DUTASTERIDE AND TAMSULOSIN HYDROCHLORIDE- dutasteride and tamsulosin hydrochloride capsules capsule
Dutasteride and Tamsulosin Hydrochloride by
Drug Labeling and Warnings
Dutasteride and Tamsulosin Hydrochloride by is a Prescription medication manufactured, distributed, or labeled by AvKARE. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
Dutasteride and Tamsulosin Hydrochloride Capsules
Rx Only
These highlights do not include all the information needed to use DUTASTERIDE AND TAMSULOSIN HYDROCHLORIDE CAPSULES safely and effectively. See full prescribing information for DUTASTERIDE AND TAMSULOSIN HYDROCHLORIDE CAPSULES.
Initial U.S. Approval: 2010RECENT MAJOR CHANGES
Contraindications (4) 12/2020
INDICATIONS AND USAGE
Dutasteride and tamsulosin hydrochloride capsules are a combination of dutasteride, a 5-alpha-reductase inhibitor, and tamsulosin, an alpha-adrenergic antagonist, indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate. ( 1.1)
Limitations of Use: Dutasteride-containing products, including dutasteride and tamsulosin hydrochloride capsules, are not approved for the prevention of prostate cancer. ( 1.2)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
0.5 mg dutasteride and 0.4 mg tamsulosin hydrochloride. ( 3)
CONTRAINDICATIONS
- Pregnancy. Dutasteride use is contraindicated in females who are pregnant.
(4,5.6,8.1)
- Patients with previously demonstrated, clinically significant hypersensitivity (e.g., serious skin reactions, angioedema, urticaria, pruritus, respiratory symptoms) to dutasteride, other 5-alpha-reductase inhibitors, tamsulosin, or any component of dutasteride and tamsulosin hydrochloride capsules. ( 4)
WARNINGS AND PRECAUTIONS
- Orthostatic hypotension and/or syncope can occur. Advise patients of symptoms related to postural hypotension and to avoid situations where injury could result if syncope occurs. ( 5.1)
- Do not use dutasteride and tamsulosin hydrochloride capsules with other alpha-adrenergic antagonists, as this may increase the risk of hypotension. ( 5.2)
- Dutasteride and tamsulosin hydrochloride capsules reduce serum prostate-specific antigen (PSA) concentration by approximately 50%. However, any confirmed increase in PSA while on dutasteride and tamsulosin hydrochloride capsules may signal the presence of prostate cancer and should be evaluated, even if those values are still within the normal range for untreated men. ( 5.3)
- Do not use dutasteride and tamsulosin hydrochloride capsules with strong inhibitors of cytochrome P450 (CYP) 3A4 (e.g., ketoconazole). Use caution in combination with moderate CYP3A4 inhibitors (e.g., erythromycin) or strong (e.g., paroxetine) or moderate CYP2D6 inhibitors, a combination of both CYP3A4 and CYP2D6 inhibitors, or known poor metabolizers of CYP2D6. Concomitant use with known inhibitors can cause a marked increase in drug exposure. ( 5.2, 7.1, 12.3)
- Exercise caution with concomitant use of phosphodiesterase-5 (PDE-5) inhibitors, as this may increase the risk of hypotension. ( 5.2)
- Drugs that contain dutasteride, including dutasteride and tamsulosin hydrochloride capsules, may increase the risk of high-grade prostate cancer. ( 5.4, 6.1)
- Prior to initiating treatment with dutasteride and tamsulosin hydrochloride capsules, consideration should be given to other urological conditions that may cause similar symptoms. ( 5.5)
- Females who are pregnant or may be pregnant should not handle dutasteride and tamsulosin hydrochloride capsules due to potential risk to a male fetus. ( 5.6, 8.1)
- Advise patients about the possibility and seriousness of priapism. ( 5.7)
- Patients should not donate blood until 6 months after their last dose of dutasteride and tamsulosin hydrochloride capsules. ( 5.8)
- Intraoperative Floppy Iris Syndrome has been observed during cataract and glaucoma surgery after alpha-adrenergic antagonist exposure. Advise patients considering cataract or glaucoma surgery to tell their ophthalmologist that they take or have taken dutasteride and tamsulosin hydrochloride capsules. ( 5.9)
- Exercise caution with concomitant use of warfarin. ( 5.2, 7.2, 12.3)
ADVERSE REACTIONS
The most common adverse reactions, reported in ≥1% of subjects treated with coadministered dutasteride and tamsulosin are ejaculation disorders, impotence, decreased libido, dizziness, and breast disorders. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact AvKARE at 1-855-361-3993 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for FDA-approved patient labeling and FDA-approved patient labeling.
Revised: 11/2023
- Pregnancy. Dutasteride use is contraindicated in females who are pregnant.
(4,5.6,8.1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
RECENT MAJOR CHANGES
1 INDICATIONS AND USAGE
1.1 Benign Prostatic Hyperplasia (BPH) Treatment
1.2 Limitations of Use
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Orthostatic Hypotension
5.2 Drug-Drug Interactions
5.3 Effects on Prostate-Specific Antigen (PSA) and the Use of PSA in Prostate Cancer Detection
5.4 Increased Risk of High-Grade Prostate Cancer
5.5 Evaluation for Other Urological Diseases
5.6 Transdermal Exposure of Dutasteride and Tamsulosin Hydrochloride in Pregnant Females -Risk to Male Fetus
5.7 Priapism
5.8 Blood Donation
5.9 Intraoperative Floppy Iris Syndrome
5.10 Sulfa Allergy
5.11 Effect on Semen Characteristics
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Cytochrome P450 Inhibition
7.2 Warfarin
7.3 Nifedipine, Atenolol, Enalapril
7.4 Digoxin and Theophylline
7.5 Furosemide
7.6 Calcium Channel Antagonists
7.7 Cholestyramine
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
The recommended dosage of dutasteride and tamsulosin hydrochloride capsules is 1 capsule (0.5 mg dutasteride and 0.4 mg tamsulosin hydrochloride) taken once daily approximately 30 minutes after the same meal each day.
The capsules should be swallowed whole and not chewed or opened. Contact with the contents of the dutasteride and tamsulosin hydrochloride capsule may result in irritation of the oropharyngeal mucosa.
-
3 DOSAGE FORMS AND STRENGTHS
Dutasteride and tamsulosin hydrochloride capsules, containing 0.5 mg dutasteride and 0.4 mg tamsulosin hydrochloride, are capsules with blue opaque cap imprinted with “C280” and white opaque body imprinted with “0.5/0.4” in black ink containing white to off-white spherical shaped pellets and one oblong, opaque yellow softgel capsule printed with “C300” in black ink.
-
4 CONTRAINDICATIONS
Dutasteride and tamsulosin hydrochloride capsules are contraindicated for use in:
- Pregnancy. Dutasteride use is contraindicated in females who are pregnant. In animal reproduction and developmental toxicity studies, dutasteride inhibited development of male fetus external genitalia. Therefore, dutasteride and tamsulosin hydrochloride capsules may cause fetal harm when administered to a pregnant female.
[seeWarnings and Precautions (
5.6), Use in Specific Populations (
8.1)
].
- Patients with previously demonstrated, clinically significant hypersensitivity (e.g., serious skin reactions, angioedema, urticaria, pruritus, respiratory symptoms) to dutasteride, other 5-alpha-reductase inhibitors, tamsulosin, or any other component of dutasteride and tamsulosin hydrochloride capsules [seeAdverse Reactions ( 6.2) ].
- Pregnancy. Dutasteride use is contraindicated in females who are pregnant. In animal reproduction and developmental toxicity studies, dutasteride inhibited development of male fetus external genitalia. Therefore, dutasteride and tamsulosin hydrochloride capsules may cause fetal harm when administered to a pregnant female.
[seeWarnings and Precautions (
5.6), Use in Specific Populations (
8.1)
].
-
5 WARNINGS AND PRECAUTIONS
5.1 Orthostatic Hypotension
As with other alpha-adrenergic antagonists, orthostatic hypotension (postural hypotension, dizziness, and vertigo) may occur in patients treated with tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules, and can result in syncope. Patients starting treatment with dutasteride and tamsulosin hydrochloride capsules should be cautioned to avoid situations where syncope could result in an injury [seeAdverse Reactions ( 6.1) ].
5.2 Drug-Drug Interactions
Strong Inhibitors of Cytochrome P450 (CYP) 3A4
Tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules, should not be coadministered with strong CYP3A4 inhibitors (e.g., ketoconazole) as this can significantly increase tamsulosin exposure [seeDrug Interactions ( 7.1), Clinical Pharmacology ( 12.3)].
Moderate Inhibitors of CYP3A4, Inhibitors of CYP2D6, or a Combination of Both CYP3A4 and CYP2D6 Inhibitors
Tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules, should be used with caution when coadministered with moderate inhibitors of CYP3A4 (e.g., erythromycin), strong (e.g., paroxetine) or moderate (e.g., terbinafine) inhibitors of CYP2D6, a combination of both CYP3A4 and CYP2D6 inhibitors, or in patients known to be poor metabolizers of CYP2D6, as there is a potential for significant increase in tamsulosin exposure [seeDrug Interactions ( 7.1), Clinical Pharmacology ( 12.3) ].
Cimetidine
Caution is advised when tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules, are coadministered with cimetidine [seeDrug Interactions ( 7.1), Clinical Pharmacology ( 12.3) ].
Other Alpha-adrenergic Antagonists
Tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules, should not be coadministered with other alpha-adrenergic antagonists because of the increased risk of symptomatic hypotension.
Phosphodiesterase-5 (PDE-5) Inhibitors
Caution is advised when alpha-adrenergic-antagonist-containing products, including dutasteride and tamsulosin hydrochloride capsules, are coadministered with PDE-5 inhibitors. Alpha-adrenergic antagonists and PDE-5 inhibitors are both vasodilators that can lower blood pressure. Concomitant use of these 2 drug classes can potentially cause symptomatic hypotension.
Warfarin
Caution should be exercised with concomitant administration of warfarin and tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules [seeDrug Interactions ( 7.2), Clinical Pharmacology ( 12.3) ].
5.3 Effects on Prostate-Specific Antigen (PSA) and the Use of PSA in Prostate Cancer Detection
Coadministration of dutasteride with tamsulosin resulted in similar changes to serum PSA as with dutasteride monotherapy.
In clinical trials, dutasteride reduced serum PSA concentration by approximately 50% within 3 to 6 months of treatment. This decrease was predictable over the entire range of PSA values in patients with symptomatic BPH, although it may vary in individuals. Dutasteride‑containing treatment, including dutasteride and tamsulosin hydrochloride capsules, may also cause decreases in serum PSA in the presence of prostate cancer. To interpret serial PSAs in men treated with a dutasteride-containing product, including dutasteride and tamsulosin hydrochloride capsules, a new baseline PSA should be established at least 3 months after starting treatment and PSA monitored periodically thereafter. Any confirmed increase from the lowest PSA value while on a dutasteride-containing treatment, including dutasteride and tamsulosin hydrochloride capsules, may signal the presence of prostate cancer and should be evaluated, even if PSA levels are still within the normal range for men not taking a 5-alpha-reductase inhibitor. Noncompliance with dutasteride and tamsulosin hydrochloride capsules may also affect PSA test results.
To interpret an isolated PSA value in a man treated with dutasteride and tamsulosin hydrochloride capsules, for 3 months or more, the PSA value should be doubled for comparison with normal values in untreated men.
The free-to-total PSA ratio (percent free PSA) remains constant, even under the influence of dutasteride. If clinicians elect to use percent free PSA as an aid in the detection of prostate cancer in men receiving dutasteride and tamsulosin hydrochloride capsules, no adjustment to its value appears necessary.
5.4 Increased Risk of High-Grade Prostate Cancer
In men aged 50 to 75 years with a prior negative biopsy for prostate cancer and a baseline PSA between 2.5 ng/mL and 10.0 ng/mL taking dutasteride in the 4-year Reduction by Dutasteride of Prostate Cancer Events (REDUCE) trial, there was an increased incidence of Gleason score 8 to 10 prostate cancer compared with men taking placebo (dutasteride 1.0% versus placebo 0.5%) [seeIndications and Usage ( 1.2), Adverse Reactions ( 6.1) ]. In a 7-year placebo-controlled clinical trial with another 5-alpha-reductase inhibitor (finasteride 5 mg, PROSCAR), similar results for Gleason score 8 to 10 prostate cancer were observed (finasteride 1.8% versus placebo 1.1%).
5-alpha-reductase inhibitors may increase the risk of development of high-grade prostate cancer. Whether the effect of 5-alpha-reductase inhibitors to reduce prostate volume or trial‑related factors impacted the results of these trials has not been established.
5.5 Evaluation for Other Urological Diseases
Prior to initiating treatment with dutasteride and tamsulosin hydrochloride capsules, consideration should be given to other urological conditions that may cause similar symptoms. In addition, BPH and prostate cancer may coexist.
5.6 Transdermal Exposure of Dutasteride and Tamsulosin Hydrochloride in Pregnant Females -Risk to Male Fetus
Dutasteride and tamsulosin hydrochloride capsules should not be handled by females who are pregnant or may be pregnant. Dutasteride can be absorbed through the skin and could result in unintended fetal exposure and potential risk to a male fetus. If a female who is or may be pregnant comes in contact with a leaking capsule, the contact area should be washed immediately with soap and water [seeUse in Specific Populations (8.1)]. Dutasteride can be absorbed through the skin based on animal studies [see Nonclinical Toxicology( 13.2) ].
5.7 Priapism
Priapism (persistent painful penile erection unrelated to sexual activity) has been associated (probably less than 1 in 50,000) with the use of alpha-adrenergic antagonists, including tamsulosin, which is a component of dutasteride and tamsulosin hydrochloride capsules. Because this condition can lead to permanent impotence if not properly treated, patients should be advised about the seriousness of the condition.
5.8 Blood Donation
Men being treated with a dutasteride-containing product, including dutasteride and tamsulosin hydrochloride capsules, should not donate blood until at least 6 months have passed following their last dose. The purpose of this deferred period is to prevent administration of dutasteride to a pregnant female transfusion recipient.
5.9 Intraoperative Floppy Iris Syndrome
Intraoperative Floppy Iris Syndrome (IFIS) has been observed during cataract and glaucoma surgery in some patients on or previously treated with alpha-adrenergic-antagonists, including tamsulosin, which is a component of dutasteride and tamsulosin hydrochloride capsules.
Most reports were in patients taking the alpha-adrenergic-antagonist when IFIS occurred, but in some cases, the alpha-adrenergic antagonist had been stopped prior to surgery. In most of these cases, the alpha-adrenergic antagonist had been stopped recently prior to surgery (2 to 14 days), but in a few cases, IFIS was reported after the patients had been off the alpha-adrenergic antagonist for a longer period (5 weeks to 9 months). IFIS is a variant of small pupil syndrome and is characterized by the combination of a flaccid iris that billows in response to intraoperative irrigation currents, progressive intraoperative miosis despite preoperative dilation with standard mydriatic drugs, and potential prolapse of the iris toward the phacoemulsification incisions. The patient’s ophthalmologist should be prepared for possible modifications to their surgical technique, such as the utilization of iris hooks, iris dilator rings, or viscoelastic substances.
IFIS may increase the risk of eye complications during and after the operation. The benefit of stopping alpha-adrenergic antagonist therapy prior to cataract or glaucoma surgery has not been established. The initiation of therapy with tamsulosin in patients for whom cataract or glaucoma surgery is scheduled is not recommended.
5.10 Sulfa Allergy
In patients with sulfa allergy, allergic reaction to tamsulosin has been rarely reported. If a patient reports a serious or life-threatening sulfa allergy, caution is warranted when administering tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules.
5.11 Effect on Semen Characteristics
Dutasteride
The effects of dutasteride 0.5 mg/day on semen characteristics were evaluated in healthy men throughout 52 weeks of treatment and 24 weeks of post-treatment follow-up. At 52 weeks, compared with placebo, dutasteride treatment resulted in mean reduction in total sperm count, semen volume, and sperm motility; the effects on total sperm count were not reversible after 24 weeks of follow-up. Sperm concentration and sperm morphology were unaffected and mean values for all semen parameters remained within the normal range at all timepoints. The clinical significance of the effect of dutasteride on semen characteristics for an individual patient’s fertility is not known. [see Use in Specific Populations ( 8.3) ].
Tamsulosin
The effects of tamsulosin hydrochloride on sperm counts or sperm function have not been evaluated.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
The clinical efficacy and safety of coadministered dutasteride and tamsulosin, which are individual components of dutasteride and tamsulosin hydrochloride capsules, have been evaluated in a multicenter, randomized, double-blind, parallel group trial (the Combination with Alpha-Blocker Therapy, or CombAT, trial). Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trial of another drug and may not reflect the rates observed in practice.
- The most common adverse reactions reported in subjects receiving coadministered dutasteride and tamsulosin were impotence, decreased libido, breast disorders (including breast enlargement and tenderness), ejaculation disorders, and dizziness. Ejaculation disorders occurred significantly more in subjects receiving coadministration therapy (11%) compared with those receiving dutasteride (2%) or tamsulosin (4%) as monotherapy.
- Trial withdrawal due to adverse reactions occurred in 6% of subjects receiving coadministered dutasteride and tamsulosin and in 4% of subjects receiving dutasteride or tamsulosin as monotherapy. The most common adverse reaction in all treatment arms leading to trial withdrawal was erectile dysfunction (1% to 1.5%).
In the CombAT trial, over 4,800 male subjects with BPH were randomly assigned to receive 0.5 mg dutasteride, 0.4 mg tamsulosin hydrochloride, or coadministration therapy (0.5 mg dutasteride and 0.4 mg tamsulosin hydrochloride) administered once daily in a 4-year double-blind trial. Overall, 1,623 subjects received monotherapy with dutasteride; 1,611 subjects received monotherapy with tamsulosin; and 1,610 subjects received coadministration therapy. The population was aged 49 to 88 years (mean age: 66 years) and 88% were white. Table 1summarizes adverse reactions reported in at least 1% of subjects receiving coadministration therapy and at a higher incidence than subjects receiving either dutasteride or tamsulosin as monotherapy.
Table 1. Adverse Reactions Reported over a 48-Month Period in ≥1% of Subjects and More Frequently in the Coadministration Therapy Group than the Dutasteride or Tamsulosin Monotherapy Group (CombAT) by Time of Onset Adverse Reaction
Adverse Reaction Time of Onset
Year 1
Year 2
Year 3
Year 4
Months 0-6
Months 7-12
Coadministration a
(n = 1,610)
(n = 1,527)
(n = 1,428)
(n = 1,283)
(n = 1,200)
Dutasteride
(n = 1,623)
(n = 1,548)
(n = 1,464)
(n = 1,325)
(n = 1,200)
Tamsulosin
(n = 1,611)
(n = 1,545)
(n = 1,468)
(n = 1,281)
(n = 1,112)
Ejaculation disorders b,c
Coadministration
7.8%
1.6%
1.0%
0.5%
<0.1%
Dutasteride
1.0%
0.5%
0.5%
0.2%
0.3%
Tamsulosin
2.2%
0.5%
0.5%
0.2%
0.3%
Impotence c,d
Coadministration
5.4%
1.1%
1.8%
0.9%
0.4%
Dutasteride
4.0%
1.1%
1.6%
0.6%
0.3%
Tamsulosin
2.6%
0.8%
1.0%
0.6%
1.1%
Decreased libido c,e
Coadministration
4.5%
0.9%
0.8%
0.2%
0.0%
Dutasteride
3.1%
0.7%
1.0%
0.2%
0.0%
Tamsulosin
2.0%
0.6%
0.7%
0.2%
<0.1%
Breast disorders ,f
Coadministration
1.1%
1.1%
0.8%
0.9%
0.6%
Dutasteride
0.9%
0.9%
1.2%
0.5%
0.7%
Tamsulosin
0.4%
0.4%
0.4%
0.2%
0.0%
Dizziness
Coadministration
1.1%
0.4%
0.1%
<0.1%
0.2%
Dutasteride
0.5%
0.3%
0.1%
<0.1%
<0.1%
Tamsulosin
0.9%
0.5%
0.4%
<0.1%
0.0%
aCoadministration = dutasteride 0.5 mg once daily plus tamsulosin 0.4 mg once daily.
bIncludes anorgasmia, retrograde ejaculation, semen volume decreased, orgasmic sensation decreased, orgasm abnormal, ejaculation delayed, ejaculation disorder, ejaculation failure, and premature ejaculation.
c These sexual adverse reactions are associated with dutasteride treatment (including monotheraphy and combination with tamsulosin). These adverse reactions may persist after treatment discontinuation. The role of dutasteride in this persistence is unknown.
dIncludes erectile dysfunction and disturbance in sexual arousal.
eIncludes libido decreased, libido disorder, loss of libido, sexual dysfunction, and male sexual dysfunction.
fIncludes breast enlargement, gynecomastia, breast swelling, breast pain, breast tenderness, nipple pain, and nipple swelling.
Cardiac Failure
In CombAT, after 4 years of treatment, the incidence of the composite term cardiac failure in the coadministration group (12/1,610; 0.7%) was higher than in either monotherapy group: dutasteride, 2/1,623 (0.1%) and tamsulosin, 9/1,611 (0.6%). Composite cardiac failure was also examined in a separate 4-year placebo-controlled trial evaluating dutasteride in men at risk for development of prostate cancer. The incidence of cardiac failure in subjects taking dutasteride was 0.6% (26/4,105) compared with 0.4% (15/4,126) in subjects on placebo. A majority of subjects with cardiac failure in both trials had comorbidities associated with an increased risk of cardiac failure. Therefore, the clinical significance of the numerical imbalances in cardiac failure is unknown. No causal relationship between dutasteride alone or coadministered with tamsulosin and cardiac failure has been established. No imbalance was observed in the incidence of overall cardiovascular adverse events in either trial.
Additional information regarding adverse reactions in placebo-controlled trials with dutasteride or tamsulosin monotherapy follows.
Dutasteride
Long-term Treatment (Up to 4 Years): High-Grade Prostate Cancer:The REDUCE trial was a randomized, double-blind, placebo-controlled trial that enrolled 8,231 men aged 50 to 75 years with a serum PSA of 2.5 ng/mL to 10 ng/mL and a negative prostate biopsy within the previous 6 months. Subjects were randomized to receive placebo (n = 4,126) or 0.5 mg daily doses of dutasteride (n = 4,105) for up to 4 years. The mean age was 63 years and 91% were white. Subjects underwent protocol-mandated scheduled prostate biopsies at 2 and 4 years of treatment or had “for-cause biopsies” at non-scheduled times if clinically indicated. There was a higher incidence of Gleason score 8 to 10 prostate cancer in men receiving dutasteride (1.0%) compared with men on placebo (0.5%) [seeIndications and Usage ( 1.2), Warnings and Precautions ( 5.4) ].In a 7-year placebo-controlled clinical trial with another 5-alpha-reductase inhibitor (finasteride 5 mg, PROSCAR ®), similar results for Gleason score 8 to 10 prostate cancer were observed (finasteride 1.8% versus placebo 1.1%).
No clinical benefit has been demonstrated in patients with prostate cancer treated with dutasteride.
Reproductive and Breast Disorders
In the 3 pivotal placebo-controlled BPH trials with dutasteride, each 4 years in duration, there was no evidence of increased sexual adverse reactions (impotence, decreased libido, and ejaculation disorder) or breast disorders with increased duration of treatment. Among these 3 trials, there was 1 case of breast cancer in the dutasteride group and 1 case in the placebo group. No cases of breast cancer were reported in any treatment group in the 4-year CombAT trial or the 4-year REDUCE trial.
The relationship between long-term use of dutasteride and male breast neoplasia is currently unknown.
Tamsulosin
According to the tamsulosin prescribing information, in two 13-week treatment trials with tamsulosin monotherapy, adverse reactions occurring in at least 2% of subjects receiving 0.4 mg tamsulosin hydrochloride and at an incidence higher than in subjects receiving placebo were: infection, asthenia, back pain, chest pain, somnolence, insomnia, rhinitis, pharyngitis, cough increased, sinusitis, and diarrhea.
Signs and Symptoms of Orthostasis:According to the tamsulosin prescribing information, in clinical trials with tamsulosin monotherapy, a positive orthostatic test result was observed in 16% (81/502) of subjects receiving 0.4 mg tamsulosin hydrochloride versus 11% (54/493) of subjects receiving placebo. Because orthostasis was detected more frequently in the tamsulosin-treated subjects than in placebo recipients, there is a potential risk of syncope [seeWarnings and Precautions ( 5.1) ].
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of the individual components of dutasteride and tamsulosin hydrochloride capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to drug exposure.
Dutasteride
Immune System Disorders:Hypersensitivity reactions, including rash, pruritus, urticaria, localized edema, serious skin reactions, and angioedema.
Neoplasms:Male breast cancer.
Psychiatric Disorders:Depressed mood.
Reproductive System and Breast Disorders: Testicular pain and testicular swelling.
Tamsulosin
Immune System Disorders:Hypersensitivity reactions, including rash, urticaria, pruritus, angioedema, and respiratory problems have been reported with positive rechallenge in some cases.
Cardiac Disorders:Palpitations, dyspnea, atrial fibrillation, arrhythmia, and tachycardia.
Skin Disorders:Skin desquamation, including Stevens-Johnson syndrome, erythema multiforme, dermatitis exfoliative.
Gastrointestinal Disorders:Constipation, vomiting, dry mouth.
Reproductive System and Breast Disorders:Priapism.
Respiratory:Epistaxis.
Vascular Disorders:Hypotension.
Ophthalmologic Disorders:Blurred vision, visual impairment. During cataract and glaucoma surgery, a variant of small pupil syndrome known as Intraoperative Floppy Iris Syndrome (IFIS) associated with alpha-adrenergic-antagonist therapy [seeWarnings and Precautions ( 5.9) ].
To report SUSPECTED ADVERSE REACTIONS, contact AvKARE at 1-855-361-3993 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
7 DRUG INTERACTIONS
There have been no drug interaction trials using dutasteride and tamsulosin hydrochloride capsules. The following sections reflect information available for the individual components.
7.1 Cytochrome P450 Inhibition
Dutasteride
Dutasteride is extensively metabolized in humans by the CYP3A4 and CYP3A5 isoenzymes. The effect of potent CYP3A4 inhibitors on dutasteride has not been studied. Because of the potential for drug-drug interactions, use caution when prescribing a dutasteride-containing product, including dutasteride and tamsulosin hydrochloride capsules, to patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir) [seeClinical Pharmacology ( 12.3) ].
Tamsulosin
Strong and Moderate Inhibitors of CYP3A4 or CYP2D6:Tamsulosin is extensively metabolized, mainly by CYP3A4 or CYP2D6.
Concomitant treatment with ketoconazole (a strong inhibitor of CYP3A4) resulted in increases in the C maxand AUC of tamsulosin by factors of 2.2 and 2.8, respectively. Concomitant treatment with paroxetine (a strong inhibitor of CYP2D6) resulted in increases in the C maxand area under the concentration-time curve (AUC) of tamsulosin by factors of 1.3 and 1.6, respectively. A similar increase in exposure is expected in poor metabolizers (PM) of CYP2D6 as compared to extensive metabolizers (EM). Since CYP2D6 PMs cannot be readily identified and the potential for significant increase in tamsulosin exposure exists when tamsulosin 0.4 mg is coadministered with strong CYP3A4 inhibitors in CYP2D6 PMs, tamsulosin 0.4 mg capsules should not be used in combination with strong inhibitors of CYP3A4 (e.g., ketoconazole). The effects of coadministration of both a CYP3A4 and a CYP2D6 inhibitor with tamsulosin have not been evaluated. However, there is a potential for significant increase in tamsulosin exposure when tamsulosin 0.4 mg is coadministered with a combination of both CYP3A4 and CYP2D6 inhibitors [seeWarnings and Precautions ( 5.2), Clinical Pharmacology ( 12.3) ].
Cimetidine: Treatment with cimetidine resulted in a moderate increase in tamsulosin hydrochloride AUC (44%) [seeWarnings and Precautions ( 5.2), Clinical Pharmacology ( 12.3) ].
7.2 Warfarin
Dutasteride
Concomitant administration of dutasteride 0.5 mg/day for 3 weeks with warfarin does not alter the steady-state pharmacokinetics of the S- or R-warfarin isomers or alter the effect of warfarin on prothrombin time [seeClinical Pharmacology ( 12.3) ].
Tamsulosin
A definitive drug-drug interaction trial between tamsulosin hydrochloride and warfarin was not conducted. Results from limited in vitroand in vivostudies are inconclusive. Caution should be exercised with concomitant administration of warfarin and tamsulosin-containing products, including dutasteride and tamsulosin hydrochloride capsules [seeWarnings and Precautions ( 5.2), Clinical Pharmacology ( 12.3) ].
7.3 Nifedipine, Atenolol, Enalapril
Tamsulosin
Dosage adjustments are not necessary when tamsulosin is administered concomitantly with nifedipine, atenolol, or enalapril [seeClinical Pharmacology ( 12.3) ].
7.4 Digoxin and Theophylline
Dutasteride
Dutasteride does not alter the steady-state pharmacokinetics of digoxin when administered concomitantly at a dose of 0.5 mg/day for 3 weeks [seeClinical Pharmacology ( 12.3)].
Tamsulosin
Dosage adjustments are not necessary when tamsulosin is administered concomitantly with digoxin or theophylline [seeClinical Pharmacology ( 12.3) ].
7.5 Furosemide
Tamsulosin
Tamsulosin had no effect on the pharmacodynamics (excretion of electrolytes) of furosemide. While furosemide produced an 11% to 12% reduction in tamsulosin hydrochloride C maxand AUC, these changes are expected to be clinically insignificant and do not require adjustment of the dose of tamsulosin [seeClinical Pharmacology ( 12.3) ].
7.6 Calcium Channel Antagonists
Dutasteride
Coadministration of verapamil or diltiazem decreases dutasteride clearance and leads to increased exposure to dutasteride. The change in dutasteride exposure is not considered to be clinically significant. No dosage adjustment of dutasteride is recommended [seeClinical Pharmacology ( 12.3) ].
7.7 Cholestyramine
Dutasteride
Administration of a single 5 mg dose of dutasteride followed 1 hour later by a 12 g dose of cholestyramine does not affect the relative bioavailability of dutasteride [seeClinical Pharmacology ( 12.3) ].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Dutasteride and tamsulosin hydrochloride capsules are contraindicated for use in pregnancy because it may cause harm to the male fetus [seeContraindications (4)]. Dutasteride and tamsulosin hydrochloride capsules are not indicated for use in females.
Dutasteride, a component of dutasteride and tamsulosin hydrochloride capsules, is a 5-alpha-reductase inhibitor that prevents conversion of testosterone to dihydrotestosterone (DHT), a hormone necessary for normal development of male genitalia. Abnormalities in the genitalia of male fetuses are an expected physiological consequence of inhibition of this conversion. These results are similar to observations in male infants with genetic 5-alpha-reductase deficiency.
In animal reproduction studies, dutasteride inhibited normal development of external genitalia in male offspring when given to rats or rabbits during organogenesis at less than the maximum recommended human dose (MRHD) of 0.5 mg daily, in the absence of maternal toxicity. At 15 times the MRHD, prolonged pregnancy, decreased reproductive organ weights, and delayed puberty in male offspring were observed in rats, with no-effect levels less than the MRHD of 0.5 mg daily. Increased placental weights in rabbits were also observed, with no-effect levels less than the MRHD of 0.5 mg daily (see Data).
Although dutasteride is secreted into human semen, the drug concentration in the human female partner is approximately 100 times less than concentrations producing abnormalities of male genitalia in animal studies (see Data). In monkeys dosed during organogenesis at blood concentrations comparable to or above levels to which a human female partner is estimated to be exposed, male offspring external genitalia was not adversely affected. No feminization occurred in male offspring of untreated female rats mated to treated male rats even though detectable blood levels of dutasteride were observed in the female rats [seeNonclinical Toxicology (13.1)].
No adverse developmental effects were observed in animal studies in which tamsulosin hydrochloride was administered to rats or rabbits during the period of organogenesis (see Data).
Data
Human Data: Dutasteride:The highest measured semen concentration of dutasteride in treated men was 14 ng/mL. Although dutasteride is detected in semen, assuming exposure of a 50 kg female to 5 mL of semen and 100% absorption, the female’s expected dutasteride blood concentration through semen would be about 0.0175 ng/mL. This concentration is approximately 100 times less than blood concentrations producing abnormalities of male genitalia in animal studies. Dutasteride is highly protein bound in human semen (greater than 96%), which may reduce the amount of dutasteride available for vaginal absorption.
Animal Data: Dutasteride:In an embryo-fetal development study in rats, oral administration of dutasteride at 10 times less than the MRHD of 0.5 mg daily (based on average blood levels in men) resulted in feminization of male genitalia in the fetus (decreased anogenital distance at 0.05 mg/kg/day with a lack of a no-effect level) in the absence of maternal toxicity. In addition, nipple development, hypospadias, and distended preputial glands occurred in fetuses of dams treated at doses of 2.5 mg/kg/day or greater (approximately 15 times the MRHD). Reduced fetal body weight and associated delayed ossification in the presence of maternal toxicity (decreased body weight gain) were observed at maternal exposure approximately 15 times the MRHD (dose of 2.5 mg/kg/day or greater). An increase in stillborn pups was observed in dams treated at 30 mg/kg/day (approximately 111 times the MRHD), with a no- effect level of 12.5 mg/kg/day.
In a rabbit embryo-fetal development study, doses 28 times the MRHD (doses of 30mg/kg/day or greater), based on average blood levels in men, were administered orally on Gestation Days 7 to 29 (during organogenesis and the late period of external genitalia development). Histological evaluation of the genital papilla of fetuses revealed evidence of feminization of the male fetus as well as fused skull bones and increased placental weights at all doses in the absence of maternal toxicity. A second embryo-fetal development study in rabbits dosed throughout pregnancy (organogenesis and later period of external genitalia development [Gestation Days 6 to 29]) at 0.3 times the MHRD doses of 0.05 mg/kg/day or greater, with no no-effect level),) also produced evidence of feminization of the genitalia in male fetuses and increased placental weights at all doses in the absence of maternal toxicity.
In an embryo-fetal development study, pregnant rhesus monkeys were exposed intravenously during organogenesis (Gestation Days 20 to 100) to a dutasteride blood level comparable to or above the estimated dutasteride exposure of a human female partner. Dutasteride was administered on Gestation Days 20 to 100 (during organogenesis) at doses of 400, 780, 1,325, or 2,010 ng/day (12 monkeys/group). No feminization of male external genitalia of monkey offspring was observed. Reduction of fetal adrenal weights, reduction in fetal prostate weights, and increases in fetal ovarian and testis weights were observed at the highest dose tested. Based on the highest measured semen concentration of dutasteride in treated men (14 ng/mL), these doses in the monkey represent up to 16 times the potential maximum exposure of a 50 kg human female to 5 mL of semen daily from a dutasteride-treated male, assuming 100% absorption. The dose levels (on a ng/kg basis) administered to monkeys in this study are 32 to 186 times the nominal (ng/kg) dose to which a female would potentially be exposed via the semen. It is not known whether rabbits or rhesus monkeys produce any of the major human metabolites.
In an oral pre- and post-natal development study in rats, feminization of the male genitalia was observed. Decreased anogenital distance was observed at 0.05 times the MRHD and greater (0.05 mg/kg/day and greater), with a lack of a no-effect level, based on average blood levels in men as an estimation of AUC. Hypospadias and nipple development were observed at 2.5 mg/kg/day or greater (14 times the MRHD or greater, with a no-effect level at 0.05 mg/kg/day). Doses of 2.5 mg/kg/day and greater also resulted in prolonged gestation in the parental females, an increase in time to balano-preputial separation in male offspring, a decrease in time to vaginal patency for female offspring, and a decrease in prostate and seminal vesicle weights in male offspring. Increased stillbirths and decreased neonatal viability in offspring were noted at 30 mg/kg/day (102 times the MRHD in the presence of maternal toxicity [decreased body weights]).
Tamsulosin:Administration of tamsulosin hydrochloride to pregnant female rats during the period of organogenesis (Gestation Days 7 to 17) at dose levels up to approximately 50 times the human therapeutic AUC exposure (300 mg/kg/day) revealed no evidence of harm to the fetus. Administration of tamsulosin hydrochloride to pregnant rabbits during the period of organogenesis (Gestation Days 6 to 18) at dose levels up to 50 mg/kg/day produced no evidence of fetal harm.
8.2 Lactation
Risk Summary
Dutasteride and tamsulosin hydrochloride capsules are not indicated for use in females.
8.3 Females and Males of Reproductive Potential
Infertility
Dutasteride: Males:The effects of dutasteride 0.5 mg/day on semen characteristics were evaluated in normal volunteers aged 18 to 52 years (n = 27 dutasteride, n = 23 placebo) throughout 52 weeks of treatment and 24 weeks of post-treatment follow-up. At 52 weeks, the mean percent reductions from baseline in total sperm count, semen volume, and sperm motility were 23%, 26%, and 18%, respectively, in the dutasteride group when adjusted for changes from baseline in the placebo group. Sperm concentration and sperm morphology were unaffected. After 24 weeks of follow-up, the mean percent change in total sperm count in the dutasteride group remained 23% lower than baseline. While mean values for all semen parameters at all timepoints remained within the normal ranges and did not meet predefined criteria for a clinically significant change (30%), 2 subjects in the dutasteride group had decreases in sperm count of greater than 90% from baseline at 52 weeks, with partial recovery at the 24-week follow-up. The clinical significance of these effects on semen characteristics for an individual patient’s fertility is not known [seeWarnings and Precautions ( 5.11) ].
Tamsulosin: Males:Abnormal ejaculation including ejaculation failure, ejaculation disorder, retrograde ejaculation, and decreased ejaculation has been associated with tamsulosin hydrochloride .Studies in rats revealed significantly reduced fertility in males, considered to be due to impairment of ejaculation, which was reversible [seeNonclinical Toxicology ( 13.1) ].
8.4 Pediatric Use
Dutasteride and tamsulosin hydrochloride capsules are not indicated for use in pediatric patients. Safety and effectiveness of dutasteride and tamsulosin hydrochloride capsules in pediatric patients have not been established.
8.5 Geriatric Use
Of 1,610 male subjects treated with coadministered dutasteride and tamsulosin in the CombAT trial, 58% of enrolled subjects were aged 65 years and older and 13% of enrolled subjects were aged 75 years and older. No overall differences in safety or efficacy were observed between these subjects and younger subjects but greater sensitivity of some older individuals cannot be ruled out [seeClinical Pharmacology ( 12.3)].
8.6 Renal Impairment
The effect of renal impairment on dutasteride and tamsulosin pharmacokinetics has not been studied using dutasteride and tamsulosin hydrochloride capsules. Because no dosage adjustment is necessary for dutasteride or tamsulosin in patients with moderate-to-severe renal impairment (10≤ CL cr<30 mL/min/1.73 m 2), no dosage adjustment is necessary for dutasteride and tamsulosin hydrochloride capsules in patients with moderate-to-severe renal impairment. However, patients with end-stage renal disease (CL cr<10 mL/min/1.73 m 2) have not been studied [seeClinical Pharmacology ( 12.3) ].
8.7 Hepatic Impairment
The effect of hepatic impairment on dutasteride and tamsulosin pharmacokinetics has not been studied using dutasteride and tamsulosin hydrochloride capsules. The following text reflects information available for the individual components.
Dutasteride
The effect of hepatic impairment on dutasteride pharmacokinetics has not been studied. Because dutasteride is extensively metabolized, exposure could be higher in hepatically impaired patients. However, in a clinical trial where 60 subjects received 5 mg (10 times the therapeutic dose) daily for 24 weeks, no additional adverse events were observed compared with those observed at the therapeutic dose of 0.5 mg [seeClinical Pharmacology ( 12.3) ].
Tamsulosin
Patients with moderate hepatic impairment do not require an adjustment in tamsulosin dosage. Tamsulosin has not been studied in patients with severe hepatic impairment [seeClinical Pharmacology ( 12.3) ].
-
10 OVERDOSAGE
No data are available with regard to overdosage with dutasteride and tamsulosin hydrochloride capsules. The following text reflects information available for the individual components.
Dutasteride
In volunteer trials, single doses of dutasteride up to 40 mg (80 times the therapeutic dose) for 7 days have been administered without significant safety concerns. In a clinical trial, daily doses of 5 mg (10 times the therapeutic dose) were administered to 60 subjects for 6 months with no additional adverse effects to those seen at therapeutic doses of 0.5 mg.
There is no specific antidote for dutasteride. Therefore, in cases of suspected overdosage symptomatic and supportive treatment should be given as appropriate, taking the long half-life of dutasteride into consideration.
Tamsulosin
Should overdosage of tamsulosin lead to hypotension [seeWarnings and Precautions ( 5.1), Adverse Reactions ( 6.1) ], support of the cardiovascular system is of first importance. Restoration of blood pressure and normalization of heart rate may be accomplished by keeping the patient in the supine position. If this measure is inadequate, then administration of intravenous fluids should be considered. If necessary, vasopressors should then be used and renal function should be monitored and supported as needed. Laboratory data indicate that tamsulosin is 94% to 99% protein bound; therefore, dialysis is unlikely to be of benefit.
-
11 DESCRIPTION
Dutasteride and tamsulosin hydrochloride capsules contain dutasteride (a selective inhibitor of both the type 1 and type 2 isoforms of steroid 5 alpha-reductase, an intracellular enzyme that converts testosterone to DHT and tamsulosin (an antagonist of alpha 1A-adrenoceptors in the prostate). Each dutasteride and tamsulosin hydrochloride capsule contains the following:
- One dutasteride oblong, opaque, yellow gelatin capsule, containing 0.5 mg of dutasteride dissolved in a mixture of butylated hydroxytoluene and mono-and di-glycerides of caprylic/capric acid. The inactive ingredients in the soft-gelatin capsule shell are ferric oxide (yellow), gelatin (from certified BSE-free bovine sources), glycerin, titanium dioxide, lecithin, medium chain triglycerides, propylene glycol, iron oxide black, polyvinyl acetate phthalate, macrogol, and ammonium hydroxide.
- Tamsulosin hydrochloride white to off-white pellets, containing 0.4 mg tamsulosin hydrochloride and the inactive ingredients: methacrylic acid copolymer, sugar sphere, ethylcellulose, polyethylene glycol, triethyl citrate and talc.
The above components are encapsulated in a hard-shell capsule made with the inactive ingredients of hypromellose, FD&C Blue #1, titanium dioxide, shellac, iron oxide black, propylene glycol, FD&C blue #2, FD&C red #40, D&C yellow #10. Blue opaque cap imprinted with “C280” and white opaque body imprinted with “0.5/0.4” in black ink containing white to off white spherical shaped pellets and one oblong, opaque yellow softgel capsule printed with “C300” in black ink.
Dutasteride:Dutasteride is a synthetic 4-azasteroid compound chemically designated as (5α,17β)-N-{2,5 bis(trifluoromethyl)phenyl}-3-oxo-4-azaandrost-1-ene-17-carboxamide. The empirical formula of dutasteride is C 27H 30F 6N 2O 2, representing a molecular weight of 528.5 with the following structural formula:
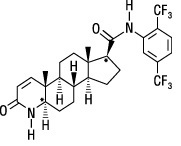
Dutasteride is a white to pale yellow powder with a melting point of 242° to 250°C. It is soluble in ethanol (44 mg/mL), methanol (64 mg/mL), and polyethylene glycol 400 (3 mg/mL), but it is insoluble in water.
Tamsulosin:Tamsulosin hydrochloride is a synthetic compound chemically designated as (-)-( R)-5-[2-[[2-( o-Ethoxyphenoxy)ethyl]amino]propyl]-2-methoxybenzenesulfonamide, monohydrochloride.
The empirical formula of tamsulosin hydrochloride is C 20H 28N 2O 5SHCl. The molecular weight of tamsulosin hydrochloride is 444.97. Its structural formula is:
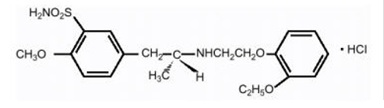
Tamsulosin hydrochloride is a white or almost white crystalline powder that melts with decomposition at approximately 234°C. It is sparingly soluble in water and slightly soluble in methanol, ethanol, acetone, and ethyl acetate.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Dutasteride and tamsulosin hydrochloride capsules are a combination of 2 drugs with different mechanisms of action to improve symptoms in patients with BPH: dutasteride, a 5-alpha-reductase inhibitor, and tamsulosin, an antagonist of alpha 1A-adrenoreceptors.
Dutasteride
Dutasteride inhibits the conversion of testosterone to DHT. DHT is the androgen primarily responsible for the initial development and subsequent enlargement of the prostate gland. Testosterone is converted to DHT by the enzyme 5-alpha-reductase, which exists as 2 isoforms, type 1 and type 2. The type 2 isoenzyme is primarily active in the reproductive tissues, while the type 1 isoenzyme is also responsible for testosterone conversion in the skin and liver.
Dutasteride is a competitive and specific inhibitor of both type 1 and type 2 5-alpha-reductase isoenzymes, with which it forms a stable enzyme complex. Dissociation from this complex has been evaluated under in vitroand in vivoconditions and is extremely slow. Dutasteride does not bind to the human androgen receptor.
Tamsulosin
Smooth muscle tone is mediated by the sympathetic nervous stimulation of alpha 1-adrenoceptors, which are abundant in the prostate, prostatic capsule, prostatic urethra, and bladder neck. Blockade of these adrenoceptors can cause smooth muscles in the bladder neck and prostate to relax, resulting in an improvement in urine flow rate and a reduction in symptoms of BPH.
Tamsulosin, an alpha 1-adrenoceptor blocking agent, exhibits selectivity for alpha 1-receptors in the human prostate. At least 3 discrete alpha 1-adrenoceptor subtypes have been identified: alpha 1A, alpha 1B, and alpha 1D; their distribution differs between human organs and tissue. Approximately 70% of the alpha 1-receptors in human prostate are of the alpha 1Asubtype. Tamsulosin is not intended for use as an antihypertensive.
12.2 Pharmacodynamics
Dutasteride
Effect on 5-Alpha-Dihydrotestosterone and Testosterone:The maximum effect of daily doses of dutasteride on the reduction of DHT is dose-dependent and is observed within 1 to 2 weeks. After 1 and 2 weeks of daily dosing with dutasteride 0.5 mg, median serum DHT concentrations were reduced by 85% and 90%, respectively. In patients with BPH treated with dutasteride 0.5 mg/day for 4 years, the median decrease in serum DHT was 94% at 1 year, 93% at 2 years, and 95% at both 3 and 4 years. The median increase in serum testosterone was 19% at both 1 and 2 years, 26% at 3 years, and 22% at 4 years, but the mean and median levels remained within the physiologic range.
In patients with BPH treated with 5 mg/day of dutasteride or placebo for up to 12 weeks prior to transurethral resection of the prostate, mean DHT concentrations in prostatic tissue were significantly lower in the dutasteride group compared with placebo (784 and 5,793 pg/g, respectively, P<0.001). Mean prostatic tissue concentrations of testosterone were significantly higher in the dutasteride group compared with placebo (2,073 and 93 pg/g, respectively, P<0.001).
Adult males with genetically inherited type 2 5-alpha-reductase deficiency also have decreased DHT levels. These 5-alpha-reductase-deficient males have a small prostate gland throughout life and do not develop BPH. Except for the associated urogenital defects present at birth, no other clinical abnormalities related to 5-alpha-reductase deficiency have been observed in these individuals.
Effects on Other Hormones:In healthy volunteers, 52 weeks of treatment with dutasteride 0.5 mg/day (n = 26) resulted in no clinically significant change compared with placebo (n = 23) in sex hormone-binding globulin, estradiol, luteinizing hormone, follicle-stimulating hormone, thyroxine (free T4), and dehydroepiandrosterone. Statistically significant, baseline-adjusted mean increases compared with placebo were observed for total testosterone at 8 weeks (97.1 ng/dL, P<0.003) and thyroid-stimulating hormone at 52 weeks (0.4 mcIU/mL, P<0.05). The median percentage changes from baseline within the dutasteride group were 17.9% for testosterone at 8 weeks and 12.4% for thyroid-stimulating hormone at 52 weeks. After stopping dutasteride for 24 weeks, the mean levels of testosterone and thyroid-stimulating hormone had returned to baseline in the group of subjects with available data at the visit. In subjects with BPH treated with dutasteride in a large randomized, double-blind, placebo-controlled trial, there was a median percent increase in luteinizing hormone of 12% at 6 months and 19% at both 12 and 24 months.
Other Effects:Plasma lipid panel and bone mineral density were evaluated following 52 weeks of dutasteride 0.5 mg once daily in healthy volunteers. There was no change in bone mineral density as measured by dual energy x-ray absorptiometry compared with either placebo or baseline. In addition, the plasma lipid profile (i.e., total cholesterol, low density lipoproteins, high density lipoproteins, triglycerides) was unaffected by dutasteride. No clinically significant changes in adrenal hormone responses to adrenocorticotropic hormone (ACTH) stimulation were observed in a subset population (n = 13) of the 1 year healthy volunteer trial.
12.3 Pharmacokinetics
The pharmacokinetics of dutasteride and tamsulosin from dutasteride and tamsulosin hydrochloride capsules are comparable to the pharmacokinetics of dutasteride and tamsulosin when administered separately.
Absorption
The pharmacokinetic parameters of dutasteride and tamsulosin observed after administration of dutasteride and tamsulosin hydrochloride capsules in a single-dose, randomized, 3-period, partial cross-over trial are summarized in Table 2below.
Table 2. Arithmetic Means (SD) of Serum Dutasteride and Tamsulosin in Single-Dose Pharmacokinetic Parameters under Fed Conditions Component
N
AUC (0-t)(ng h/mL)
C max(ng/mL)
T max(h) a
t ½(h)
Dutasteride
92
39.6 (23.1)
2.14 (0.77)
3.00 (1.00-10.00)
Tamsulosin
92
187.2 (95.7)
11.3 (4.44)
6.00 (2.00-24.00)
13.5 (3.92) b
aMedian (range).
bN = 91.
Dutasteride:Following administration of a single 0.5 mg dose of a soft gelatin capsule, time to peak absolute bioavailability in 5 healthy subjects is approximately 60% (range: 40% to 94%).
Tamsulosin:Absorption of tamsulosin is essentially complete (>90%) following oral administration of 0.4 mg tamsulosin hydrochloride capsules under fasting conditions. Tamsulosin exhibits linear kinetics following single and multiple dosing, with achievement of steady-state concentrations by the fifth day of once-daily dosing.
Effect of Food
Food does not affect the pharmacokinetics of dutasteride following administration of dutasteride and tamsulosin hydrochloride capsules. However, a mean 30% decrease in tamsulosin C maxwas observed when dutasteride and tamsulosin hydrochloride capsules was administered with food, similar to that seen when tamsulosin monotherapy was administered under fed versus fasting conditions.
Distribution
Dutasteride:Pharmacokinetic data following single and repeat oral doses show that dutasteride has a large volume of distribution (300 to 500 L). Dutasteride is highly bound to plasma albumin (99.0%) and alpha-1 acid glycoprotein (AAG, 96.6%).
In a trial of healthy subjects (n = 26) receiving dutasteride 0.5 mg/day for 12 months, semen dutasteride concentrations averaged 3.4 ng/mL (range: 0.4 to 14 ng/mL) at 12 months and, similar to serum, achieved steady-state concentrations at 6 months. On average, at 12 months 11.5% of serum dutasteride concentrations partitioned into semen.
Tamsulosin:The mean steady-state apparent volume of distribution of tamsulosin after intravenous administration to 10 healthy male adults was 16 L, which is suggestive of distribution into extracellular fluids in the body.
Tamsulosin is extensively bound to human plasma proteins (94% to 99%), primarily AAG, with linear binding over a wide concentration range (20 to 600 ng/mL). The results of 2-way in vitrostudies indicate that the binding of tamsulosin to human plasma proteins is not affected by amitriptyline, diclofenac, glyburide, simvastatin plus simvastatin-hydroxy acid metabolite, warfarin, diazepam, or propranolol. Likewise, tamsulosin had no effect on the extent of binding of these drugs.
Metabolism
Dutasteride:Dutasteride is extensively metabolized in humans. In vitrostudies showed that dutasteride is metabolized by the CYP3A4 and CYP3A5 isoenzymes. Both of these isoenzymes produced the 4’-hydroxydutasteride, 6-hydroxydutasteride, and the 6,4’-dihydroxydutasteride metabolites. In addition, the 15-hydroxydutasteride metabolite was formed by CYP3A4. Dutasteride is not metabolized in vitroby human cytochrome P450 isoenzymes CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP2E1. In human serum following dosing to steady state, unchanged dutasteride, 3 major metabolites (4’-hydroxydutasteride, 1,2-dihydrodutasteride, and 6-hydroxydutasteride), and 2 minor metabolites (6,4’-dihydroxydutasteride and 15-hydroxydutasteride), as assessed by mass spectrometric response, have been detected. The absolute stereochemistry of the hydroxyl additions in the 6 and 15 positions is not known. In vitro, the 4’-hydroxydutasteride and 1,2-dihydrodutasteride metabolites are much less potent than dutasteride against both isoforms of human 5α-reductase. The activity of 6β-hydroxydutasteride is comparable to that of dutasteride.
Tamsulosin:There is no enantiomeric bioconversion from tamsulosin [R(-) isomer] to the S(+) isomer in humans. Tamsulosin is extensively metabolized by cytochrome P450 enzymes in the liver and less than 10% of the dose is excreted in urine unchanged. However, the pharmacokinetic profile of the metabolites in humans has not been established. In vitrostudies indicate that CYP3A4 and CYP2D6 are involved in metabolism of tamsulosin as well as some minor participation of other CYP isoenzymes. Inhibition of hepatic drug metabolizing enzymes may lead to increased exposure to tamsulosin [seeDrug Interactions ( 7.1) ]. The metabolites of tamsulosin undergo extensive conjugation to glucuronide or sulfate prior to renal excretion.
Incubations with human liver microsomes showed no evidence of clinically significant metabolic interactions between tamsulosin and amitriptyline, albuterol, glyburide, and finasteride. However, results of the in vitrotesting of the tamsulosin interaction with diclofenac and warfarin were equivocal.
Excretion
Dutasteride:Dutasteride and its metabolites were excreted mainly in feces. As a percent of dose, there was approximately 5% unchanged dutasteride (approximately 1% to approximately 15%) and 40% as dutasteride-related metabolites (approximately 2% to approximately 90%). Only trace amounts of unchanged dutasteride were found in urine (<1%). Therefore, on average, the dose unaccounted for approximated 55% (range: 5% to 97%). The terminal elimination half-life of dutasteride is approximately 5 weeks at steady state. The average steady-state serum dutasteride concentration was 40 ng/mL following 0.5 mg/day for 1 year. Following daily dosing, dutasteride serum concentrations achieve 65% of steady-state concentration after 1 month and approximately 90% after 3 months. Due to the long half-life of dutasteride, serum concentrations remain detectable (greater than 0.1 ng/mL) for up to 4 to 6 months after discontinuation of treatment.
Tamsulosin:On administration of the radiolabeled dose of tamsulosin to 4 healthy volunteers, 97% of the administered radioactivity was recovered, with urine (76%) representing the primary route of excretion compared with feces (21%) over 168 hours.
Following intravenous or oral administration of an immediate-release formulation, the elimination half-life of tamsulosin in plasma ranges from 5 to 7 hours. Because of absorption rate-controlled pharmacokinetics with tamsulosin hydrochloride capsules, the apparent half-life of tamsulosin is approximately 9 to 13 hours in healthy volunteers and 14 to 15 hours in the target population.
Tamsulosin undergoes restrictive clearance in humans, with a relatively low systemic clearance (2.88 L/h).
Specific Populations
Pediatric Patients:The pharmacokinetics of dutasteride and tamsulosin administered together have not been investigated in subjects younger than 18 years.
Geriatric Patients:Dutasteride and tamsulosin pharmacokinetics using dutasteride and tamsulosin hydrochloride capsules have not been studied in geriatric patients. The following text reflects information for the individual components.
Dutasteride:No dosage adjustment is necessary in the elderly. The pharmacokinetics and pharmacodynamics of dutasteride were evaluated in 36 healthy male subjects aged between 24 and 87 years following administration of a single 5 mg dose of dutasteride. In this single-dose trial, dutasteride half-life increased with age (approximately 170 hours in men aged 20 to 49 years, approximately 260 hours in men aged 50 to 69 years, and approximately 300 hours in men older than 70 years).
Tamsulosin:Cross-study comparison of tamsulosin overall exposure (AUC) and half-life indicate that the pharmacokinetic disposition of tamsulosin may be slightly prolonged in geriatric males compared with young, healthy male volunteers. Intrinsic clearance is independent of tamsulosin binding to AAG, but diminishes with age, resulting in a 40% overall higher exposure (AUC) in subjects aged 55 to 75 years compared with subjects aged 20 to 32 years.
Male and Female Patients:Dutasteride:Dutasteride is contraindicated in females who are pregnant and is not indicated for use in females [seeCONTRAINDICATIONS (4), Warnings and Precautions (5.6)]. The pharmacokinetics of dutasteride in females have not been studied.
Tamsulosin:Tamsulosin is not indicated for use in females. No information is available on the pharmacokinetics of tamsulosin in females.
Racial and Ethnic Groups:The effect of race on the pharmacokinetics of dutasteride and tamsulosin administered together or separately has not been studied.
Patients with Renal Impairment:The effect of renal impairment on dutasteride and tamsulosin pharmacokinetics has not been studied using dutasteride and tamsulosin hydrochloride capsules. The following text reflects information for the individual components.
Dutasteride:The effect of renal impairment on dutasteride pharmacokinetics has not been studied. However, less than 0.1% of a steady-state 0.5 mg dose of dutasteride is recovered in human urine, so no adjustment in dosage is anticipated for patients with renal impairment.
Tamsulosin:The pharmacokinetics of tamsulosin have been compared in 6 subjects with mild-moderate (30≤ CL cr<70 mL/min/1.73 m 2) or moderate-severe (10≤ CL cr<30 mL/min/1.73 m 2) renal impairment and 6 normal subjects (CL cr>90 mL/min/1.73 m 2). While a change in the overall plasma concentration of tamsulosin was observed as the result of altered binding to AAG, the unbound (active) concentration of tamsulosin, as well as the intrinsic clearance, remained relatively constant. Therefore, patients with renal impairment do not require an adjustment in tamsulosin dosing. However, patients with end-stage renal disease (CL cr<10 mL/min/1.73 m 2) have not been studied.
Patients with Hepatic Impairment:The effect of hepatic impairment on dutasteride and tamsulosin pharmacokinetics has not been studied using dutasteride and tamsulosin hydrochloride capsules. The following text reflects information available for the individual components.
Dutasteride:The effect of hepatic impairment on dutasteride pharmacokinetics has not been studied. Because dutasteride is extensively metabolized, exposure could be higher in hepatically impaired patients.
Tamsulosin:The pharmacokinetics of tamsulosin have been compared in 8 subjects with moderate hepatic impairment (Child-Pugh classification: Grades A and B) and 8 normal subjects. While a change in the overall plasma concentration of tamsulosin was observed as the result of altered binding to AAG, the unbound (active) concentration of tamsulosin does not change significantly with only a modest (32%) change in intrinsic clearance of unbound tamsulosin. Therefore, patients with moderate hepatic impairment do not require an adjustment in tamsulosin dosage. Tamsulosin has not been studied in patients with severe hepatic impairment.
Drug Interaction Studies
There have been no drug interaction studies using dutasteride and tamsulosin hydrochloride capsules. The following text reflects information available for the individual components.
Cytochrome P450 Inhibitors: Dutasteride:No clinical drug interaction trials have been performed to evaluate the impact of CYP3A enzyme inhibitors on dutasteride pharmacokinetics. However, based on in vitrodata, blood concentrations of dutasteride may increase in the presence of inhibitors of CYP3A4/5 such as ritonavir, ketoconazole, verapamil, diltiazem, cimetidine, troleandomycin, and ciprofloxacin.
Dutasteride does not inhibit the in vitrometabolism of model substrates for the major human cytochrome P450 isoenzymes (CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4) at a concentration of 1,000 ng/mL, 25 times greater than steady-state serum concentrations in humans.
Tamsulosin: Strong and Moderate Inhibitors of CYP3A4 or CYP2D6:The effects of ketoconazole (a strong inhibitor of CYP3A4) at 400 mg once daily for 5 days on the pharmacokinetics of a single tamsulosin hydrochloride capsule 0.4 mg dose was investigated in 24 healthy volunteers (age range: 23 to 47 years). Concomitant treatment with ketoconazole resulted in increases in the C maxand AUC of tamsulosin by factors of 2.2 and 2.8, respectively. The effects of concomitant administration of a moderate CYP3A4 inhibitor (e.g., erythromycin) on the pharmacokinetics of tamsulosin have not been evaluated.
The effects of paroxetine (a strong inhibitor of CYP2D6) at 20 mg once daily for 9 days on the pharmacokinetics of a single tamsulosin capsule 0.4 mg dose was investigated in 24 healthy volunteers (age range: 23 to 47 years). Concomitant treatment with paroxetine resulted in increases in the C maxand AUC of tamsulosin by factors of 1.3 and 1.6, respectively. A similar increase in exposure is expected in poor metabolizers (PM) of CYP2D6 as compared with extensive metabolizers (EM). A fraction of the population (about 7% of whites and 2% of African-Americans) are CYP2D6 PMs. Since CYP2D6 PMs cannot be readily identified and the potential for significant increase in tamsulosin exposure exists when tamsulosin 0.4 mg is coadministered with strong CYP3A4 inhibitors in CYP2D6 PMs, tamsulosin 0.4 mg capsules should not be used in combination with strong inhibitors of CYP3A4 (e.g., ketoconazole).
The effects of concomitant administration of a moderate CYP2D6 inhibitor (e.g., terbinafine) on the pharmacokinetics of tamsulosin have not been evaluated.
The effects of coadministration of both a CYP3A4 and a CYP2D6 inhibitor with tamsulosin capsules have not been evaluated. However, there is a potential for significant increase in tamsulosin exposure when tamsulosin 0.4 mg is coadministered with a combination of both CYP3A4 and CYP2D6 inhibitors.
Cimetidine:The effects of cimetidine at the highest recommended dose (400 mg every 6 hours for 6 days) on the pharmacokinetics of a single tamsulosin capsule 0.4 mg dose was investigated in 10 healthy volunteers (age range: 21 to 38 years). Treatment with cimetidine resulted in a significant decrease (26%) in the clearance of tamsulosin hydrochloride, which resulted in a moderate increase in tamsulosin hydrochloride AUC (44%).
Alpha-adrenergic Antagonists:Dutasteride:In a single-sequence, crossover trial in healthy volunteers, the administration of tamsulosin or terazosin in combination with dutasteride had no effect on the steady-state pharmacokinetics of either alpha-adrenergic antagonist. Although the effect of administration of tamsulosin or terazosin on dutasteride pharmacokinetic parameters was not evaluated, the percent change in DHT concentrations was similar for dutasteride, alone or in combination with tamsulosin or terazosin.
Warfarin:Dutasteride:In a trial of 23 healthy volunteers, 3 weeks of treatment with dutasteride 0.5 mg/day did not alter the steady-state pharmacokinetics of the S- or R-warfarin isomers or alter the effect of warfarin on prothrombin time when administered with warfarin.
Tamsulosin:A definitive drug-drug interaction trial between tamsulosin and warfarin was not conducted. Results from limited in vitroand in vivostudies are inconclusive. Therefore, caution should be exercised with concomitant administration of warfarin and tamsulosin.
Nifedipine, Atenolol, Enalapril: Tamsulosin:In 3 trials in hypertensive subjects (age range: 47 to 79 years) whose blood pressure was controlled with stable doses of nifedipine extended-release, atenolol, or enalapril for at least 3 months, tamsulosin hydrochloride capsules 0.4 mg for 7 days followed by tamsulosin hydrochloride capsules 0.8 mg for another 7 days (n = 8 per trial) resulted in no clinically significant effects on blood pressure and pulse rate compared with placebo (n = 4 per trial). Therefore, dosage adjustments are not necessary when tamsulosin is administered concomitantly with nifedipine extended-release, atenolol, or enalapril.
Digoxin and Theophylline:Dutasteride:In a trial of 20 healthy volunteers, dutasteride did not alter the steady-state pharmacokinetics of digoxin when administered concomitantly at a dose of 0.5 mg/day for 3 weeks.
Tamsulosin:In 2 trials in healthy volunteers (n = 10 per trial; age range: 19 to 39 years) receiving tamsulosin capsules 0.4 mg/day for 2 days, followed by tamsulosin capsules 0.8 mg/day for 5 to 8 days, single intravenous doses of digoxin 0.5 mg or theophylline 5 mg/kg resulted in no change in the pharmacokinetics of digoxin or theophylline. Therefore, dosage adjustments are not necessary when a tamsulosin capsule is administered concomitantly with digoxin or theophylline.
Furosemide: Tamsulosin:The pharmacokinetic and pharmacodynamic interaction between tamsulosin hydrochloride capsules 0.8 mg/day (steady-state) and furosemide 20 mg intravenously (single dose) was evaluated in 10 healthy volunteers (age range: 21 to 40 years). Tamsulosin had no effect on the pharmacodynamics (excretion of electrolytes) of furosemide. While furosemide produced an 11% to 12% reduction in tamsulosin C maxand AUC, these changes are expected to be clinically insignificant and do not require dose adjustment for tamsulosin.
Calcium Channel Antagonists: Dutasteride:In a population pharmacokinetics analysis, a decrease in clearance of dutasteride was noted when coadministered with the CYP3A4 inhibitors verapamil (-37%, n = 6) and diltiazem (-44%, n = 5). In contrast, no decrease in clearance was seen when amlodipine, another calcium channel antagonist that is not a CYP3A4 inhibitor, was coadministered with dutasteride (+7%, n = 4). The decrease in clearance and subsequent increase in exposure to dutasteride in the presence of verapamil and diltiazem is not considered to be clinically significant. No dosage adjustment is recommended.
Cholestyramine:Dutasteride:Administration of a single 5 mg dose of dutasteride followed 1 hour later by 12 g cholestyramine did not affect the relative bioavailability of dutasteride in 12 normal volunteers.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No non-clinical studies have been conducted with dutasteride and tamsulosin hydrochloride capsules. The following information is based on studies performed with dutasteride or tamsulosin.
Carcinogenesis
Dutasteride:A 2-year carcinogenicity study was conducted in B6C3F1 mice at doses of 3, 35, 250, and 500 mg/kg/day for males and 3, 35, and 250 mg/kg/day for females; an increased incidence of benign hepatocellular adenomas was noted at 250 mg/kg/day (290-fold the MRHD of a 0.5 mg daily dose) in female mice only. Two of the 3 major human metabolites have been detected in mice. The exposure to these metabolites in mice is either lower than in humans or is not known.
In a 2-year carcinogenicity study in Han Wistar rats, at doses of 1.5, 7.5, and 53 mg/kg/day in males and 0.8, 6.3, and 15 mg/kg/day in females, there was an increase in Leydig cell adenomas in the testes at 135-fold the MRHD (53 mg/kg/day and greater). An increased incidence of Leydig cell hyperplasia was present at 52-fold the MRHD (male rat doses of 7.5 mg/kg/day and greater). A positive correlation between proliferative changes in the Leydig cells and an increase in circulating luteinizing hormone levels has been demonstrated with 5-alpha-reductase inhibitors and is consistent with an effect on the hypothalamic-pituitary-testicular axis following 5-alpha-reductase inhibition. At tumorigenic doses, luteinizing hormone levels in rats were increased by 167%. In this study, the major human metabolites were tested for carcinogenicity at approximately 1 to 3 times the expected clinical exposure.
Tamsulosin:In a rat carcinogenicity assay, no increases in tumor incidence was observed in rats administered up to 3 times the MRHD of 0.8 mg/day (based on AUC of animal doses up to 43 mg/kg/day in males and up to 52 mg/kg/day in females), with the exception of a modest increase in the frequency of mammary gland fibroadenomas in female rats receiving doses of 5.4 mg/kg or greater.
In a carcinogenicity assay, mice were administered up to 8 times the MRHD of tamsulosin (oral doses up to 127 mg/kg/day in males and 158 mg/kg/day in females). There were no significant tumor findings in male mice. Female mice treated for 2 years with the 2 highest doses of 45 and 158 mg/kg/day had statistically significant increases in the incidence of mammary gland fibroadenomas ( P<0.0001) and adenocarcinomas.
The increased incidences of mammary gland neoplasms in female rats and mice were considered secondary to tamsulosin-induced hyperprolactinemia. It is not known if tamsulosin elevates prolactin in humans. The relevance for human risk of the findings of prolactin-mediated endocrine tumors in rodents is not known.
Mutagenesis
Dutasteride:Dutasteride was tested for genotoxicity in a bacterial mutagenesis assay (Ames test), a chromosomal aberration assay in Chinese hamster ovary (CHO) cells, and a micronucleus assay in rats. The results did not indicate any genotoxic potential of the parent drug. Two major human metabolites were also negative in either the Ames test or an abbreviated Ames test.
Tamsulosin:Tamsulosin produced no evidence of mutagenic potential in vitroin the Ames reverse mutation test, mouse lymphoma thymidine kinase assay, unscheduled DNA repair synthesis assay, and chromosomal aberration assays in CHO cells or human lymphocytes. There were no mutagenic effects in the in vivosister chromatid exchange and mouse micronucleus assay.
Impairment of Fertility
Dutasteride:Treatment of sexually mature male rats with dutasteride at 0.1 times the MRHD (animal doses of 0.05 mg/kg/day or greater for up to 31 weeks) based on mean serum concentration resulted in dose- and time-dependent decreases in fertility at all doses; reduced cauda epididymal (absolute) sperm counts but not sperm concentration (at 50 and 500 mg/kg/day); reduced weights of the epididymis, prostate, and seminal vesicles; and microscopic changes (cytoplasmic vacuolation of tubular epithelium in the epididymides and/or decreased cytoplasmic content of epithelium, consistent with decreased secretory activity in the prostate and seminal vesicles) in the reproductive organs at all doses in the absence of paternal toxicity. The fertility effects were reversed by Recovery Week 6 at all doses, and sperm counts were normal at the end of a 14-week recovery period. The microscopic changes were no longer present at Recovery Week 14 at 0.1 times the MHRD and were partly recovered in the remaining treatment groups. Low levels of dutasteride (0.6 to 17 ng/mL) were detected in the serum of untreated female rats mated to treated males (10 to 500 mg/kg/day for 29 to 30 weeks) which are 16 to 110 times the MRHD based on mean serum concentration. No feminization occurred in male offspring of untreated female rats mated to treated male rats even though detectable blood levels of dutasteride were observed in the female rats.
In a fertility study in female rats with dosing 4 weeks prior to mating through early gestation, oral administration of dutasteride at doses of 0.05, 2.5, 12.5, and 30 mg/kg/day resulted in reduced litter size due to increased resorptions and in feminization of male fetuses (decreased anogenital distance) at 2 to 10 times the MRHD (animal doses of 2.5 mg/kg/day or greater) based on mean serum concentration, in the presence of maternal toxicity (decreased body weight gain). Fetal body weights were also reduced at approximately 0.02 times the MRHD (rat dose of 0.05 mg/kg/day or greater) based on mean serum concentration, with no no-effect level, in the absence of maternal toxicity.
Tamsulosin:Studies in rats revealed significantly reduced fertility in males dosed with single or multiple daily doses of 300 mg/kg/day of tamsulosin hydrochloride (AUC exposure in rats about 50 times the human exposure with the maximum therapeutic dose). The mechanism of decreased fertility in male rats is considered to be an effect of the compound on the vaginal plug formation possibly due to changes of semen content or impairment of ejaculation. The effects on fertility were reversible, showing improvement by 3 days after a single dose and 4 weeks after multiple daily dosing. Effects on fertility in males were completely reversed within 9 weeks after discontinuation of multiple daily dosing. Multiple doses of 10 and 100 mg/kg/day tamsulosin hydrochloride (1/5 and 16 times the anticipated human AUC exposure) did not significantly alter fertility in male rats. Effects of tamsulosin on sperm counts or sperm function have not been evaluated.
Studies in female rats revealed significant reductions in fertility after single or multiple daily doses of 300 mg/kg/day of the R-isomer or racemic mixture of tamsulosin hydrochloride, respectively. In female rats, the reductions in fertility after single doses were considered to be associated with impairments in fertilization. Multiple daily doses of 10 or 100 mg/kg/day of the racemic mixture did not significantly alter fertility in female rats.
13.2 Animal Toxicology and/or Pharmacology
Central Nervous System Toxicology Studies
Dutasteride:In rats and dogs, repeated oral administration of dutasteride resulted in some animals showing signs of non-specific, reversible, centrally-mediated toxicity without associated histopathological changes at exposures 425- and 315-fold the expected clinical exposure (of parent drug), respectively.
Rabbit Dermal Absorption
In a rabbit dermal pharmacokinetics study, dermal absorption of dutasteride in CAPMUL (glyceryl oleate) in rabbits resulted in serum concentrations of 2.7 to 40.5 mcg/h/mL for doses of 1 to 20 mg/mL, respectively, or 56% to 100% of applied dutasteride to be absorbed under occluded and prolonged conditions. Dutasteride and tamsulosin hydrochloride soft gelatin capsules administered orally contain 0.5 mg dutasteride dissolved in a mixture of mono-di-glycerides of caprylic/capric acid and butylated hydroxytoluene. Dutasteride in water was minimally absorbed in rabbits (2,000 mg/kg).
-
14 CLINICAL STUDIES
The trial supporting the efficacy of dutasteride and tamsulosin hydrochloride capsules was a 4-year multicenter, randomized, double-blind, parallel-group trial (CombAT trial) investigating the efficacy of the coadministration of dutasteride 0.5 mg/day and tamsulosin hydrochloride 0.4 mg/day (n = 1,610) compared with dutasteride alone (n = 1,623) or tamsulosin alone (n = 1,611). Subjects were aged at least 50 years with a serum PSA ≥1.5 ng/mL and <10 ng/mL and BPH diagnosed by medical history and physical examination, including enlarged prostate (≥30 cc) and BPH symptoms that were moderate to severe according to the International Prostate Symptom Score (IPSS). Eighty-eight percent (88%) of the enrolled trial population was white. Approximately 52% of subjects had previous exposure to 5-alpha-reductase inhibitor or alpha-adrenergic antagonist treatment. Of the 4,844 subjects randomly assigned to receive treatment, 69% of subjects in the coadministration group, 67% in the dutasteride group, and 61% in the tamsulosin group completed 4 years of double-blind treatment.
Effect on Symptom Score
Symptoms were quantified using the first 7 questions of the International Prostate Symptom Score (IPSS). The baseline score was approximately 16.4 units for each treatment group. Coadministration therapy was statistically superior to each of the monotherapy treatments in decreasing symptom score at Month 24, the primary time point for this endpoint. At Month 24, the mean changes from baseline (±SD) in IPSS total symptom scores were -6.2 (±7.14) for the coadministration group, -4.9 (±6.81) for dutasteride, and -4.3 (±7.01) for tamsulosin, with a mean difference between coadministration and dutasteride of -1.3 units ( P<0.001; [95% CI: -1.69, -0.86]), and between coadministration and tamsulosin of -1.8 units ( P<0.001; [95% CI: -2.23, -1.40]). A significant difference was seen by Month 9 and continued through Month 48. At Month 48 the mean changes from baseline (±SD) in IPSS total symptom scores were -6.3 (±7.40) for coadministration, -5.3 (±7.14) for dutasteride, and -3.8 (±7.74) for tamsulosin, with a mean difference between coadministration and dutasteride of -0.96 units ( P<0.001; [95% CI: -1.40, -0.52]), and between coadministration and tamsulosin of -2.5 units ( P<0.001; [95% CI: -2.96, -2.07]). See Figure 1.
Figure 1. International Prostate Symptom Score Change from Baseline over a 48-MonthPeriod (Randomized, Double-blind, Parallel-group Trial [CombAT Trial])
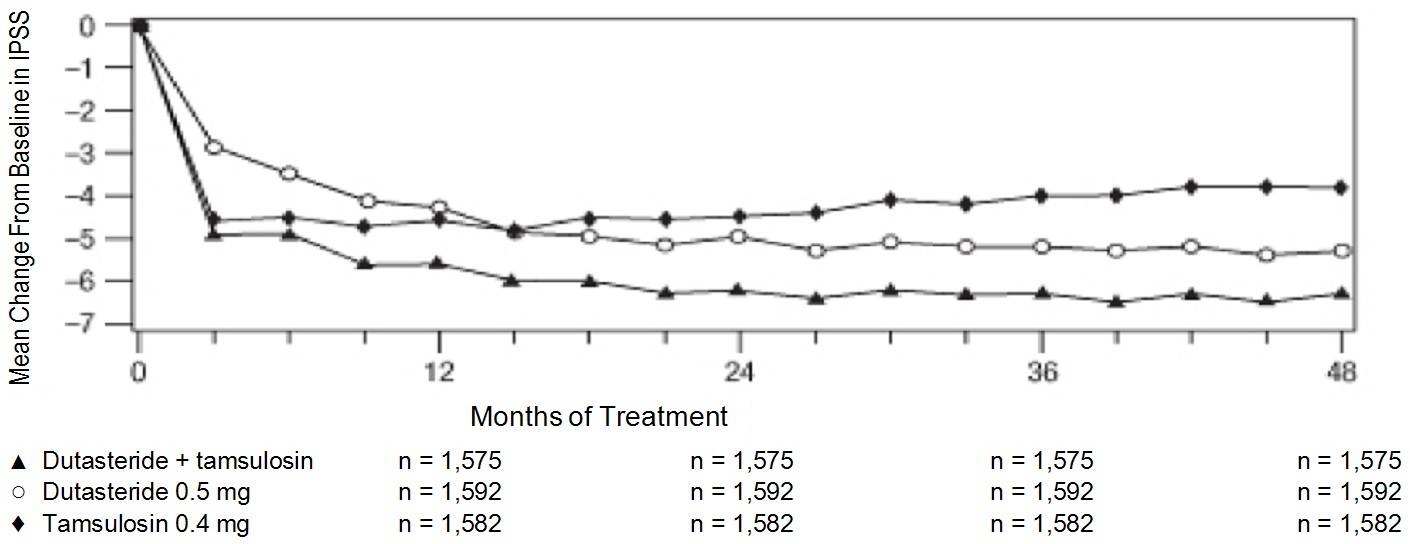
Effect on Acute Urinary Retention (AUR) or the Need for BPH-Related Surgery
After 4 years of treatment, coadministration therapy with dutasteride and tamsulosin did not provide benefit over dutasteride monotherapy in reducing the incidence of AUR or BPH-related surgery.
In separate 2-year randomized, double-blind trials, compared with placebo, dutasteride monotherapy was associated with a statistically significantly lower incidence of AUR (1.8% for dutasteride versus 4.2% for placebo; 57% reduction in risk) and with a statistically significantly lower incidence of BPH-related surgery (2.2% for dutasteride versus. 4.1% for placebo; 48% reduction in risk).
Effect on Maximum Urine Flow Rate
The baseline Q maxwas approximately 10.7 mL/sec for each treatment group. Coadministration therapy was statistically superior to each of the monotherapy treatments in increasing Q maxat Month 24, the primary time point for this endpoint. At Month 24, the mean increases from baseline (±SD) in Q maxwere 2.4 (±5.26) mL/sec for coadministration group, 1.9 (±5.10) mL/sec for dutasteride, and 0.9 (±4.57) mL/sec for tamsulosin, with a mean difference between coadministration and dutasteride of 0.5 mL/sec ( P= 0.003; [95% CI: 0.17, 0.84]), and between coadministration and tamsulosin of 1.5 mL/sec ( P<0.001; [95% CI: 1.19, 1.86]). This difference was seen by Month 6 and continued through Month 24. See Figure 2.
The additional improvement in Q maxof coadministration therapy over dutasteride monotherapy was no longer statistically significant at Month 48.
Figure 2. Q maxChange from Baseline over a 24-Month Period (Randomized, Double-blind, Parallel-group Trial [CombAT Trial])
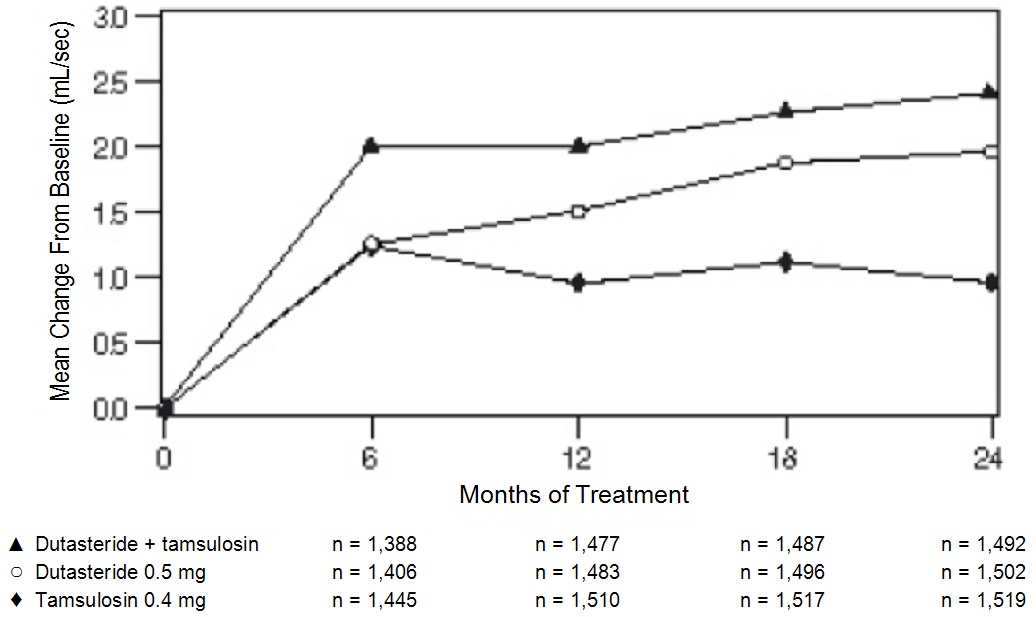
Effect on Prostate Volume
The mean prostate volume at trial entry was approximately 55 cc. At Month 24, the primary time point for this endpoint, the mean percent changes from baseline (±SD) in prostate volume were -26.9% (±22.57) for coadministration therapy, -28.0% (±24.88) for dutasteride, and 0% (±31.14) for tamsulosin, with a mean difference between coadministration and dutasteride of 1.1% ( P= NS; [95% CI: -0.6, 2.8]), and between coadministration and tamsulosin of -26.9% ( P<0.001; [95% CI: -28.9, -24.9]). Similar changes were seen at Month 48: -27.3% (±24.91) for coadministration therapy, -28.0% (±25.74) for dutasteride, and +4.6% (±35.45) for tamsulosin.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Dutasteride and tamsulosin hydrochloride capsules, containing 0.5 mg dutasteride and 0.4 mg tamsulosin hydrochloride, are blue, opaque cap imprinted with “C280” and white, opaque body imprinted with “0.5/0.4” in black ink containing white to off-white spherical shaped pellets and one oblong, opaque yellow softgel capsule printed with “C300” in black ink. They are available in bottles with child-resistant closures as follows:
Bottle of 30 (NDC: 42291-031-30).
Storeat 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Capsules may become deformed and/or discolored if kept at high temperatures.
Dutasteride is absorbed through the skin. Dutasteride and tamsulosin hydrochloride capsules should not be handled by females who are pregnant or who could become pregnant because of the potential for absorption of dutasteride and the subsequent potential risk to a developing male fetus [seeWarnings and Precautions ( 5.6) ].
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Orthostatic Hypotension
Inform patients about the possible occurrence of symptoms related to orthostatic hypotension, such as dizziness and vertigo, and the potential risk of syncope when taking dutasteride and tamsulosin hydrochloride capsules. Caution patients starting treatment with dutasteride and tamsulosin hydrochloride capsules to avoid situations where injury could result should syncope occur (e.g., driving, operating machinery, performing hazardous tasks). Advise patients to sit or lie down at the first signs of orthostatic hypotension [seeWarnings and Precautions ( 5.1) ].
Drug Interactions
Advise patients that dutasteride and tamsulosin hydrochloride capsules should not be used in combination with strong inhibitors of CYP3A4 [see Warnings and Precautions ( 5.2), Drug Interactions ( 7.1) ].
PSA Monitoring
Inform patients that dutasteride and tamsulosin hydrochloride capsules reduces serum PSA levels by approximately 50% within 3 to 6 months of therapy, although it may vary for each individual. For patients undergoing PSA screening, increases in PSA levels while on treatment with dutasteride and tamsulosin hydrochloride capsules may signal the presence of prostate cancer and should be evaluated by a healthcare provider [seeWarnings and Precautions ( 5.3) ].
Increased Risk of High-Grade Prostate Cancer
Inform patients that there was an increase in high-grade prostate cancer in men treated with 5-alpha-reductase inhibitors (which are indicated for BPH treatment), including dutasteride, which is a component of dutasteride and tamsulosin hydrochloride capsules, compared with those treated with placebo in trials looking at the use of these drugs to reduce the risk of prostate cancer [seeIndications and Usage ( 1.2), Warnings and Precautions ( 5.4), Adverse Reactions ( 6.1) ].
Transdermal Exposure of Dutasteride and Tamsulosin hydrochlorideinPregnant or Potentially Pregnant Females -Risk to Male Fetus
Inform patients that dutasteride and tamsulosin hydrochloride capsules should not be handled by females who are pregnant or may potentially be pregnant because of the potential for absorption of dutasteride and the subsequent potential risk to a developing male fetus. Dutasteride can be absorbed through the skin and could result in unintended fetal exposure. If a pregnant or potentially pregnant female comes in contact with leaking dutasteride and tamsulosin hydrochloride capsules, the contact area should be washed immediately with soap and water [seeWarnings and Precautions (5.6), Use in Specific Populations (8.1)].
Effects on Semen Parameters
Advise men that dutasteride and tamsulosin hydrochloride capsules may affect sperm characteristics but the effect on fertility is unknown [seeWarnings and Precautions (5.11), Use in Specific Populations (8.3)].
Administration Instructions
Dutasteride and tamsulosin hydrochloride capsules should be swallowed whole and not chewed, crushed, or opened. Dutasteride and tamsulosin hydrochloride capsules may become deformed and/or discolored if kept at high temperatures. If this occurs, capsules should not be used.
Priapism
Inform patients about the possibility of priapism as a result of treatment with dutasteride and tamsulosin hydrochloride capsules or other alpha-adrenergic-antagonist-containing medications. Inform patients that this reaction is extremely rare, but can lead to permanent erectile dysfunction if not brought to immediate medical attention [seeWarnings and Precautions (5.7)].
Blood Donation
Inform men treated with dutasteride and tamsulosin hydrochloride capsules that they should not donate blood until at least 6 months following their last dose to prevent pregnant females from receiving dutasteride through blood transfusion [seeWarnings and Precautions (5.8)]. Serum levels of dutasteride are detectable for 4 to 6 months after treatment ends [seeClinical Pharmacology (12.3)].
Intraoperative Floppy Iris Syndrome (IFIS)
Advise patients considering cataract or glaucoma surgery to tell their ophthalmologist that they take or have taken dutasteride and tamsulosin hydrochloride capsules, an alpha adrenergic antagonist-containing product [seeWarnings and Precautions (5.9)].
For Patient Information Leaflet, please visit www.avkare.com.
-
PATIENT INFORMATION
Dutasteride (doo tas' ter ide) and Tamsulosin Hydrochloride (tam soo' loe sin hye'' droe klor' ide) Capsules
Dutasteride and tamsulosin hydrochloride capsules are for use by men only.
What are dutasteride and tamsulosin hydrochloride capsules?
Dutasteride and tamsulosin hydrochloride capsules are a prescription medicine that contains 2 medicines: dutasteride and tamsulosin. Dutasteride and tamsulosin hydrochloride capsules are used to treat the symptoms of benign prostatic hyperplasia (BPH) in men with an enlarged prostate. The 2 medications in dutasteride and tamsulosin hydrochloride capsules work in different ways to improve symptoms of BPH. Dutasteride shrinks the enlarged prostate and tamsulosin relaxes muscles in the prostate and neck of the bladder. These 2 medications, when used together, can improve symptoms of BPH better than either medication when used alone.
Do not take dutasteride and tamsulosin hydrochloride capsules if you are:
- pregnant or may be pregnant. Dutasteride and tamsulosin hydrochloride capsules may harm your unborn baby. Pregnant females should not touch dutasteride and tamsulosin hydrochloride capsules. If a female who is pregnant with a male baby gets enough dutasteride and tamsulosin hydrochloride capsules in her body by swallowing or touching dutasteride and tamsulosin hydrochloride capsules, the male baby may be born with sex organs that are not normal. If a pregnant female comes in contact with leaking dutasteride and tamsulosin hydrochloride capsules, the contact area should be washed immediately with soap and water.
- allergic to dutasteride, tamsulosin, or any of the ingredients in dutasteride and tamsulosin hydrochloride capsules. See the end of this leaflet for a complete list of ingredients in dutasteride and tamsulosin hydrochloride capsules.
- taking another medicine that contains an alpha-blocker.
- allergic to other 5-alpha-reductase inhibitors, for example, PROSCAR (finasteride) tablets.
Before you take dutasteride and tamsulosin hydrochloride capsules, tell your healthcare provider about all of your medical conditions, including if you:
- have a history of low blood pressure
- take medicines to treat high blood pressure
- plan to have cataract or glaucoma surgery
- have liver problems
- are allergic to sulfa medications
- have any other medical conditions
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Dutasteride and tamsulosin hydrochloride capsules and other medicines may affect each other, causing side effects. Dutasteride and tamsulosin hydrochloride capsules may affect the way other medicines work, and other medicines may affect how dutasteride and tamsulosin hydrochloride capsules works.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take dutasteride and tamsulosin hydrochloride capsules?
- Take dutasteride and tamsulosin hydrochloride capsules exactly as your healthcare provider tells you to take it.
- Swallow dutasteride and tamsulosin hydrochloride capsules whole. Do not crush, chew, or open dutasteride and tamsulosin hydrochloride capsules because the contents of the capsule may irritate your lips, mouth, or throat.
- Take your dutasteride and tamsulosin hydrochloride capsules 1 time each day, about 30 minutes after the same meal every day. For example, you may take dutasteride and tamsulosin hydrochloride capsules 30 minutes after dinner every day.
- If you miss a dose, you can take it later that same day, 30 minutes after a meal. Do not take 2 dutasteride and tamsulosin hydrochloride capsules in the same day. If you stop or forget to take dutasteride and tamsulosin hydrochloride capsules for several days, talk with your healthcare provider before starting again.
- If you take too much dutasteride and tamsulosin hydrochloride capsules, call your healthcare provider or go to the nearest hospital emergency room right away.
What should I avoid while taking dutasteride and tamsulosin hydrochloride capsules?
- Avoid driving, operating machinery, or other dangerous activities when starting treatment with dutasteride and tamsulosin hydrochloride capsules until you know how dutasteride and tamsulosin hydrochloride capsules affects you. Dutasteride and tamsulosin hydrochloride capsules can cause a sudden drop in your blood pressure, especially at the start of treatment. A sudden drop in blood pressure may cause you to faint, feel dizzy or lightheaded.
- You should not donate blood while taking dutasteride and tamsulosin hydrochloride capsules or for 6 months after you have stopped dutasteride and tamsulosin hydrochloride capsules. This is important to prevent pregnant females from receiving dutasteride and tamsulosin hydrochloride capsules through blood transfusions.
What are the possible side effects of dutasteride and tamsulosin hydrochloride capsules?
Dutasteride and tamsulosin hydrochloride capsulesmay cause serious side effects including:
- Decreased blood pressure.Dutasteride and tamsulosin hydrochloride capsules may cause a sudden drop in your blood pressure upon standing from a sitting or lying position, especially at the start of treatment. Symptoms of low blood pressure may include:
- fainting
- dizziness
- feeling lightheaded
- Rare and serious allergic reactions, including:
- swelling of your face, tongue, or throat
- difficulty breathing
- serious skin reactions, such as skin peeling
Get medical help right away if you have these serious allergic reactions.
- Higher chance of a more serious form of prostate cancer.
- Eye problems during cataract or glaucoma surgery.During cataract or glaucoma surgery, a condition called Intraoperative Floppy Iris Syndrome (IFIS) can happen if you take or have taken dutasteride and tamsulosin hydrochloride capsules in the past. If you need to have cataract or glaucoma surgery, tell your surgeon if you take or have taken dutasteride and tamsulosin hydrochloride capsules.
- A painful erection that will not go away.Rarely, dutasteride and tamsulosin hydrochloride capsules can cause a painful erection (priapism), which cannot be relieved by having sex. If this happens, get medical help right away. If priapism is not treated, there could be lasting damage to your penis, including not being able to have an erection.
The most common side effects of dutasteride and tamsulosin hydrochloride capsules include:
- ejaculation problems*
- trouble getting or keeping an erection (impotence)*
- a decrease in sex drive (libido)*
- dizziness
- enlarged or painful breasts. If you notice breast lumps or nipple discharge, you should talk to your healthcare provider.
- runny nose
*Some of these events may continue after you stop taking dutasteride and tamsulosin hydrochloride capsules.
Depressed mood has been reported in patients receiving dutasteride, an ingredient of dutasteride and tamsulosin hydrochloride capsules.
Dutasteride, an ingredient of dutasteride and tamsulosin hydrochloride capsules, has been shown to reduce sperm count, semen volume, and sperm movement. However, the effect of dutasteride and tamsulosin hydrochloride capsules on male fertility is not known.
Prostate-Specific Antigen (PSA) Test:Your healthcare provider may check you for other prostate problems, including prostate cancer before you start and while you take dutasteride and tamsulosin hydrochloride capsules. A blood test called PSA (prostate-specific antigen) is sometimes used to see if you might have prostate cancer. Dutasteride and tamsulosin hydrochloride capsules will reduce the amount of PSA measured in your blood. Your healthcare provider is aware of this effect and can still use PSA to see if you might have prostate cancer. Increases in your PSA levels while on treatment with dutasteride and tamsulosin hydrochloride capsules (even if the PSA levels are in the normal range) should be evaluated by your healthcare provider. These are not all the possible side effects with dutasteride and tamsulosin hydrochloride capsules. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store dutasteride and tamsulosin hydrochloride capsules?
- Store dutasteride and tamsulosin hydrochloride capsules at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
- Dutasteride and tamsulosin hydrochloride capsules may become deformed and/or discolored if kept at high temperatures.
- Do not use or touch dutasteride and tamsulosin hydrochloride capsules if your capsules are deformed, discolored, or leaking.
- Safely throw away medicine that is no longer needed.
Keep dutasteride and tamsulosin hydrochloride capsules and all medicines out of the reach of children.
General information about the safe and effective use of dutasteride and tamsulosin hydrochloride capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use dutasteride and tamsulosin hydrochloride capsules for a condition for which it was not prescribed. Do not give dutasteride and tamsulosin hydrochloride capsules to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about dutasteride and tamsulosin hydrochloride capsules that is written for health professionals.
For more information, call 1-855-361-3993.
What are the ingredients in dutasteride and tamsulosin hydrochloride capsules?
Active ingredients:dutasteride and tamsulosin hydrochloride
Inactive ingredients:butylated hydroxytoluene, ethylcellulose, gelatin, glycerin, lecithin, medium chain triglycerides, methacrylic acid copolymer, mono- and di-glycerides of capryl/capric acid, polyethylene glycol, sugar spheres, talc, triethyl citrate, iron oxide yellow, hypromellose, titanium dioxide, D&C yellow #10, iron oxide black, FD&C blue #2, FD&C blue #1, propylene glycol, FD&C red #40, shellac, polyvinyl acetate phthalate, macrogol, ammonium hydroxide.
Trademarks are the properties of their respective owners.
This Patient Information has been approved by the U.S. Food and Drug Administration.
For Patient Information Leaflet, please visit please visit www.avkare.com
Manufactured for:
AvKARE
Pulaski, TN 38478
Mfg. Rev. 10/21
AV 11/23 (M)
- PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DUTASTERIDE AND TAMSULOSIN HYDROCHLORIDE
dutasteride and tamsulosin hydrochloride capsules capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 42291-031(NDC:10370-280) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DUTASTERIDE (UNII: O0J6XJN02I) (DUTASTERIDE - UNII:O0J6XJN02I) DUTASTERIDE 0.5 mg TAMSULOSIN HYDROCHLORIDE (UNII: 11SV1951MR) (TAMSULOSIN - UNII:G3P28OML5I) TAMSULOSIN HYDROCHLORIDE 0.4 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) ETHYLCELLULOSE (10 MPA.S) (UNII: 3DYK7UYZ62) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) HYPROMELLOSES (UNII: 3NXW29V3WO) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) FD&C RED NO. 40 (UNII: WZB9127XOA) SHELLAC (UNII: 46N107B71O) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) Product Characteristics Color white (body) , blue (cap) Score no score Shape CAPSULE Size 23mm Flavor Imprint Code C280;0504;C300 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 42291-031-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 11/15/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202509 11/15/2023 Labeler - AvKARE (796560394)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
