronounced presystemic metabolism. Biotransformation of methylphenidate by the carboxylesterase CES1A1 is rapid and extensive leading to the main, de-esterified metabolite α-phenyl-2-piperidine acetic acid (ritalinic acid), which has little or no pharmacologic activity. Only small amounts of hydroxylated metabolites (e.g., hydroxymethylphenidate and hydroxyritalinic acid) are detectable in plasma. Therapeutic activity is principally due to the parent compound.
Excretion
After oral administration of an immediate release formulation of methylphenidate, 78% to 97% of the dose is excreted in the urine and 1% to 3% in the feces in the form of metabolites within 48 to 96 hours. Only small quantities (less than 1%) of unchanged methylphenidate appear in the urine. Most of the dose is excreted in the urine as ritalinic acid (60% to 86%), the remainder being accounted for by minor metabolite.
Studies in Specific Populations
Male and Female Patients
There were no apparent gender differences in the pharmacokinetics of methylphenidate between healthy male and female adults when administered methylphenidate hydrochloride extended-release capsules.
Racial or Ethnic Groups
There is insufficient experience with the use of methylphenidate hydrochloride extended-release capsules to detect ethnic variations in pharmacokinetics.
Pediatric Patients
The pharmacokinetics of methylphenidate hydrochloride extended-release capsules were examined in 18 children with ADHD between 7 and 12 years of age. Fifteen of these children were between 10 and 12 years of age. The time until the between peak minimum, and the time until the second peak were delayed and more variable in children compared to adults. After a 20-mg dose of methylphenidate hydrochloride extended-release capsules, concentrations in children were approximately twice the concentrations observed in 18 to 35 year old adults. This higher exposure is almost completely due to the smaller body size and total volume of distribution in children, as apparent clearance normalized to body weight is independent of age.
Patients with Renal Impairment
Methylphenidate hydrochloride extended-release capsules have not been studied in renally-impaired patients. Renal impairment is expected to have minimal effect on the pharmacokinetics of methylphenidate since less than 1% of a radiolabeled dose is excreted in the urine as unchanged compound, and the major metabolite (ritalinic acid), has little or no pharmacologic activity.
Patients with Hepatic Impairment
Methylphenidate hydrochloride extended-release capsules have not been studied in patients with hepatic impairment. Hepatic impairment is expected to have minimal effect on the pharmacokinetics of methylphenidate since it is metabolized primarily to ritalinic acid by nonmicrosomal hydrolytic esterases that are widely distributed throughout the body.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
Carcinogenesis
In a lifetime carcinogenicity study carried out in B6C3F1 mice, methylphenidate caused an increase in hepatocellular adenomas, and in males only, an increase in hepatoblastomas at a daily dose of approximately 60 mg/kg/day. This dose is approximately 2 times the MRHD of 60 mg/day given to children on a mg/m2 basis. Hepatoblastoma is a relatively rare rodent malignant tumor type. There was no increase in total malignant hepatic tumors. The mouse strain used is sensitive to the development of hepatic tumors, and the significance of these results to humans is unknown.Methylphenidate did not cause any increase in tumors in a lifetime carcinogenicity study carried out in F344 rats; the highest dose used was approximately 45 mg/kg/day, which is approximately 4 times the MRHD (children) on a mg/m2 basis.
In a 24-week carcinogenicity study in the transgenic mouse strain p53+/-, which is sensitive to genotoxic carcinogens, there was no evidence of carcinogenicity. Male and female mice were fed diets containing the same concentration of methylphenidate as in the lifetime carcinogenicity study; the high-dose groups were exposed to 60 to 74 mg/kg/day of methylphenidate.
Mutagenesis
Methylphenidate was not mutagenic in the in vitro Ames reverse mutation assay, in the in vitro mouse lymphoma cell forward mutation assay, or in the in vitro chromosomal aberration assay using human lymphocytes. Sister chromatid exchanges and chromosome aberrations were increased, indicative of a weak clastogenic response, in an in vitro assay in cultured Chinese Hamster Ovary (CHO) cells. Methylphenidate was negative in vivo in males and females in the mouse bone marrow micronucleus assay.
Impairment of Fertility
Methylphenidate did not impair fertility in male or female mice that were fed diets containing the drug in an 18-week continuous breeding study. The study was conducted at doses up to 160 mg/kg/day, approximately 10 times the maximum recommended dose of 60 mg/day given to adolescents on a mg/m2 basis.
-
14 CLINICAL STUDIES
14.1 Children and Adolescents
Methylphenidate hydrochloride extended-release capsules were evaluated in a randomized, double-blind, placebo-controlled, parallel group clinical study in which 134 children, ages 6 to 12, with DSM-IV diagnoses of ADHD received a single morning dose of methylphenidate hydrochloride extended-release capsules in the range of 10 to 40 mg/day, or placebo, for up to 2 weeks. The doses used were the optimal doses established in a previous individual dose titration phase. In that titration phase, 53 of 164 patients (32%) started on a daily dose of 10 mg and 111 of 164 patients (68%) started on a daily dose of 20 mg or higher. The patient’s regular schoolteacher completed the Conners ADHD/DSM-IV Scale for Teachers (CADS-T) at baseline and the end of each week. The CADS-T assesses symptoms of hyperactivity and inattention. The change from baseline of the (CADS-T) scores during the last week of treatment was analyzed as the primary efficacy parameter. Patients treated with methylphenidate hydrochloride extended-release capsules showed a statistically significant improvement in symptom scores from baseline [Mean (final score - baseline) = -10.7 points] over patients who received placebo [Mean (final score - baseline) = +2.8 points]. The lower the final score on the CADS-T scale from baseline, the less severe the disease is. This demonstrates that a single morning dose of methylphenidate hydrochloride extended-release capsules exerts a treatment effect in ADHD.
Figure 2: CADS-T Total Subscale - Mean Change From Baseline*

-
16 HOW SUPPLIED/STORAGE AND HANDLING
Methylphenidate Hydrochloride Extended-Release Capsules, 10 mg are available as two-piece, hard gelatin capsules, with light brown opaque cap and white opaque body, filled with white to off-white pellets. Imprinted in black ink TEVA on the cap and 5345 on the body, containing 10 mg of methylphenidate hydrochloride, USP packaged in bottles of 100 capsules (NDC: 0093-5345-01).
Methylphenidate Hydrochloride Extended-Release Capsules, 20 mg are available as two-piece, hard gelatin capsules, with white opaque cap and white opaque body, filled with white to off-white pellets. Imprinted in black ink TEVA on the cap and 5346 on the body, containing 20 mg of methylphenidate hydrochloride, USP packaged in bottles of 100 capsules (NDC: 0093-5346-01).
Methylphenidate Hydrochloride Extended-Release Capsules, 30 mg are available as two-piece, hard gelatin capsules, with ivory opaque cap and ivory opaque body, filled with white to off-white pellets. Imprinted in black ink TEVA on the cap and 5347 on the body, containing 30 mg of methylphenidate hydrochloride, USP packaged in bottles of 100 capsules (NDC: 0093-5347-01).
Methylphenidate Hydrochloride Extended-Release Capsules, 40 mg are available as two-piece, hard gelatin capsules, with light brown opaque cap and light brown opaque body, filled with white to off-white pellets. Imprinted in black ink TEVA on the cap and 5348 on the body, containing 40 mg of methylphenidate hydrochloride, USP packaged in bottles of 100 capsules (NDC: 0093-5348-01).
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Disposal
Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired methylphenidate hydrochloride extended-release capsules by a medicine takeback program or by an authorized collector registered with the Drug Enforcement Administration. If no take-back program or authorized collector is available, mix methylphenidate hydrochloride extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard methylphenidate hydrochloride extended-release capsules in the household trash.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Controlled Substance Status/High Potential for Abuse and DependenceAdvise patients that methylphenidate hydrochloride extended-release capsules are a controlled substance, and they can be abused and lead to dependence. Instruct patients that they should not give methylphenidate hydrochloride extended-release capsules to anyone else. Advise patients to store methylphenidate hydrochloride extended-release capsules in a safe place, preferably locked, to prevent abuse. Advise patients to comply with laws and regulations on drug disposal. Advise patients to dispose of remaining, unused, or expired methylphenidate hydrochloride extended-release capsules by a medicine take-back program if available [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.1, 9.2, 9.3), How Supplied/Storage and Handling (16)].
Serious Cardiovascular Risks
Advise patients that there is a potential serious cardiovascular risk including sudden death, myocardial infarction, stroke, and hypertension with methylphenidate hydrochloride extended-release capsules use. Instruct patients to contact a healthcare provider immediately if they develop symptoms such as exertional chest pain, unexplained syncope, or other symptoms suggestive of cardiac disease [see Warnings and Precautions (5.2)].
Blood Pressure and Heart Rate Increases
Instruct patients that methylphenidate hydrochloride extended-release capsules can cause elevations of their blood pressure and pulse rate [see Warnings and Precautions (5.3)].
Psychiatric Risks
Advise patients that methylphenidate hydrochloride extended-release capsules, at recommended doses, can cause psychotic or manic symptoms, even in patients without prior history of psychotic symptoms or mania [see Warnings and Precautions (5.4)].
Priapism
Advise patients of the possibility of painful or prolonged penile erections (priapism). Instruct them to seek immediate medical attention in the event of priapism [see Warnings and Precautions (5.5)].
Circulation Problems in Fingers and Toes [Peripheral Vasculopathy, Including Raynaud’s Phenomenon]
Instruct patients beginning treatment with methylphenidate hydrochloride extended-release capsules about the risk of peripheral vasculopathy, including Raynaud’s Phenomenon, and associated signs and symptoms: fingers or toes may feel numb, cool, painful, and/or may change color from pale, to blue, to red. Instruct patients to report to their physician any new numbness, pain, skin color change, or sensitivity to temperature in fingers or toes.
Instruct patients to call their physician immediately with any signs of unexplained wounds appearing on fingers or toes while taking methylphenidate hydrochloride extended-release capsules. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients [see Warnings and Precautions (5.6)].
Suppression of Growth
Advise patients that methylphenidate hydrochloride extended-release capsules may cause slowing of growth and weight loss [see Warnings and Precautions (5.7)].
Alcohol Effect
Advise patients to avoid alcohol while taking methylphenidate hydrochloride extended-release capsules. Consumption of alcohol while taking methylphenidate hydrochloride extended-release capsules may result in a more rapid release of the dose of methylphenidate [see Clinical Pharmacology (12.3)].
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in patients exposed to methylphenidate hydrochloride extended-release capsules during pregnancy [see Use in Specific Populations (8.1)].
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454
Rev. F 12/2019
-
MEDICATION GUIDE
Methylphenidate Hydrochloride (meth″ il fen′ i date hye″ droe klor′ ide)
Extended-Release Capsules LA CIIWhat is the most important information I should know about methylphenidate hydrochloride extended-release capsules?
Methylphenidate hydrochloride extended-release capsules are a federal controlled substance (CII) because it can be abused or lead to dependence. Keep methylphenidate hydrochloride extended-release capsules in a safe place to prevent misuse and abuse. Selling or giving away methylphenidate hydrochloride extended-release capsules may harm others, and is against the law. Tell your doctor if you or your child have ever abused or been dependent on alcohol, prescription medicines, or street drugs.
The following have been reported with use of methylphenidate hydrochloride and other stimulant medicines:
- Heart-related problems:
- sudden death in patients who have heart problems or heart defects
- stroke and heart attack in adults
- increased blood pressure and heart rate
Tell your doctor if you or your child have any heart problems, heart defects, high blood pressure, or a family history of these problems.
Your doctor should check you or your child carefully for heart problems before starting methylphenidate hydrochloride extended-release capsules.
Your doctor should check you or your child’s blood pressure and heart rate regularly during treatment with methylphenidate hydrochloride extended-release capsules.
Call your doctor right away if you or your child has any signs of heart problems such as chest pain, shortness of breath, or fainting while taking methylphenidate hydrochloride extended-release capsules.
- Mental (Psychiatric) problems:
All Patients
- new or worse behavior and thought problems
- new or worse bipolar illness
- new or worse aggressive behavior or hostility
- new psychotic symptoms (such as hearing voices, believing things that are not true, are suspicious) or new manic symptoms
Tell your doctor about any mental problems you or your child have, or about a family history of suicide, bipolar illness, or depression.
Call your doctor right away if you or your child have any new or worsening mental symptoms or problems while taking methylphenidate hydrochloride extended-release capsules, especially seeing or hearing things that are not real, believing things that are not real, or are suspicious.
What are methylphenidate hydrochloride extended-release capsules?Methylphenidate hydrochloride extended-release capsules are a central nervous system stimulant prescription medicine. It is used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD). Methylphenidate hydrochloride extended-release capsules may help increase attention and decrease impulsiveness and hyperactivity in patients with ADHD.
Methylphenidate hydrochloride extended-release capsules should be used as a part of a total treatment program for ADHD that may include counseling or other therapies.
It is not known if methylphenidate hydrochloride extended-release capsules are safe and effective in children under 6 years of age.
Who should not take methylphenidate hydrochloride extended-release capsules?
Methylphenidate hydrochloride extended-release capsules should not be taken if you or your child:
- are allergic to methylphenidate hydrochloride, or any of the ingredients in methylphenidate hydrochloride extended-release capsules. See the end of this Medication Guide for a complete list of ingredients in methylphenidate hydrochloride extended-release capsules.
- are taking or have taken within the past 14 days an anti-depression medicine called a monoamine oxidase inhibitor or MAOI.
Methylphenidate hydrochloride extended-release capsules may not be right for you or your child. Before starting methylphenidate hydrochloride extended-release capsules, tell your or your child’s doctor about all health conditions (or a family history of) including:
- heart problems, heart defects, high blood pressure
- mental problems including psychosis, mania, bipolar illness, or depression
- circulation problems in fingers or toes
- if you are pregnant or plan to become pregnant. It is not known if methylphenidate hydrochloride will harm your unborn baby.
- There is a pregnancy registry for females who are exposed to ADHD medications, including methylphenidate hydrochloride extended-release capsules during pregnancy. The purpose of the registry is to collect information about the health of females exposed to methylphenidate hydrochloride extended-release capsules and their baby. If you or your child becomes pregnant during treatment with methylphenidate hydrochloride extended-release capsules, talk to your healthcare provider about registering with the National Pregnancy Registry of ADHD medications at 1-866-961-2388 or visit online at https://womensmentalhealth.org/adhd-medications/.
- if you are breastfeeding or plan to breastfeed. Methylphenidate hydrochloride passes into your breast milk. Talk to your healthcare provider about the best way to feed the baby during treatment with methylphenidate hydrochloride extended-release capsules.
Tell your doctor about all of the medicines that you or your child take including prescription and over- the-counter medicines, vitamins, and herbal supplements. Methylphenidate hydrochloride extended-release capsules and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted while taking methylphenidate hydrochloride extended-release capsules.
Your doctor will decide whether methylphenidate hydrochloride extended-release capsules can be taken with other medicines.
Especially tell your doctor if you or your child takes:
- anti-depression medicines including MAOIs
- blood pressure medicines (anti-hypertensive)
Know the medicines that you or your child takes. Keep a list of your medicines with you to show your doctor and pharmacist.
Do not start any new medicine while taking methylphenidate hydrochloride extended-release capsules without talking to your doctor first.
How should methylphenidate hydrochloride extended-release capsules be taken?
- Take methylphenidate hydrochloride extended-release capsules exactly as prescribed. Your doctor may adjust the dose until it is right for you or your child.
- Take methylphenidate hydrochloride extended-release capsules once a day in the morning. Methylphenidate hydrochloride extended-release capsules are an extended-release capsule.
- Do not chew or crush methylphenidate hydrochloride extended-release capsules or the medicine inside the capsule. Swallow methylphenidate hydrochloride extended-release capsules whole with water or other liquids.
- If you cannot swallow the capsule whole, open it and sprinkle the medicine over a spoonful of applesauce.
Swallow the applesauce and medicine mixture without chewing. Follow with a drink of water or other liquid.
- You should avoid drinking alcohol during treatment with methylphenidate hydrochloride extended-release capsules. This may cause a faster release of methylphenidate hydrochloride extended-release capsules.
- From time to time, your doctor may stop methylphenidate hydrochloride extended-release capsule treatment for a while to check ADHD symptoms.
- Your doctor may do regular checks of the blood, heart, and blood pressure while taking methylphenidate hydrochloride extended-release capsules. Children should have their height and weight checked often while taking methylphenidate hydrochloride extended-release capsules. Methylphenidate hydrochloride extended-release capsules treatment may be stopped if a problem is found during these check-ups.
In case of poisoning, call your poison control center at 1-800-222-1222 right away or go to the nearest hospital emergency room.
What are possible side effects of methylphenidate hydrochloride extended-release capsules?
- See “What is the most important information I should know about methylphenidate hydrochloride extended-release capsules?” for information on reported heart and mental problems.
- painful and prolonged erections (priapism) have occurred with methylphenidate. If you or your child develops priapism, seek medical help right away. Because of the potential for lasting damage, priapism should be evaluated by a doctor immediately.
- circulation problems in fingers and toes (Peripheral Vasculopathy, Including Raynaud’s Phenomenon):
- fingers or toes may feel numb, cool, painful
- fingers or toes may change color from pale, to blue, to red
Tell your doctor if you or your child have numbness, pain, skin color change, or sensitivity to temperature in the fingers or toes.
- Call your doctor right away if you have or your child has any signs of unexplained wounds appearing on fingers or toes while taking methylphenidate hydrochloride extended-release capsules.
- slowing of growth (height and weight) in children
- fast heart beat
- abnormal
heartbeat
(palpitations)
- headache
- trouble
sleeping
- nervousness
- sweating a lot
- decreased
appetite
- dry mouth
- nausea
- stomach pain
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store methylphenidate hydrochloride extended-release capsules?
- Store methylphenidate hydrochloride extended-release capsules in a safe place and in a tightly closed container at room temperature, 68°F to 77°F (20°C to 25°C).
- Protect from moisture.
- Dispose of remaining, unused, or expired methylphenidate hydrochloride extended-release capsules by a medicine take-back program at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix methylphenidate hydrochloride extended-release capsules with an undesirable, nontoxic substance such as dirt, cat litter, or used coffee grounds to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and throw away (discard) methylphenidate hydrochloride extended-release capsules in the household trash.
- Keep methylphenidate hydrochloride extended-release capsules and all medicines out of the reach of children.
General information about the safe and effective use methylphenidate hydrochloride extended-release capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or doctor for information about methylphenidate hydrochloride extended-release capsules that is written for healthcare professionals. Do not use methylphenidate hydrochloride extended-release capsules for a condition for which it was not prescribed. Do not give methylphenidate hydrochloride extended-release capsules to other people, even if they have the same symptoms. It may harm them and it is against the law.
What are the ingredients in methylphenidate hydrochloride extended-release capsules?
Active Ingredient: methylphenidate HCL
Inactive Ingredients: ammonio methacrylate copolymer, black iron oxide, D&C Yellow #10 Aluminum Lake, FD&C Blue #1 Aluminum Lake, FD&C Blue #2 Aluminum Lake, FD&C Red #40 Aluminum Lake, gelatin, hypromellose, methacrylic acid copolymer, polyethylene glycol, propylene glycol, shellac glaze (modified), sugar spheres, talc, titanium dioxide and triethyl citrate. The 10 mg and 40 mg capsule shell contains FD&C blue no. 1, FD&C red no. 40 and FD&C yellow no. 6. The 30 mg capsule shell contains D&C red no. 33 and D&C yellow no. 10.
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454For more information about methylphenidate hydrochloride extended-release capsules call 1-888-838-2872.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Rev. E 12/2019
- Package/Label Display Panel
- Package/Label Display Panel
- Package/Label Display Panel
-
INGREDIENTS AND APPEARANCE
METHYLPHENIDATE HYDROCHLORIDE (LA)
methylphenidate hydrochloride capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0093-5346 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLPHENIDATE HYDROCHLORIDE (UNII: 4B3SC438HI) (METHYLPHENIDATE - UNII:207ZZ9QZ49) METHYLPHENIDATE HYDROCHLORIDE 20 mg Inactive Ingredients Ingredient Name Strength AMMONIO METHACRYLATE COPOLYMER TYPE B (UNII: 161H3B14U2) FERROSOFERRIC OXIDE (UNII: XM0M87F357) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) HYPROMELLOSE 2208 (3 MPA.S) (UNII: 9H4L916OBU) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color WHITE Score no score Shape CAPSULE Size 18mm Flavor Imprint Code TEVA;5346 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0093-5346-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 07/16/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA079031 07/16/2012 METHYLPHENIDATE HYDROCHLORIDE (LA)
methylphenidate hydrochloride capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0093-5347 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLPHENIDATE HYDROCHLORIDE (UNII: 4B3SC438HI) (METHYLPHENIDATE - UNII:207ZZ9QZ49) METHYLPHENIDATE HYDROCHLORIDE 30 mg Inactive Ingredients Ingredient Name Strength AMMONIO METHACRYLATE COPOLYMER TYPE B (UNII: 161H3B14U2) FERROSOFERRIC OXIDE (UNII: XM0M87F357) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) HYPROMELLOSE 2208 (3 MPA.S) (UNII: 9H4L916OBU) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Product Characteristics Color WHITE (ivory) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code TEVA;5347 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0093-5347-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 07/16/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA079031 07/16/2012 METHYLPHENIDATE HYDROCHLORIDE (LA)
methylphenidate hydrochloride capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0093-5348 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLPHENIDATE HYDROCHLORIDE (UNII: 4B3SC438HI) (METHYLPHENIDATE - UNII:207ZZ9QZ49) METHYLPHENIDATE HYDROCHLORIDE 40 mg Inactive Ingredients Ingredient Name Strength AMMONIO METHACRYLATE COPOLYMER TYPE B (UNII: 161H3B14U2) FERROSOFERRIC OXIDE (UNII: XM0M87F357) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) HYPROMELLOSE 2208 (3 MPA.S) (UNII: 9H4L916OBU) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color BROWN (light brown) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code TEVA;5348 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0093-5348-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 07/16/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA079031 07/16/2012 Labeler - Teva Pharmaceuticals USA, Inc. (001627975)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.


