Acetaminophen by Cardinal Health ACETAMINOPHEN solution
Acetaminophen by
Drug Labeling and Warnings
Acetaminophen by is a Otc medication manufactured, distributed, or labeled by Cardinal Health. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each 5 mL teaspoonful)
- Purpose
- Uses
-
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take:
- adult takes more than 6 doses in 24 hours, which is the maximum daily amount
- child takes more than 5 doses in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert
Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription).
- if you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- for more than 10 days for pain unless directed by a doctor
- for more than 3 days for fever unless directed by a doctor
- if you are allergic to acetaminophen or any of the inactive ingredients of this product
Stop use and ask a doctor if
- symptoms do not improve
- new symptoms occur
- pain or fever persists or gets worse
Overdose warning
Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222). Quick medical attention is critical for adults as well as children even if you do not notice any signs or symptoms.
-
Directions
- do not take more than directed (see overdose warning)
age dose adults and children 12 years of age and over
20.3 mL (650 mg) every 4 to 6 hours
not to exceed 6 doses in a 24-hour periodchildren 6 to under 12 years of age
10.15 mL (325 mg) every 4 hours
not to exceed 5 doses in a 24-hour periodchildren 4 to under 6 years of age
7.5 mL (240 mg) every 4 hours
not to exceed 5 doses in a 24-hour periodchildren 2 to under 4 years of age
5 mL (160 mg) every 4 hours
not to exceed 5 doses in a 24-hour periodchildren under 2 years of age
consult a doctor
-
Other information
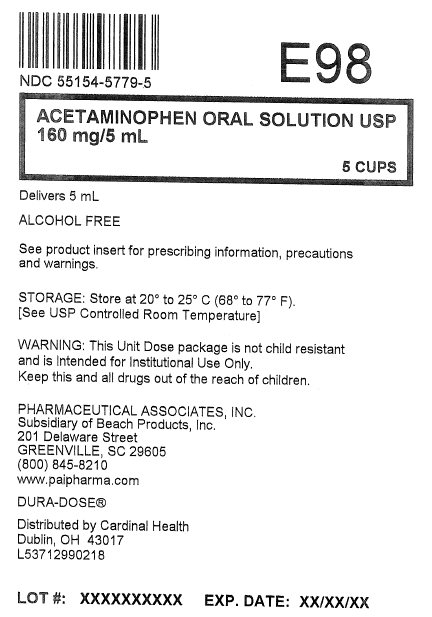
■ sodium content: 2 mg/5 mL
■ store at 20° to 25°C (68° to 77°F). [See USP controlled room temperature]
■ keep tightly closed ■ protect from light
■ a red, cherry flavored solution supplied in the following oral dosage forms:
Overbagged with 5 x 5 mL unit dose cup per bag, NDC: 55154-5779-5
- Inactive ingredients
- Questions or comments?
- MANUFACTURED BY:
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN
acetaminophen solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 55154-5779(NDC:0121-0657) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SORBITOL (UNII: 506T60A25R) SUCROSE (UNII: C151H8M554) Product Characteristics Color red Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 55154-5779-5 5 in 1 BAG 08/01/2007 1 5 mL in 1 CUP, UNIT-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 08/01/2007 Labeler - Cardinal Health (603638201)
Trademark Results [Acetaminophen]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ACETAMINOPHEN 85615223 not registered Dead/Abandoned |
General Merchandise importers and Expoters 2012-05-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.