Publix Super Markets, Inc. Dual Action Drug Facts
dual action by
Drug Labeling and Warnings
dual action by is a Otc medication manufactured, distributed, or labeled by Publix Super Markets Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
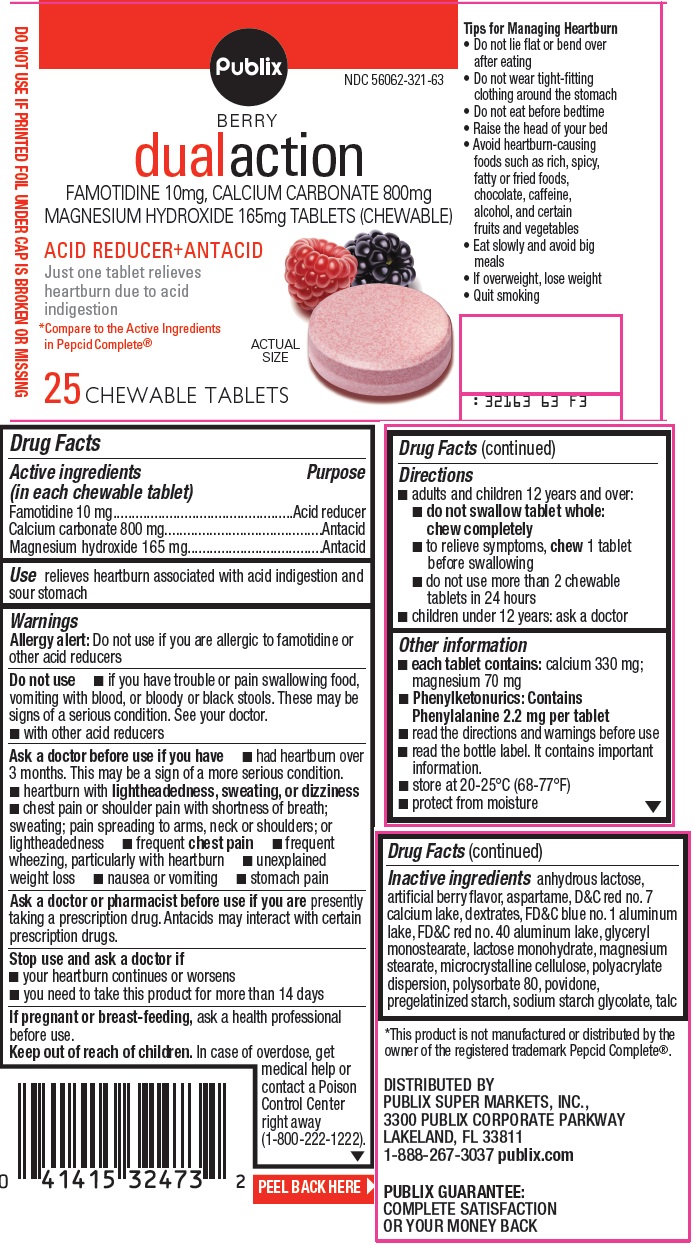
DUAL ACTION- famotidine, calcium carbonate and magnesium hydroxide tablet, chewable
Publix Super Markets Inc
----------
Publix Super Markets, Inc. Dual Action Drug Facts
Active ingredients (in each chewable tablet)
Famotidine 10 mg
Calcium carbonate 800 mg
Magnesium hydroxide 165 mg
Warnings
Allergy alert: Do not use if you are allergic to famotidine or other acid reducers
Do not use
- if you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools. These may be signs of a serious condition. See your doctor.
- with other acid reducers
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating, or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Ask a doctor or pharmacist before use if you are
presently taking a prescription drug. Antacids may interact with certain prescription drugs.
Directions
- adults and children 12 years and over:
- do not swallow tablet whole: chew completely
- to relieve symptoms, chew 1 tablet before swallowing
- do not use more than 2 chewable tablets in 24 hours
- children under 12 years: ask a doctor
Other information
- each tablet contains: calcium 330 mg; magnesium 70 mg
- Phenylketonurics: Contains Phenylalanine 2.2 mg per tablet
- read the directions and warnings before use
- read the bottle label. It contains important information.
- store at 20-25°C (68-77°F)
- protect from moisture
Inactive ingredients
anhydrous lactose, artificial berry flavor, aspartame, D&C red no. 7 calcium lake, dextrates, FD&C blue no. 1 aluminum lake, FD&C red no. 40 aluminum lake, glyceryl monostearate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyacrylate dispersion, polysorbate 80, povidone, pregelatinized starch, sodium starch glycolate, talc
| DUAL ACTION
famotidine, calcium carbonate and magnesium hydroxide tablet, chewable |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Publix Super Markets Inc (006922009) |
Trademark Results [dual action]
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.