MEMANTINE AND DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE- memantine and donepezil capsule, extended release
Memantine and Donepezil Hydrochlorides Extended-release by
Drug Labeling and Warnings
Memantine and Donepezil Hydrochlorides Extended-release by is a Prescription medication manufactured, distributed, or labeled by Amneal Pharmaceuticals NY LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use MEMANTINE and DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE CAPSULES safely and effectively. See full prescribing information for MEMANTINE and DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE CAPSULES.
MEMANTINE and DONEPEZIL HYDROCHLORIDES extended-release capsules, for oral use
Initial U.S. Approval: 2014INDICATIONS AND USAGE
Memantine and donepezil hydrochlorides extended-release capsules are a combination of memantine hydrochloride, an NMDA receptor antagonist, and donepezil hydrochloride, an acetylcholinesterase inhibitor, indicated for the treatment of moderate to severe dementia of the Alzheimer’s type in patients stabilized on 10 mg of donepezil hydrochloride once daily. (1)
DOSAGE AND ADMINISTRATION
- For patients on donepezil hydrochloride 10 mg only, the recommended starting dose of memantine and donepezil hydrochlorides extended-release capsules is 7 mg/10 mg, taken once daily in the evening. The dose should be increased in 7 mg increments to the recommended maintenance dose of 28 mg/10 mg. The minimum recommended interval between dose increases is one week. (2.1)
- Patients on memantine hydrochloride (10 mg twice daily or 28 mg extended-release once daily) and donepezil hydrochloride 10 mg once daily can be switched to memantine and donepezil hydrochlorides extended-release capsules 28 mg/10 mg, taken once daily in the evening. (2.1)
- Memantine and donepezil hydrochlorides extended-release capsules can be taken with or without food, whole or sprinkled on applesauce; do not divide, chew, or crush. (2.2)
- Severe renal impairment: the recommended maintenance dose for memantine and donepezil hydrochlorides extended-release capsules is 14 mg/10 mg once daily in the evening. (2.3)
DOSAGE FORMS AND STRENGTHS
Memantine and donepezil hydrochlorides extended-release capsules:
CONTRAINDICATIONS
Memantine and donepezil hydrochlorides extended-release capsules are contraindicated in patients with known hypersensitivity to memantine hydrochloride, donepezil hydrochloride, piperidine derivatives, or to any excipients used in the formulation. (4)
WARNINGS AND PRECAUTIONS
- Memantine and donepezil hydrochlorides extended-release is likely to exaggerate succinylcholine-type muscle relaxation during anesthesia. (5.1)
- Memantine and donepezil hydrochlorides extended-release may have vagotonic effects on the sinoatrial and atrioventricular nodes manifesting as bradycardia or heart block. (5.2)
- Monitor patients for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers. (5.3)
- Memantine and donepezil hydrochlorides extended-release can cause diarrhea, nausea, and vomiting. (5.4)
- Memantine and donepezil hydrochlorides extended-release may cause bladder outflow obstructions. (5.5)
- Conditions that raise urine pH may decrease the urinary elimination of memantine, resulting in increased plasma levels of memantine. (5.5, 7.1)
ADVERSE REACTIONS
- The most common adverse reactions, occurring at a frequency of at least 5% and greater than placebo with memantine hydrochloride extended-release 28 mg/day, were headache, diarrhea, and dizziness. (6.1)
- The most common adverse reactions occurring at a frequency of at least 5% in patients receiving donepezil and at twice or more the placebo rate, include diarrhea, anorexia, vomiting, nausea, and ecchymosis. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Amneal Pharmaceuticals at 1-877-835-5472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Combined use with NMDA antagonists: use with caution. (7.2)
- Memantine and donepezil hydrochlorides extended-release may interfere with anticholinergic medications. (7.4)
- Concomitant administration of succinylcholine, similar neuromuscular blocking agents, or cholinergic agonists may lead to synergistic effect. (7.5)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 5/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
2.2 Administration Information
2.3 Dosing in Patients with Severe Renal Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Anesthesia
5.2 Cardiovascular Conditions
5.3 Peptic Ulcer Disease and Gastrointestinal Bleeding
5.4 Nausea and Vomiting
5.5 Genitourinary Conditions
5.6 Seizures
5.7 Pulmonary Conditions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Use of Memantine with Drugs That Make the Urine Alkaline
7.2 Use of Memantine with Other N-methyl-D-aspartate (NMDA) Antagonists
7.3 Effect of Other Drugs on the Metabolism of Donepezil
7.4 Use of Donepezil with Anticholinergics
7.5 Use of Donepezil with Cholinomimetics and Other Cholinesterase Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
The recommended dose of memantine and donepezil hydrochlorides extended-release capsules is 28 mg/10 mg once daily.
For patients stabilized on donepezil and not currently on memantine:
For patients stabilized on donepezil hydrochloride 10 mg and not currently on memantine hydrochloride, the recommended starting dose of memantine and donepezil hydrochlorides extended-release capsules is 7 mg/10 mg, taken once a day in the evening. The dose should be increased in 7 mg increments of the memantine hydrochloride component to the recommended maintenance dose of 28 mg/10 mg once daily. The minimum recommended interval between dose increases is one week. The dose should only be increased if the previous dose has been well tolerated. The maximum dose is 28 mg/10 mg once daily.
For patients stabilized on both donepezil and memantine:
Patients stabilized on memantine hydrochloride (10 mg twice daily or 28 mg extended-release once daily) and donepezil hydrochloride 10 mg once daily can be switched to memantine and donepezil hydrochlorides extended-release capsules 28 mg/10 mg, taken once a day in the evening. Patients should start memantine and donepezil hydrochlorides extended-release capsules the day following the last dose of memantine and donepezil hydrochlorides extended-release administered separately.
If a patient misses a single dose of memantine and donepezil hydrochlorides extended-release capsules, the next dose should be taken as scheduled, without doubling up the dose.
2.2 Administration Information
Memantine and donepezil hydrochlorides extended-release capsules can be taken with or without food. Memantine and donepezil hydrochlorides extended-release capsules can be taken intact or may be opened, sprinkled on applesauce, and swallowed without chewing. The entire contents of each memantine and donepezil hydrochlorides extended-release capsule should be consumed; the dose should not be divided.
Except when opened and sprinkled on applesauce, as described above, memantine and donepezil hydrochlorides extended-release capsules should be swallowed whole. Memantine and donepezil hydrochlorides extended-release capsules should not be divided, chewed, or crushed.
2.3 Dosing in Patients with Severe Renal Impairment
For patients stabilized on donepezil and not currently on memantine:
For patients with severe renal impairment (creatinine clearance 5 to 29 mL/min, based on the Cockcroft-Gault equation), stabilized on donepezil hydrochloride 10 mg once daily and not currently on memantine hydrochloride, the recommended starting dose of memantine and donepezil hydrochlorides extended-release capsules is 7 mg/10 mg taken once a day in the evening. The dose should be increased to the recommended maintenance dose of 14 mg/10 mg once daily in the evening after a minimum of one week [see Use in Specific Populations (8.6)].
For patients stabilized on both donepezil and memantine:
Patients with severe renal impairment, stabilized on memantine hydrochloride (5 mg twice daily or 14 mg extended-release once daily) and donepezil hydrochloride 10 mg once daily, can be switched to memantine and donepezil hydrochlorides extended-release capsules 14 mg/10 mg, taken once daily in the evening.
-
3 DOSAGE FORMS AND STRENGTHS
Memantine and donepezil hydrochlorides extended-release capsules:
- 7 mg memantine hydrochloride extended-release/10 mg donepezil hydrochloride capsules: Two piece hard gelatin capsules with green opaque cap and white opaque body filled with white to off- white pellets. Imprinted in black ink AN on the cap and imprinted in black ink 1552 on the body.
- 14 mg memantine hydrochloride extended-release/10 mg donepezil hydrochloride capsules: Two-piece hard gelatin capsules with green opaque cap and green opaque body filled with white to off-white pellets. Imprinted in white ink AN on the cap and imprinted in white ink 1248 on the body.
- 21 mg memantine hydrochloride extended-release/10 mg donepezil hydrochloride capsules: Two piece hard gelatin capsules with yellow opaque cap and white opaque body filled with white to off-white pellets. Imprinted in black ink AN on the cap and imprinted in black ink 1553 on the body.
- 28 mg memantine hydrochloride extended-release/10 mg donepezil hydrochloride capsules: Two-piece hard gelatin capsules with yellow opaque cap and yellow opaque body filled with white to off-white pellets. Imprinted in black ink AN on the cap and imprinted in black ink 1247 on the body.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Anesthesia
Donepezil hydrochloride, an active ingredient in memantine and donepezil hydrochlorides extended-release capsules, as a cholinesterase inhibitor, is likely to exaggerate succinylcholine-type muscle relaxation during anesthesia.
5.2 Cardiovascular Conditions
Because of their pharmacological action, cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes. This effect may manifest as bradycardia or heart block in patients both with and without known underlying cardiac conduction abnormalities. Syncopal episodes have been reported in association with the use of donepezil hydrochloride, an active ingredient in memantine and donepezil hydrochlorides extended-release capsules.
5.3 Peptic Ulcer Disease and Gastrointestinal Bleeding
Through their primary action, cholinesterase inhibitors may be expected to increase gastric acid secretion due to increased cholinergic activity. Clinical studies of donepezil hydrochloride in a dose of 5 mg/day to 10 mg/day have shown no increase, relative to placebo, in the incidence of either peptic ulcer disease or gastrointestinal bleeding. Patients treated with memantine and donepezil hydrochlorides extended-release should be monitored closely for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e.g., those with a history of ulcer disease or those receiving concurrent nonsteroidal anti-inflammatory drugs (NSAIDs).
5.4 Nausea and Vomiting
Donepezil hydrochloride, when initiated, as a predictable consequence of its pharmacological properties, has been shown to produce diarrhea, nausea, and vomiting. Although in most cases, these effects have been mild and transient, sometimes lasting one to three weeks, and have resolved during continued use of donepezil hydrochloride, patients should be observed closely at the initiation of treatment.
5.5 Genitourinary Conditions
Although not observed in clinical trials of donepezil hydrochloride, an active ingredient in memantine and donepezil hydrochlorides extended-release capsules, cholinomimetics may cause bladder outflow obstruction.
Conditions that raise urine pH may decrease the urinary elimination of memantine, an active ingredient in memantine and donepezil hydrochlorides extended-release capsules, resulting in increased plasma levels of memantine [see Drug Interactions (7.1)].
5.6 Seizures
Cholinomimetics, including donepezil hydrochloride, an active ingredient in memantine and donepezil hydrochlorides extended-release capsules, are believed to have some potential to cause generalized convulsions. However, seizure activity also may be a manifestation of Alzheimer’s disease.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed below and elsewhere in the labeling.
- Cardiovascular Conditions [see Warnings and Precautions (5.2)]
- Peptic Ulcer Disease and Gastrointestinal Bleeding [see Warnings and Precautions (5.3)]
- Nausea and Vomiting [see Warnings and Precautions (5.4)]
- Genitourinary Conditions [see Warnings and Precautions (5.5)]
- Seizures [see Warnings and Precautions (5.6)]
- Pulmonary Conditions [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Memantine Hydrochloride
Memantine hydrochloride extended-release was evaluated in a double-blind, placebo-controlled trial in 676 patients with moderate to severe dementia of the Alzheimer’s type (341 patients treated with memantine 28 mg/day dose and 335 patients treated with placebo) for a treatment period up to 24 weeks. Of the patients randomized, 236 treated with memantine 28 mg/day and 227 treated with placebo were on a stable dose of donepezil for 3 months prior to screening.
Adverse Reactions Leading to Discontinuation with Memantine Hydrochloride
In the placebo-controlled clinical trial of memantine hydrochloride extended-release, the proportion of patients in the memantine hydrochloride extended-release 28 mg/day dose group and in the placebo group who discontinued treatment due to adverse reactions was 10% and 6%, respectively. The most common adverse reaction in the memantine hydrochloride extended-release treated group that led to treatment discontinuation was dizziness, at a rate of 1.5%.
Most Common Adverse Reactions with Memantine Hydrochloride
The most common adverse reactions with memantine hydrochloride extended-release in patients with moderate to severe Alzheimer’s disease, defined as those occurring at a frequency of at least 5% in the memantine hydrochloride extended-release group and at a higher frequency than placebo, were headache, diarrhea, and dizziness.
Table 1 lists adverse reactions that occurred at an incidence of ≥ 2% in the memantine hydrochloride extended-release treated group and occurred at a rate greater than placebo.
Table 1: Adverse reactions with memantine hydrochloride extended-release in patients with moderate to severe Alzheimer’s disease
Adverse Reaction
Placebo
(n = 335)
%
Memantine hydrochloride extended-release
28 mg
(n = 341)
%
Gastrointestinal Disorders
Diarrhea
4
5
Constipation
1
3
Abdominal pain
1
2
Vomiting
1
2
Infections and Infestations
Influenza
3
4
Investigations
Increased weight
1
3
Musculoskeletal and Connective Tissue Disorders
Back pain
1
3
Nervous System Disorders
Headache
5
6
Dizziness
1
5
Somnolence
1
3
Psychiatric Disorders
Anxiety
3
4
Depression
1
3
Aggression
1
2
Renal and Urinary Disorders
Urinary incontinence
1
2
Vascular Disorders
Hypertension
2
4
Hypotension
1
2
Donepezil hydrochloride
Adverse Reactions Leading to Discontinuation with Donepezil Hydrochloride
In controlled clinical trials of donepezil hydrochloride, the rate of discontinuation due to adverse reactions for patients treated with donepezil hydrochloride was approximately 12%, compared to 7% for patients treated with placebo. The most common adverse reactions leading to discontinuation, defined as those occurring in at least 2% of donepezil hydrochloride patients and at twice or more the incidence seen with placebo, were anorexia (2%), nausea (2%), diarrhea (2%) and urinary tract infection (2%).
Most Common Adverse Reactions with Donepezil Hydrochloride
The most common adverse reactions reported with donepezil hydrochloride in controlled clinical trials in patients with severe Alzheimer’s disease, defined as those occurring at a frequency of at least 5% in the donepezil hydrochloride group and at twice or more the placebo rate, were diarrhea, anorexia, vomiting, nausea, and ecchymosis. The most common adverse reactions reported with donepezil hydrochloride in controlled clinical trials in patients with mild to moderate Alzheimer’s disease were insomnia, muscle cramp, and fatigue.
Table 2 lists adverse reactions that occurred at an incidence of ≥ 2% in the donepezil hydrochloride group and at a rate greater than placebo in controlled trials in patients with severe Alzheimer’s disease.
Table 2: Adverse reactions with donepezil hydrochloride in patients with severe Alzheimer’s disease
Body System/Adverse Event
Placebo (n = 392) %
Donepezil hydrochloride
10 mg/day
(n = 501)
%
Percent of Patients with any Adverse Event
73
81
Body as a Whole
Accident
12
13
Infection
9
11
Headache
3
4
Pain
2
3
Back pain
2
3
Fever
1
2
Chest pain
< 1
2
Cardiovascular System
Hypertension
2
3
Hemorrhage
1
2
Syncope
1
2
Digestive System
Diarrhea
4
10
Vomiting
4
8
Anorexia
4
8
Nausea
2
6
Hemic and Lymphatic System
Ecchymosis
2
5
Metabolic and Nutritional Systems
Increased creatine phosphokinase
1
3
Dehydration
1
2
Hyperlipemia
< 1
2
Nervous System
Insomnia
4
5
Hostility
2
3
Nervousness
2
3
Hallucinations
1
3
Somnolence
1
2
Dizziness
1
2
Depression
1
2
Confusion
1
2
Emotional lability
1
2
Personality disorder
1
2
Skin and Appendages
Eczema
2
3
Urogenital System
Urinary incontinence
1
2
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of memantine and donepezil hydrochlorides extended-release. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Memantine Hydrochloride
Acute renal failure, agranulocytosis, cardiac failure congestive, hepatitis, leukopenia (including neutropenia), pancreatitis, pancytopenia, Stevens-Johnson syndrome, suicidal ideation, thrombocytopenia, and thrombotic thrombocytopenic purpura.
Donepezil Hydrochloride
Abdominal pain, agitation, cholecystitis, confusion, convulsions, hallucinations, heart block (all types), hemolytic anemia, hepatitis, hyponatremia, neuroleptic malignant syndrome, pancreatitis, and rash.
-
7 DRUG INTERACTIONS
7.1 Use of Memantine with Drugs That Make the Urine Alkaline
The clearance of memantine was reduced by about 80% under alkaline urine conditions at pH 8. Therefore,
alterations of urine pH towards the alkaline condition may lead to an accumulation of the
drug with a possible increase in adverse reactions. Urine pH is altered by diet, drugs (e.g., carbonic anhydrase inhibitors, sodium bicarbonate) and clinical state of the patient (e.g., renal tubular acidosis or severe infections of the urinary tract). Hence, memantine should be used with caution under these conditions.
7.2 Use of Memantine with Other N-methyl-D-aspartate (NMDA) Antagonists
The combined use of memantine hydrochloride with other NMDA antagonists (amantadine, ketamine, and dextromethorphan) has not been systematically evaluated and such use should be approached with caution.
7.3 Effect of Other Drugs on the Metabolism of Donepezil
Inhibitors of CYP3A4 (e.g., ketoconazole) and CYP2D6 (e.g., quinidine), inhibit donepezil metabolism in vitro. Whether there is a clinical effect of quinidine is not known. Inducers of CYP3A4 (e.g., phenytoin, carbamazepine, dexamethasone, rifampin, and phenobarbital) could increase the rate of elimination of donepezil.
7.4 Use of Donepezil with Anticholinergics
Because of their mechanism of action, cholinesterase inhibitors, including donepezil hydrochloride, have the potential to interfere with the activity of anticholinergic medications.
7.5 Use of Donepezil with Cholinomimetics and Other Cholinesterase Inhibitors
A synergistic effect may be expected when cholinesterase inhibitors, including donepezil hydrochloride, are given concurrently with succinylcholine, similar neuromuscular blocking agents, or cholinergic agonists such as bethanechol.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate data on the developmental risk associated with the use of memantine and donepezil hydrochlorides extended-release capsules or its active ingredients (memantine hydrochloride and donepezil hydrochloride) in pregnant women.
Adverse developmental effects (mortality and decreased body weight and skeletal ossification) were observed in the offspring of rats administered memantine or donepezil during pregnancy at doses associated with minimal maternal toxicity. These doses are higher than those used in humans at the recommended daily dose of memantine and donepezil hydrochlorides extended-release [see Data].
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
Data
Animal Data
Memantine Hydrochloride
Oral administration of memantine (2, 6, or 18 mg/kg/day) to rats during the period of organogenesis resulted in decreased skeletal ossification in fetuses at the highest dose tested. The higher no-effect dose for adverse developmental effects (6 mg/kg) is 2 times the dose of memantine at the recommended human daily dose [RHD] of memantine and donepezil hydrochlorides extended-release (28 mg memantine/10 mg donepezil) on a body surface area (mg/m2) basis.
Oral administration of memantine to rabbits (3, 10, or 30 mg/kg/day) during the period of organogenesis resulted in no adverse developmental effects. The highest dose tested is approximately 20 times the dose of memantine at the RHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis.
In rats, memantine (2, 6, or 18 mg/kg/day) was administered orally prior to and throughout mating and, in females, through the period of organogenesis or continuing throughout lactation to weaning. Decreased skeletal ossification in fetuses and decreased body weight in pups were observed at the highest dose tested. The higher no-effect dose for adverse developmental effects (6 mg/kg/day) is 2 times the dose of memantine at the RHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis.
Oral administration of memantine (2, 6, or 18 mg/kg/day) to rats from late gestation throughout lactation to weaning, resulted in decreased pup weights at the highest dose tested. The higher no-effect dose (6 mg/kg/day) is approximately 2 times the dose of memantine at the RHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis.
Donepezil Hydrochloride
Oral administration of donepezil to rats and rabbits during the period of organogenesis resulted in no adverse developmental effects. The highest doses (16 and 10 mg/kg/day, respectively) were approximately 15 and 7 times, respectively, the dose of donepezil at the RHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis.
Oral administration of donepezil (1, 3, or 10 mg/kg/day) to rats during late gestation and throughout lactation to weaning resulted in an increase in stillbirths and offspring mortality at the highest dose tested. The higher no-effect dose (3 mg/kg/day) is approximately 3 times the dose of donepezil at the RHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis.
8.2 Lactation
Risk Summary
There are no data on the presence of memantine or donepezil in human milk, the effects on the breastfed infant, or the effects of memantine and donepezil hydrochlorides extended-release capsules or its metabolites on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for memantine and donepezil hydrochlorides extended-release capsules and any potential adverse effects on the breastfed infant from memantine and donepezil hydrochlorides extended-release capsules or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of memantine and donepezil hydrochlorides extended-release in pediatric patients have not been established.
Memantine failed to demonstrate efficacy in two 12-week controlled clinical studies of 578 pediatric patients aged 6 to 12 years with autism spectrum disorders (ASD), including autism, Asperger’s disorder and Pervasive Development Disorder - Not Otherwise Specified (PDD-NOS). Memantine has not been studied in pediatric patients under 6 years of age or over 12 years of age. Memantine treatment was initiated at 3 mg/day and the dose was escalated to the target dose (weight-based) by week 6. Oral doses of memantine 3, 6, 9, or 15 mg extended-release capsules were administered once daily to patients with weights < 20 kg, 20 to 39 kg, 40 to 59 kg and ≥ 60 kg, respectively.
In a randomized, 12-week double-blind, placebo-controlled parallel study (Study A) in patients with autism, there was no statistically significant difference in the Social Responsiveness Scale (SRS) total raw score between patients randomized to memantine (n=54) and those randomized to placebo (n=53). In a 12-week responder-enriched randomized withdrawal study (Study B) in 471 patients with ASD, there was no statistically significant difference in the loss of therapeutic response rates between patients randomized to remain on full-dose memantine (n=153) and those randomized to switch to placebo (n=158).
The overall risk profile of memantine in pediatric patients was generally consistent with the known risk profile in adults [see Adverse Reactions (6.1)].
In Study A, the adverse reactions in the memantine group (n=56) that were reported in at least 5% of patients and at least twice the frequency of the placebo group (N=58) are listed in Table 3:
Table 3: Study A Commonly Reported Adverse Reactions With a Frequency ≥ 5% and Twice That of Placebo
Adverse Reaction
Memantine
N=56
Placebo
N=58
Cough
8.9%
3.4%
Influenza
7.1%
3.4%
Rhinorrhea
5.4%
0%
Agitation
5.4%
1.7%
Discontinuations due to adverse reactionsa
Aggression
3.6%
1.7%
Irritability
1.8%
3.4%
a Reported adverse reactions leading to discontinuation in more than one patient in either treatment group.
The adverse reactions that were reported in at least 5% of patients in the 12 to 48 week open-label study to identify responders to enroll in Study B are listed in Table 4:
Table 4: 12 to 48 Week Open Label Lead-In study to Study B Commonly Reported Adverse Reactions with a Frequency ≥ 5%
Adverse Reaction
Memantine
N=903
Headache
8%
Nasopharyngitis
6.3%
Pyrexia
5.8%
Irritability
5.4%
Discontinuations due to adverse reactionsa
Irritability
1.2%
Aggression
1%
a At least 1% incidence of adverse reactions leading to premature discontinuation.
In the randomized withdrawal study (Study B), the adverse reaction in patients randomized to placebo (n=160) and reported in at least 5% of patients and at twice the frequency of the full-dose memantine treatment group (n=157) was irritability (5% vs 2.5%).
In a juvenile animal study, male and female juvenile rats were administered memantine (15, 30, and 45 mg/kg/day) starting on postnatal day (PND) 14 through PND 70. Body weights were reduced at 45 mg/kg/day. Delays in sexual maturation were noted in male and female rats at doses ≥ 30 mg/kg/day. Memantine induced neuronal lesions in several areas of the brain on PND 15 and 17 at doses ≥ 30 mg/kg/day. Behavioral toxicity (decrease percent of auditory startle habituation) was noted for animals in the 45 mg/kg/day dose group. The 15 mg/kg/day dose was considered the No-Observed-Adverse-Effect-Level (NOAEL) for this study.
In a second juvenile rat toxicity study, male and female juvenile rats were administered memantine (1, 3, 8, 15, 30, and 45 mg/kg/day) starting on postnatal day (PND) 7 through PND 70. Due to early memantine-related mortality, the 30 and 45 mg/kg/day dose groups were terminated without further evaluation. Memantine induced apoptosis or neuronal degeneration in several areas of the brain on PND 8, 10, and 17 at a dose of 15 mg/kg/day. The NOAEL for apoptosis and neuronal degeneration was 8 mg/kg/day. Behavioral toxicity (effects on motor activity, auditory startle habituation, and learning and memory) was noted at doses ≥ 3 mg/kg/day during treatment, but was not seen after drug discontinuation. Therefore, the 1 mg/kg/day dose was considered the NOAEL for the neurobehavioral effect in this study.
8.5 Geriatric Use
Memantine Hydrochloride
The majority of people with Alzheimer’s disease are 65 years and older. In the clinical study of memantine hydrochloride extended-release, the mean age of patients was approximately 77 years; over 91% of patients were 65 years of age and older, 67% were 75 years of age and older, and 14% were 85 years of age and older. The efficacy and safety data presented in the clinical trials section were obtained from these patients. There were no clinically meaningful differences in most adverse events reported by patients ≥ 65 years old and < 65 years old.
Donepezil Hydrochloride
The mean age of patients enrolled in the clinical studies with donepezil was 73 years; 80% of these patients were between 65 and 84 years of age, and 49% of patients 75 years of age and older. The efficacy and safety data presented in the clinical trials section were obtained from these patients. There were no clinically significant differences in most adverse events reported by patients ≥ 65 years old and < 65 years old.
8.6 Renal Impairment
A dosage reduction is recommended in patients with severe renal impairment [see Dosage and Administration (2.3) andClinical Pharmacology (12.3)]. No dosage adjustment is needed in patients with mild or moderate renal impairment.
8.7 Hepatic Impairment
No dosage adjustment is needed in patients with mild or moderate hepatic impairment. Memantine and donepezil hydrochlorides extended-release has not been studied in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Memantine hydrochloride and donepezil hydrochloride are the two active ingredients of memantine and donepezil hydrochlorides extended-release. No specific antidote for memantine hydrochloride overdose is known; however, elimination of memantine can be increased by acidification of the urine. Tertiary anticholinergics such as atropine may be used as an antidote for donepezil hydrochloride overdose. In managing cases of overdose, consider the possibility of multiple drug involvement. In case of overdose, call Poison Control Center at 1-800-222-1222 for the latest recommendation. In general, supportive measures should be utilized, and treatment should be symptomatic.
Memantine Hydrochloride
Signs and symptoms most often accompanying overdosage with other formulations of memantine in clinical trials and from worldwide marketing experience, alone or in combination with other drugs and/or alcohol, include agitation, asthenia, bradycardia, confusion, coma, dizziness, ECG changes, increased blood pressure, lethargy, loss of consciousness, psychosis, restlessness, slowed movement, somnolence, stupor, unsteady gait, visual hallucinations, vertigo, vomiting, and weakness. The largest known ingestion of memantine worldwide was 2 grams in an individual who took memantine in conjunction with unspecified antidiabetic medications. This person experienced coma, diplopia, and agitation, but subsequently recovered.
One patient participating in a memantine hydrochloride extended-release clinical trial unintentionally took 112 mg of memantine hydrochloride extended-release daily for 31 days and experienced an elevated serum uric acid, elevated serum alkaline phosphatase, and low platelet count.
No fatalities have been noted with overdoses of memantine alone. A fatal outcome has very rarely been reported when memantine has been ingested as part of overdosing with multiple drugs; in those instances, the relationship between memantine and a fatal outcome has been unclear.
Donepezil Hydrochloride
Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse, and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Tertiary anticholinergics such as atropine may be used as an antidote for donepezil hydrochloride overdosage. Intravenous atropine sulfate titrated to effect is recommended: an initial dose of 1 to 2 mg IV with subsequent doses based upon clinical response. Atypical responses in blood pressure and heart rate have been reported with other cholinomimetics when co-administered with quaternary anticholinergics such as glycopyrrolate. It is not known whether donepezil hydrochloride and/or its metabolites can be removed by dialysis (hemodialysis, peritoneal dialysis, or hemofiltration).
Dose-related signs of toxicity in animals included reduced spontaneous movement, prone position, staggering gait, lacrimation, clonic convulsions, depressed respiration, salivation, miosis, tremors, fasciculation, and lower body surface temperature.
-
11 DESCRIPTION
Memantine and donepezil hydrochlorides extended-release capsules contain memantine, an orally active NMDA receptor antagonist, as the hydrochloride salt and donepezil, a reversible inhibitor of the enzyme acetylcholinesterase, as the hydrochloride salt.
Memantine Hydrochloride, USP
The chemical name for memantine hydrochloride, USP is 1-amino-3,5-dimethyladamantane hydrochloride with the following structural formula:
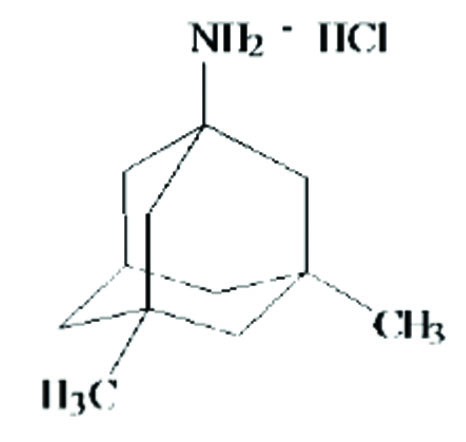
The molecular formula is C12H21N HCl and the molecular weight is 215.76. Memantine hydrochloride, USP occurs as a fine white to off-white powder.
Donepezil Hydrochloride, USP
The chemical name for donepezil hydrochloride, USP is (±)-2-[(1-benzyl- 4-piperidyl) methyl]-5,6-dimethoxy-1-indanone hydrochloride
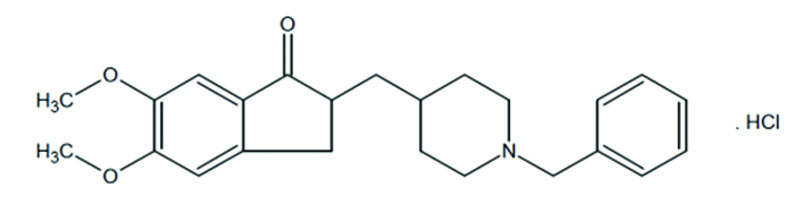
The molecular formula is C24H29NO3HCl and the molecular weight is 415.95. Donepezil hydrochloride, USP is off white to cream coloured powder.
Memantine and donepezil hydrochlorides extended-release capsules contain 7 mg memantine hydrochloride extended-release and 10 mg donepezil hydrochloride (7 mg/10 mg), 14 mg memantine hydrochloride extended-release and 10 mg donepezil hydrochloride (14 mg/10 mg), 21 mg memantine hydrochloride extended-release and 10 mg donepezil hydrochloride (21 mg/10 mg) or 28 mg memantine hydrochloride extended-release and 10 mg donepezil hydrochloride (28 mg/10 mg).
Memantine and donepezil hydrochlorides extended-release capsules (7 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no. 10, FD&C blue no. 1, gelatin, sodium lauryl sulfate and titanium dioxide. The black monogramming ink contains black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, shellac, strong ammonia solution and purified water.
Memantine and donepezil hydrochlorides extended-release capsules (14 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no.10, FD&C blue no. 1, gelatin, sodium lauryl sulfate and titanium dioxide. The white monogramming ink contains butyl alcohol, dehydrated alcohol, isopropyl alcohol, sodium hydroxide, propylene glycol, povidone, shellac and titanium dioxide.
Memantine and donepezil hydrochlorides extended-release capsules (21 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no. 10, FD&C red no. 40, gelatin, sodium lauryl sulfate and titanium dioxide. The black monogramming ink contains black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, shellac, strong ammonia solution and purified water.
Memantine and donepezil hydrochlorides extended-release capsules (28 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no.10, FD&C red no. 40, gelatin, sodium lauryl sulfate and titanium dioxide. The black monogramming ink contains black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, purified water, shellac and strong ammonia solution.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Memantine and donepezil hydrochlorides extended-release capsules contain two approved medications: memantine hydrochloride extended-release and donepezil hydrochloride. Each of those medications is postulated to have a different mechanism in Alzheimer’s disease.
Memantine
Persistent activation of central nervous system NMDA receptors by the excitatory amino acid glutamate has been hypothesized to contribute to the symptomatology of Alzheimer’s disease. Memantine is postulated to exert its therapeutic effect through its action as a low to moderate affinity uncompetitive (open channel) NMDA receptor antagonist which binds preferentially to the NMDA receptor-operated cation channels. There is no evidence that memantine prevents or slows neurodegeneration in patients with Alzheimer’s disease.
Donepezil
Current theories on the pathogenesis of the cognitive signs and symptoms of Alzheimer’s disease attribute some of them to a deficiency of cholinergic neurotransmission. Donepezil is postulated to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine in the central nervous system through reversible inhibition of its hydrolysis by acetylcholinesterase. There is no evidence that donepezil prevents or slows neurodegeneration in patients with Alzheimer’s disease.
12.3 Pharmacokinetics
Memantine and donepezil hydrochlorides extended-release
Memantine and donepezil hydrochlorides extended-release was bioequivalent to co-administration of individual memantine hydrochloride extended-release and donepezil hydrochloride.
Exposure (AUC and Cmax) of memantine and donepezil following memantine and donepezil hydrochlorides extended-release administration in the fed or fasted state was similar. Further, exposure of memantine and donepezil following memantine and donepezil hydrochlorides extended-release administration as intact capsule or capsule contents sprinkled on applesauce was similar in healthy subjects.
Memantine Hydrochloride
Memantine is well absorbed after oral administration and has linear pharmacokinetics over the therapeutic dose range. It is excreted predominantly unchanged in urine and has a terminal elimination half-life of about 60 to 80 hours. In a study comparing 28 mg once-daily memantine hydrochloride extended-release to 10 mg twice-daily memantine hydrochloride, Cmax and AUC0-24 values were 48% and 33% higher for the memantine hydrochloride extended-release dosage regimen, respectively.
Absorption
After multiple dose administration of memantine hydrochloride extended-release, memantine peak concentrations occur around 9 to 12 hours postdose. There is no difference in the absorption of memantine hydrochloride extended-release when the capsule is taken intact or when the contents are sprinkled on applesauce.
After single-dose administration, there is no difference in memantine exposure, based on Cmax or AUC, for memantine hydrochloride extended-release when the drug product is administered with food or on an empty stomach. However, peak plasma concentrations are achieved about 18 hours after administration with food versus approximately 25 hours after administration on an empty stomach.
Distribution
The mean volume of distribution of memantine is 9 to 11 L/kg and the plasma protein binding is low (45%).
Metabolism
Memantine undergoes partial hepatic metabolism. The hepatic microsomal CYP450 enzyme system does not play a significant role in the metabolism of memantine.
Elimination
Memantine is excreted predominantly in the urine, unchanged, and has a terminal elimination half-life of about 60 to 80 hours. About 48% of administered drug is excreted unchanged in urine; the remainder is converted primarily to three polar metabolites which possess minimal NMDA receptor antagonistic activity: the N-glucuronide conjugate, 6-hydroxy memantine, and 1-nitroso-deaminated memantine. A total of 74% of the administered dose is excreted as the sum of the parent drug and the N-glucuronide conjugate. Renal clearance involves active tubular secretion moderated by pH dependent tubular reabsorption.
Pharmacokinetics in Special Populations
Renal Impairment
Memantine pharmacokinetics were evaluated following single oral administration of 20 mg memantine hydrochloride in 8 subjects with mild renal impairment (creatinine clearance, CLcr, > 50 to 80 mL/min), 8 subjects with moderate renal impairment (CLcr 30 to 49 mL/min), 7 subjects with severe renal impairment (CLcr 5 to 29 mL/min) and 8 healthy subjects (CLcr > 80 mL/min) matched as closely as possible by age, weight and gender to the subjects with renal impairment. Mean AUC0-∞ increased by 4%, 60%, and 115% in subjects with mild, moderate, and severe renal impairment, respectively, compared to healthy subjects. The terminal elimination half-life increased by 18%, 41%, and 95% in subjects with mild, moderate, and severe renal impairment, respectively, compared to healthy subjects [see Dosage and Administration (2.3) and Use in Specific Populations (8.6)].
Hepatic Impairment
Memantine pharmacokinetics were evaluated following the administration of single oral doses of 20 mg in 8 subjects with moderate hepatic impairment (Child-Pugh Class B, score 7 to 9) and 8 subjects who were age-, gender-, and weight-matched to the hepatically-impaired subjects. There was no change in memantine exposure (based on Cmax and AUC) in subjects with moderate hepatic impairment as compared with healthy subjects. However, terminal elimination half-life increased by about 16% in subjects with moderate hepatic impairment as compared with healthy subjects. The pharmacokinetics of memantine has not been evaluated in patients with severe hepatic impairment.
Gender
Following multiple dose administration of memantine hydrochloride 20 mg daily, females had about 45% higher exposure than males, but there was no difference in exposure when body weight was taken into account.
Elderly
The pharmacokinetics of memantine in young and elderly subjects are similar.
Drug-Drug Interactions
Use with Cholinesterase Inhibitors
Co-administration of memantine with the AChE inhibitor donepezil hydrochloride did not affect the pharmacokinetics of either compound. Furthermore, memantine did not affect AChE inhibition by donepezil. In a 24-week controlled clinical study in patients with moderate to severe Alzheimer’s disease, the adverse event profile observed with a combination of memantine immediate-release and donepezil was similar to that of donepezil alone.
Effect of Memantine on the Metabolism of Other Drugs
In vitro studies conducted with marker substrates of CYP450 enzymes (CYP1A2, -2A6, -2C9, -2D6, -2E1, -3A4) showed minimal inhibition of these enzymes by memantine. In addition, in vitro studies indicate that at concentrations exceeding those associated with efficacy, memantine does not induce the cytochrome P450 isozymes CYP1A2, -2C9, -2E1 and -3A4/5. No pharmacokinetic interactions with drugs metabolized by these enzymes are expected.
Pharmacokinetic studies evaluated the potential of memantine for interaction with warfarin and bupropion. Memantine did not affect the pharmacokinetics of the CYP2B6 substrate bupropion or its metabolite hydroxybupropion. Furthermore, memantine did not affect the pharmacokinetics or pharmacodynamics of warfarin as assessed by prothrombin INR.
Effect of Other Drugs on Memantine
Memantine is predominantly renally eliminated, and drugs that are substrates and/or inhibitors of the CYP450 system are not expected to alter the pharmacokinetics of memantine. A single dose of bupropion did not affect the pharmacokinetics of memantine at steady state.
Drugs Eliminated via Renal Mechanisms
Because memantine is eliminated in part by tubular secretion, co-administration of drugs that use the same renal cationic system, including hydrochlorothiazide (HCTZ), triamterene (TA), metformin, cimetidine, ranitidine, quinidine, and nicotine, could potentially result in altered plasma levels of both agents. However, co-administration of memantine hydrochloride and HCTZ/TA did not affect the bioavailability of either memantine or TA, and the bioavailability of HCTZ decreased by 20%. In addition, co-administration of memantine hydrochloride with the antihyperglycemic drug Glucovance® (glyburide and metformin hydrochloride) did not affect the pharmacokinetics of memantine, metformin, and glyburide. Furthermore, memantine did not modify the serum glucose lowering effect of Glucovance®, indicating the absence of a pharmacodynamic interaction.
Drugs Highly Bound to Plasma Proteins
Because the plasma protein binding of memantine is low (45%), an interaction with drugs that are highly bound to plasma proteins, such as warfarin and digoxin, is unlikely.
Donepezil Hydrochloride
Pharmacokinetics of donepezil are linear over a dose range of 1 to 10 mg given once daily. The rate and extent of absorption of donepezil hydrochloride tablets are not influenced by food.
Donepezil is absorbed with a relative oral bioavailability of 100% and reaches peak plasma concentrations in 3 to 4 hours.
The elimination half-life of donepezil is about 70 hours, and the mean apparent plasma clearance (Cl/F) is 0.13 to 0.19 L/hr/kg. Following multiple dose administration, donepezil accumulates in plasma by 4 to 7 fold, and steady state is reached within 15 days. The steady-state volume of distribution is 12 to 16 L/kg. Donepezil is approximately 96% bound to human plasma proteins, mainly to albumins (about 75%) and alpha-1-acid glycoprotein (about 21%) over the concentration range of 2 to 1,000 ng/mL.
Donepezil is both excreted in the urine intact and extensively metabolized to four major metabolites, two of which are known to be active, and a number of minor metabolites, not all of which have been identified. Donepezil is metabolized by CYP 450 isoenzymes 2D6 and 3A4 and undergoes glucuronidation. Following administration of 14C-labeled donepezil, plasma radioactivity, expressed as a percent of the administered dose, was present primarily as intact donepezil (53%) and as 6-O-desmethyl donepezil (11%), which has been reported to inhibit AChE to the same extent as donepezil in vitro and was found in plasma at concentrations equal to about 20% of donepezil. Approximately 57% and 15% of the total radioactivity was recovered in urine and feces, respectively, over a period of 10 days, while 28% remained unrecovered, with about 17% of the donepezil dose recovered in the urine as unchanged drug. Examination of the effect of CYP2D6 genotype in Alzheimer’s patients showed differences in clearance values among CYP2D6 genotype subgroups. When compared to the extensive metabolizers, poor metabolizers had a 31.5% slower clearance and ultra-rapid metabolizers had a 24% faster clearance. These results suggest CYP2D6 has a minor role in the metabolism of donepezil.
Renal Impairment
In a study of 11 patients with moderate to severe renal impairment (CLcr < 18 mL/min/ 1.73 m2) the clearance of donepezil hydrochloride did not differ from 11 age- and sex-matched healthy subjects.
Hepatic Disease
In a study of 10 patients with stable alcoholic cirrhosis, the clearance of donepezil hydrochloride was decreased by 20% relative to 10 healthy age- and sex-matched subjects.
Age
No formal pharmacokinetic study was conducted to examine age-related differences in the pharmacokinetics of donepezil hydrochloride. Population pharmacokinetic analysis suggested that the clearance of donepezil in patients decreases with increasing age. When compared with 65-year old, subjects, 90-year old subjects have a 17% decrease in clearance, while 40-year old subjects have a 33% increase in clearance. The effect of age on donepezil clearance may not be clinically significant.
Gender and Race
No specific pharmacokinetic study was conducted to investigate the effects of gender and race on the disposition of donepezil hydrochloride. However, retrospective pharmacokinetic analysis and population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer’s disease indicate that gender and race (Japanese and Caucasians) did not affect the clearance of donepezil hydrochloride to an important degree.
Body weight
There was a relationship noted between body weight and clearance. Over the range of body weight from 50 kg to 110 kg, clearance increased from 7.77 L/h to 14.04 L/h, with a value of 10 L/h for 70 kg individuals.
Drug-Drug Interactions
Effect of Donepezil hydrochloride on the Metabolism of Other Drugs
No in vivo clinical trials have investigated the effect of donepezil hydrochloride on the clearance of drugs metabolized by CYP3A4 (e.g., cisapride, terfenadine) or by CYP2D6 (e.g., imipramine). However, in vitro studies show a low rate of binding to these enzymes (mean Ki about 50 to 130 μM), that, given the therapeutic plasma concentrations of donepezil (164 nM), indicates little likelihood of interference. Based on in vitro studies, donepezil shows little or no evidence of direct inhibition of CYP2B6, CYP2C8, and CYP2C19 at clinically relevant concentrations.
Whether donepezil hydrochloride has any potential for enzyme induction is not known. Formal pharmacokinetic studies evaluated the potential of donepezil hydrochloride for interaction with theophylline, cimetidine, warfarin, digoxin, and ketoconazole. No effects of donepezil hydrochloride on the pharmacokinetics of these drugs were observed.
Effect of Other Drugs on the Metabolism of Donepezil hydrochloride
A small effect of CYP2D6 inhibitors was identified in a population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer’s disease. Donepezil clearance was reduced by approximately 17% in patients taking 10 or 23 mg in combination with a known CYP2D6 inhibitor. This result is consistent with the conclusion that CYP2D6 is a minor metabolic pathway of donepezil.
Formal pharmacokinetic studies demonstrated that the metabolism of donepezil hydrochloride is not significantly affected by concurrent administration of digoxin or cimetidine.
An in vitro study showed that donepezil was not a substrate of P-glycoprotein.
Drugs Highly Bound to Plasma Proteins
Drug displacement studies have been performed in vitro between this highly bound drug (96%) and other drugs such as furosemide, digoxin, and warfarin. Donepezil hydrochloride at concentrations of 0.3 to 10 micrograms/mL did not affect the binding of furosemide (5 micrograms/mL), digoxin (2 ng/mL), and warfarin (3 micrograms/mL) to human albumin. Similarly, the binding of donepezil hydrochloride to human albumin was not affected by furosemide, digoxin, and warfarin.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Memantine
There was no evidence of carcinogenicity in a 113-week oral study in mice at doses up to 40 mg/kg/day (7 times the dose of memantine at the maximum recommended human dose [MRHD] of memantine and donepezil hydrochlorides extended-release [28 mg/10 mg] on a mg/m2 basis). There was also no evidence of carcinogenicity in rats orally dosed at up to 40 mg/kg/day for 71 weeks followed by 20 mg/kg/day (14 and 7 times the MRHD on a mg/m2 basis, respectively) through 128 weeks.
Memantine produced no evidence of genotoxic potential when evaluated in the in vitro S. typhimurium or E. coli reverse mutation assay, an in vitro chromosomal aberration test in human lymphocytes, an in vivo cytogenetics assay for chromosome damage in rats, and the in vivo mouse micronucleus assay. The results were equivocal in an in vitro gene mutation assay using Chinese hamster V79 cells.
No impairment of fertility or reproductive performance was seen in rats administered up to 18 mg/kg/day (6 times the dose of memantine at the MRHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis) orally from 14 days prior to mating through gestation and lactation in females, or for 60 days prior to mating in males.
Donepezil
No evidence of carcinogenic potential was obtained in an 88-week carcinogenicity study of donepezil conducted in mice at oral doses up to 180 mg/kg/day (approximately 90 times the dose of donepezil at the MRHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis), or in a 104-week carcinogenicity study in rats at oral doses up to 30 mg/kg/day (approximately 30 times the dose of donepezil at the MRHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis).
Donepezil was negative in a battery of genotoxicity assays (in vitro bacterial reverse mutation, in vitro mouse lymphoma tk, in vitro chromosomal aberration, and in vivo mouse micronucleus).
Donepezil had no effect on fertility in rats at oral doses up to 10 mg/kg/day (approximately 10 times the dose of donepezil at the MRHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis) when administered to males and females prior to and during mating and continuing in females through implantation.
13.2 Animal Toxicology and/or Pharmacology
Memantine induced neuronal lesions (vacuolation and necrosis) in the multipolar and pyramidal cells in cortical layers III and IV of the posterior cingulate and retrosplenial neocortices in rats, similar to those which are known to occur in rodents administered other NMDA receptor antagonists. Lesions were seen after a single dose of memantine. In a study in which rats were given daily oral doses of memantine for 14 days, the no-effect dose for neuronal necrosis was 4 times the dose of memantine at the MRHD of memantine and donepezil hydrochlorides extended-release on a mg/m2 basis.
In acute and repeat-dose neurotoxicity studies in female rats, oral administration of memantine and donepezil in combination resulted in increased incidence, severity, and distribution of neurodegeneration compared with memantine alone. The no-effect levels of the combination were associated with clinically relevant plasma memantine and donepezil exposures.
The relevance of these findings to humans is unknown.
-
14 CLINICAL STUDIES
The effectiveness of memantine and donepezil hydrochlorides extended-release as a treatment for patients with moderate to severe Alzheimer’s disease was established by demonstrating the bioequivalence of memantine and donepezil hydrochlorides extended-release with co-administered memantine and donepezil hydrochlorides extended-release [see Clinical Pharmacology (12.3)].
Memantine Hydrochloride
The effectiveness of memantine hydrochloride extended-release as a treatment for patients with moderate to severe Alzheimer’s disease when co-administered with acetylcholinesterase inhibitors, including donepezil hydrochloride, was based on the results of a double-blind, placebo-controlled trial.
24-week Study of Memantine Hydrochloride Extended-Release
This was a randomized, double-blind clinical investigation in 677 outpatients with moderate to severe Alzheimer’s disease (diagnosed by DSM-IV criteria and NINCDS-ADRDA criteria for AD with a Mini Mental State Examination [MMSE] score ≥ 3 and ≤ 14 at Screening and Baseline) receiving acetylcholinesterase inhibitor (AChEI) therapy at a stable dose for 3 months prior to screening. Approximately 68% of the patients received donepezil as the AChEI. The mean age of patients participating in this trial was 76.5 years, with a range of 49 to 97 years. Approximately 72% of patients were female and 94% were Caucasian.
Study Outcome Measures
The effectiveness of memantine hydrochloride extended-release was evaluated in this study using the co-primary efficacy parameters of Severe Impairment Battery (SIB) and the Clinician’s Interview-Based Impression of Change (CIBIC-Plus).
The ability of memantine hydrochloride extended-release to improve cognitive performance was assessed with the Severe Impairment Battery (SIB), a multi-item instrument that has been validated for the evaluation of cognitive function in patients with moderate to severe dementia. The SIB examines selected aspects of cognitive performance, including elements of attention, orientation, language, memory, visuospatial ability, construction, praxis, and social interaction. The SIB scoring range is from 0 to 100, with lower scores indicating greater cognitive impairment.
The ability of memantine hydrochloride extended-release to produce an overall clinical effect was assessed using a Clinician’s Interview Based Impression of Change that required the use of caregiver information, the CIBIC-Plus. The CIBIC-Plus is not a single instrument and is not a standardized instrument like the ADCS-ADL or SIB. Clinical trials for investigational drugs have used a variety of CIBIC formats, each different in terms of depth and structure. As such, results from a CIBIC-Plus reflect clinical experience from the trial or trials in which it was used and cannot be compared directly with the results of CIBIC-Plus evaluations from other clinical trials. The CIBIC-Plus used in this trial was a structured instrument based on a comprehensive evaluation at baseline and subsequent time points of four domains: general (overall clinical status), functional
(including activities of daily living), cognitive, and behavioral. It represents the assessment of a skilled clinician using validated scales based on his/her observation during an interview with the patient, in combination with information supplied by a caregiver familiar with the behavior of the patient over the interval rated. The CIBIC-Plus is scored as a seven point categorical rating, ranging from a score of 1, indicating “marked improvement” to a score of 4, indicating “no change” to a score of 7, indicating “marked worsening.” The CIBIC-Plus has not been systematically compared directly to assessments not using information from caregivers (CIBIC) or other global methods.
Study Results
In this study, 677 patients were randomized to one of the following 2 treatments: memantine hydrochloride extended-release 28 mg/day or placebo, while still receiving an AChEI (either donepezil, galantamine, or rivastigmine).
Effects on Severe Impairment Battery (SIB)
Figure 1 shows the time course for the change from baseline in SIB score for the two treatment groups completing the 24 weeks of the study. At 24 weeks of treatment, the mean difference in the SIB change scores for the memantine hydrochloride extended-release 28 mg/AChEI-treated (combination therapy) patients compared to the patients on placebo/AChEI (monotherapy) was 2.6 units. Using an LOCF analysis, memantine hydrochloride extended-release 28 mg/AChEI treatment was statistically significantly superior to placebo/AChEI.
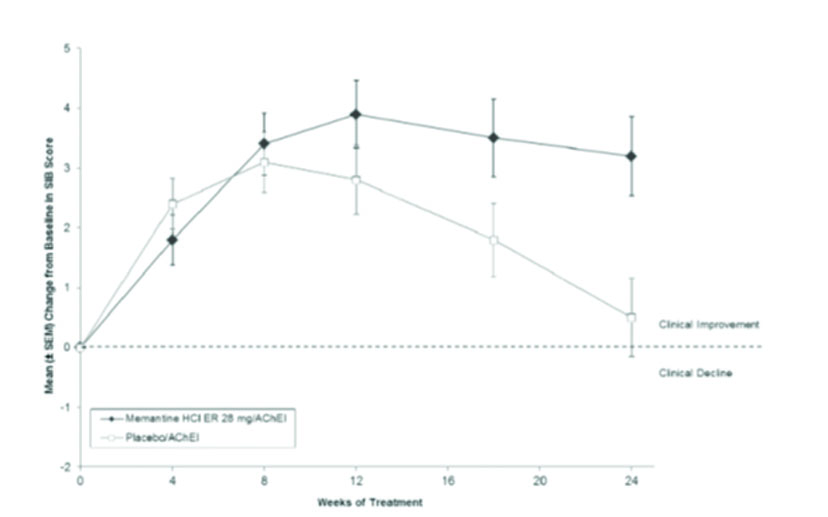
Figure 1: Time course of the change from baseline in SIB score for patients completing 24 weeks of treatment.
Figure 2 shows the cumulative percentages of patients from each treatment group who had attained at least the measure of improvement in SIB score shown on the X-axis. The curves show that both patients assigned to memantine hydrochloride extended-release 28 mg/AChEI and placebo/AChEI have a wide range of responses, but that the memantine hydrochloride extended-release 28 mg/AChEI group is more likely to show an improvement or a smaller decline.
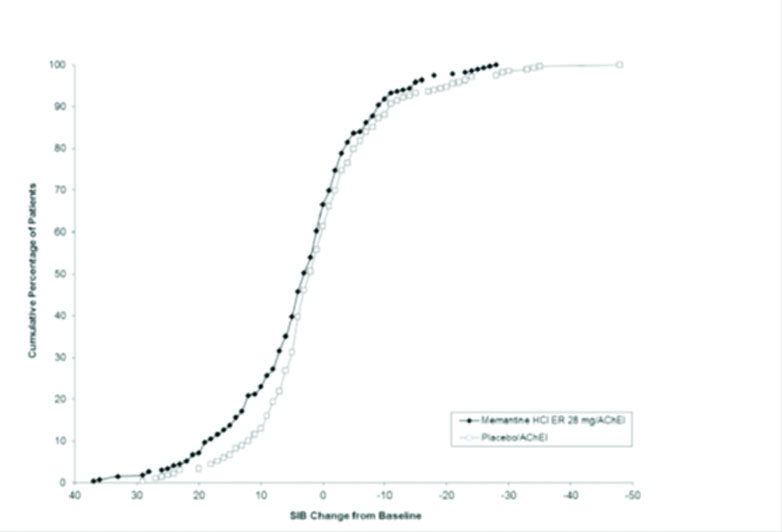
Figure 2: Cumulative percentage of patients completing 24 weeks of double-blind treatment with specified changes from baseline in SIB scores.
Effects on Severe Impairment Battery (SIB) in the Subset of Patients on Concomitant Donepezil Therapy
Approximately 68% of the patients randomized to receive either memantine hydrochloride extended-release 28 mg or placebo were taking donepezil at Baseline and throughout the study. At 24 weeks of treatment, in patients on concomitant donepezil treatment, the mean difference in the SIB change scores for the memantine hydrochloride extended-release 28 mg-treated patients compared to patients on placebo (2.7 units) was similar to that observed for the entire study population (2.6 units).
Effects of Clinician's Interview-Based Impression of Change Plus Caregiver Input (CIBIC-Plus)
Figure 3 shows the time course for the CIBIC-Plus score for patients in the two treatment groups completing the 24 weeks of the study. At 24 weeks of treatment, the mean difference in the CIBIC-Plus scores for the memantine hydrochloride extended-release 28 mg/AChEI-treated patients compared to the patients on placebo/AChEI was 0.3 units. Using an LOCF analysis, memantine hydrochloride extended-release 28 mg/AChEI treatment was statistically significantly superior to placebo/AChEI.
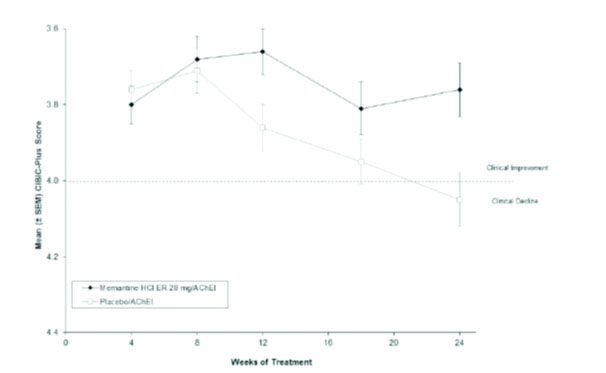
Figure 3: Time course of the CIBIC-Plus score for patients completing 24 weeks of treatment.
Figure 4 is a histogram of the percentage distribution of CIBIC-Plus scores attained by patients assigned to each of the treatment groups who completed 24 weeks of treatment.
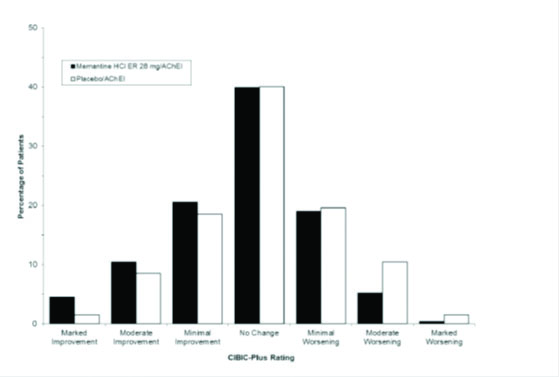
Figure 4: Distribution of CIBIC-Plus ratings at week 24.
Effects on CIBIC-Plus in the Subset of Patients on Concomitant Donepezil Therapy
Approximately 68% of the patients randomized to receive either memantine hydrochloride extended-release 28 mg or placebo were taking donepezil at baseline and throughout the study. At 24 weeks of treatment, in patients on concomitant donepezil, the mean difference in the CIBIC-Plus scores for the memantine hydrochloride extended-release 28 mg-treated patients compared to patients on placebo (0.3 units) was similar to that observed for the entire study population (0.3 units).
Donepezil Hydrochloride
The effectiveness of donepezil hydrochloride as a treatment for patients with severe Alzheimer’s disease was based on the results of two double-blind, placebo-controlled trials.
6-Month Study of Donepezil Hydrochloride
This was a randomized, double-blind, placebo-controlled clinical study conducted in Sweden in patients with probable or possible Alzheimer's disease diagnosed by NINCDS-ADRDA and DSM-IV criteria, MMSE: range of 1 to 10. Two hundred and forty eight (248) patients with severe Alzheimer's disease were randomized to donepezil hydrochloride or placebo. For patients randomized to donepezil hydrochloride, treatment was initiated at 5 mg once daily for 28 days and then increased to 10 mg once daily. At the end of the 6 month treatment period, 90.5% of the donepezil hydrochloride treated patients were receiving the 10 mg/day dose. The mean age of patients was 84.9 years, with a range of 59 to 99. Approximately 77% of patients were women, and 23% were men. Almost all patients were Caucasian. Probable AD was diagnosed in the majority of the patients (83.6% of donepezil hydrochloride treated patients and 84.2% of placebo treated patients).
Study Outcome Measures
The effectiveness of treatment with donepezil hydrochloride was evaluated using a dual outcome assessment strategy that evaluated cognitive function using an instrument designed for more impaired patients and overall function through caregiver-rated assessment. This study showed that patients on donepezil hydrochloride experienced significant improvement on both measures compared to placebo.
The ability of donepezil hydrochloride to improve cognitive performance was assessed with the SIB.
Daily function was assessed using the Modified Alzheimer's Disease Cooperative Study Activities of Daily Living Inventory for Severe Alzheimer's Disease (ADCS-ADL-severe). The ADCS- ADL-severe is derived from the Alzheimer's Disease Cooperative Study Activities of Daily Living Inventory, which is a comprehensive battery of ADL questions used to measure the functional capabilities of patients. Each ADL item is rated from the highest level of independent performance to complete loss. The ADCS-ADL-severe is a subset of 19 items, including ratings of the patient's ability to eat, dress, bathe, use the telephone, get around (or travel), and perform other activities of daily living; it has been validated for the assessment of patients with moderate to severe dementia. The ADCS-ADL-severe has a scoring range of 0 to 54, with the lower scores indicating greater functional impairment. The investigator performs the inventory by interviewing a caregiver, in this study a nurse staff member, familiar with the functioning of the patient.
Effects on the SIB
Figure 5 shows the time course for the change from baseline in SIB score for the two treatment groups over the 6 months of the study. At 6 months of treatment, the mean difference in the SIB change scores for donepezil hydrochloride treated patients compared to patients on placebo was
5.9 points. Donepezil hydrochloride treatment was statistically significantly superior to placebo.
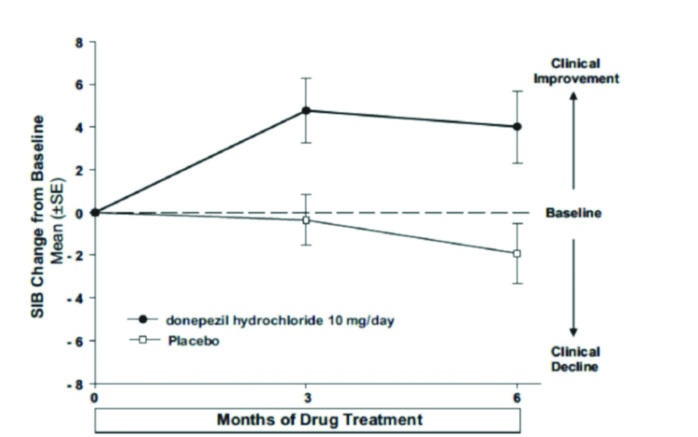
Figure 5. Time Course of the Change from Baseline in SIB Score for Patients Completing 6 months of Treatment.
Figure 6 illustrates the cumulative percentages of patients from each of the two treatment groups who attained the measure of improvement in SIB score shown on the X-axis. While patients assigned both to donepezil hydrochloride and to placebo have a wide range of responses, the curves show that the donepezil hydrochloride group is more likely to show a greater improvement in cognitive performance.
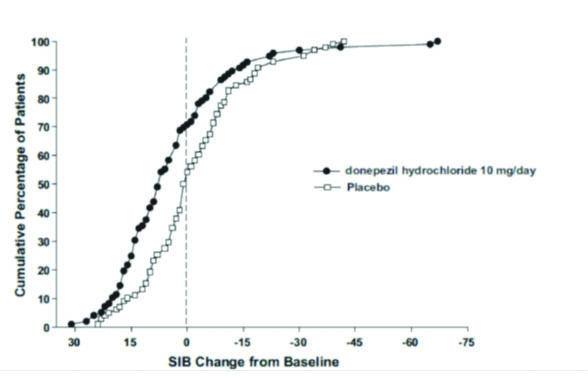
Figure 6. Cumulative Percentage of Patients Completing 6 Months of Double-blind Treatment with Particular Changes from Baseline in SIB Scores.
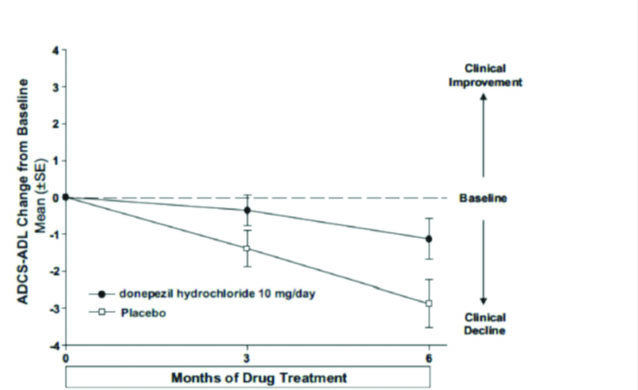
Figure 7. Time Course of the Change from Baseline in ADCS-ADL-Severe Score for Patients Completing 6 Months of Treatment.
Effects on the ADCS-ADL-severe
Figure 7 illustrates the time course for the change from baseline in ADCS-ADL-severe scores for patients in the two treatment groups over the 6 months of the study. After 6 months of treatment, the mean difference in the ADCS-ADL-severe change scores for donepezil hydrochloride treated patients compared to patients on placebo was 1.8 points. Donepezil hydrochloride treatment was statistically significantly superior to placebo.
Figure 8 shows the cumulative percentages of patients from each treatment group with specified changes from baseline ADCS-ADL-severe scores. While both patients assigned to donepezil hydrochloride and placebo have a wide range of responses, the curves demonstrate that the donepezil hydrochloride group is more likely to show a smaller decline or an improvement.
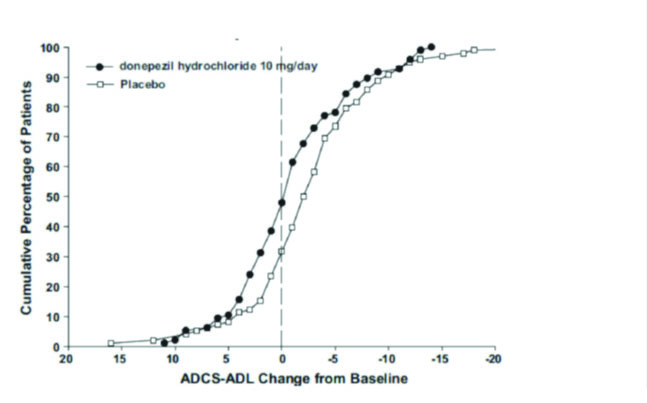
Figure 8. Cumulative Percentage of Patients Completing 6 Months of Double-blind Treatment with Particular Changes from Baseline in ADCS-ADL-Severe Scores.
24-Week Study of Donepezil Hydrochloride
In a randomized, double-blind, placebo-controlled study conducted in Japan, 325 patients with severe Alzheimer's disease received doses of 5 mg/day or 10 mg/day of donepezil hydrochloride, administered once daily, or placebo. Patients randomized to treatment with donepezil hydrochloride were to achieve their assigned doses by titration, beginning at 3 mg/day, and extending over a maximum of 6 weeks. Two hundred and forty eight (248) patients completed the study, with similar proportions of patients completing the study in each treatment group. The primary efficacy measures for this study were the SIB and CIBIC-plus.
At 24 weeks of treatment, statistically significant treatment differences were observed between the 10 mg/day dose of donepezil hydrochloride and placebo on both the SIB and CIBIC-plus. The 5 mg/day dose of donepezil hydrochloride showed a statistically significant superiority to placebo on the SIB, but not on the CIBIC-plus.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Memantine and donepezil hydrochlorides extended-release capsules, 7 mg/10 mg, are supplied as two-piece hard gelatin capsules with green opaque cap and white opaque body filled with white to off-white pellets. Imprinted in black ink AN on the cap and imprinted in black ink 1552 on the body.
They are available as follows:
Bottles of 30: NDC: 69238-1552-3
Bottles of 90: NDC: 69238-1552-9Memantine and donepezil hydrochlorides extended-release capsules, 14 mg/10 mg, are supplied as two-piece hard gelatin capsules with green opaque cap and green opaque body filled with white to off-white pellets. Imprinted in white ink AN on the cap and imprinted in white ink 1248 on the body.
They are available as follows:
Bottle of 30: NDC: 69238-1248-3
Bottle of 90: NDC: 69238-1248-9Memantine and donepezil hydrochlorides extended-release capsules, 21 mg/10 mg, are supplied as two-piece hard gelatin capsules with yellow opaque cap and white opaque body filled with white to off-white pellets. Imprinted in black ink AN on the cap and imprinted in black ink 1553 on the body.
They are available as follows:
Bottles of 30: NDC: 69238-1553-3
Bottles of 90: NDC: 69238-1553-9Memantine and donepezil hydrochlorides extended-release capsules, 28 mg/10 mg, are supplied as two-piece hard gelatin capsules with yellow opaque cap and yellow opaque body filled with white to off-white pellets. Imprinted in black ink AN on the cap and imprinted in black ink 1247 on the body. They are available as follows:
Bottle of 30: NDC: 69238-1247-3
Bottle of 90: NDC: 69238-1247-9 -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Dosing Information
- Instruct patients and caregivers to take memantine and donepezil hydrochlorides extended-release only once daily in the evening, as prescribed [see Dosage and Administration (2.1)].
- If a patient misses a single dose of memantine and donepezil hydrochlorides extended-release, that patient should not double up on the next dose. The next dose should be taken as scheduled [see Dosage and Administration (2.1)].
- Instruct patients and caregivers that memantine and donepezil hydrochlorides extended-release capsules should be swallowed whole. Alternatively, memantine and donepezil hydrochlorides extended-release capsules may be opened and sprinkled on applesauce and the entire contents should be consumed. The capsules should not be divided, chewed, or crushed. Warn patients and caregivers not to use any capsules of memantine and donepezil hydrochlorides extended-release that are damaged or show signs of tampering [see Dosage and Administration (2.2)].
Common Adverse Reactions
Advise patients and caregivers that memantine and donepezil hydrochlorides extended-release may cause headache, diarrhea, dizziness, anorexia, vomiting, nausea, and ecchymosis [see Adverse Reactions (6.1)].
Distributed by:
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807 -
PATIENT INFORMATION
Memantine (meh-MAN-teen) and Donepezil (doe-NEP-e-zil) Hydrochlorides Extended-release Capsules
What are memantine and donepezil hydrochlorides extended-release capsules?
- Memantine and donepezil hydrochlorides extended-release capsules is a prescription medicine used to treat moderate to severe dementia in people with Alzheimer’s disease. Memantine and donepezil hydrochlorides extended-release capsules contain 2 medicines, memantine HCl, an NMDA receptor antagonist, and donepezil HCl, an acetylcholinesterase inhibitor. Memantine and donepezil hydrochlorides extended-release capsules are for people who are taking donepezil HCl 10 mg.
- It is not known if memantine and donepezil hydrochlorides extended-release capsules are safe and effective in children.
Do not take memantine and donepezil hydrochlorides extended-release capsules if you have an allergy to memantine HCl, donepezil HCl, medicines that contains piperidines, or any of the ingredients in memantine and donepezil hydrochlorides extended-release capsules. See the end of this leaflet for a complete list of ingredients in memantine and donepezil hydrochlorides extended-release capsules.
Before taking memantine and donepezil hydrochlorides extended-release capsules, tell your doctor about all of your medical conditions, including if you:
- have heart problems including an irregular, slow, or fast heartbeat.
- have asthma or lung problems.
- have seizures.
- have stomach ulcers.
- have bladder or kidney problems.
- have liver problems.
- have surgical, dental, or other medical procedures scheduled and anesthesia may be used.
- are pregnant or plan to become pregnant. It is not known if memantine and donepezil hydrochlorides extended-release capsules can harm an unborn baby.
- are breast-feeding or plan to breastfeed. It is not known if memantine hydrochloride and donepezil hydrochloride passes into breast milk. Talk to your doctor about the best way to feed your baby if you take memantine and donepezil hydrochlorides extended-release capsules.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I take memantine and donepezil hydrochlorides extended-release capsules?
- Take memantine and donepezil hydrochlorides extended-release capsules exactly as your doctor tells you to take it.
- Do not change your dose or stop taking memantine and donepezil hydrochlorides extended-release capsules without talking to your doctor.
- Take memantine and donepezil hydrochlorides extended-release capsules by mouth 1 time each evening before going to bed.
- Take memantine and donepezil hydrochlorides extended-release capsules with or without food.
- Memantine and donepezil hydrochlorides extended-release capsules may be opened and sprinkled on applesauce before swallowing. Sprinkle all of the medicine in the capsule on the applesauce. Do not divide the dose.
- If you do not open and sprinkle memantine and donepezil hydrochlorides extended-release capsules on applesauce, the memantine and donepezil hydrochlorides extended-release capsules must be swallowed whole. Do not divide, chew, or crush memantine and donepezil hydrochlorides extended-release capsules.
- If you miss a dose take memantine and donepezil hydrochlorides extended-release capsules at your next scheduled dose. Do not take 2 doses of memantine and donepezil hydrochlorides extended-release capsules at the same time.
- Do not use any memantine and donepezil hydrochlorides extended-release capsules that are damaged or show signs of tampering.
- If you take too much memantine and donepezil hydrochlorides extended-release capsules, call Poison Control Center at 1-800-222-1222, or go to the nearest hospital emergency room right away.
What are the possible side effects of memantine and donepezil hydrochlorides extended-release capsules?
Memantine and donepezil hydrochlorides extended-release capsules may cause serious side effects, including:
- muscle problems if you need anesthesia
- slow heartbeat and fainting. This happens more often in people with heart problems. Call the doctor right away if the patient faints while taking memantine and donepezil hydrochlorides extended-release capsules.
- more stomach acid. This raises the chance of ulcers and bleeding especially when taking memantine and donepezil hydrochlorides extended-release capsules. The risk is higher for patients who had ulcers, or take aspirin or other NSAIDs.
- nausea and vomiting
- difficulty passing urine
- seizures
- worsening of lung problems in people with asthma or other lung disease.
The most common side effects of memantine HCl include:
- headache
- diarrhea
- dizziness
The most common side effects of donepezil HCl include:
- diarrhea
- not wanting to eat (anorexia)
- bruising
These are not all the possible side effects of memantine and donepezil hydrochlorides extended-release capsules. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store memantine and donepezil hydrochlorides extended-release capsules?
Store memantine and donepezil hydrochlorides extended-release capsules at room temperature, about 68°F to 77°F (20°C to 25°C). Capsules should be stored in the original prescription container or other light resistant container.
General information about the safe and effective use of memantine and donepezil hydrochlorides extended-release capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use memantine and donepezil hydrochlorides extended-release capsules for a condition for which it was not prescribed. Do not give memantine and donepezil hydrochlorides extended-release capsules to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or doctor for information about memantine and donepezil hydrochlorides extended-release capsules that is written for health professionals.
What are the ingredients in memantine and donepezil hydrochlorides extended-release capsules?
Active ingredients: memantine hydrochloride, USP and donepezil hydrochloride, USP
Inactive ingredients:
Memantine and donepezil hydrochlorides extended-release capsules (7 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no.10, FD&C blue no. 1, gelatin, sodium lauryl sulfate and titanium dioxide. The black monogramming ink contains black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, shellac, strong ammonia solution and purified water.
Memantine and donepezil hydrochlorides extended-release capsules (14 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no.10, FD&C blue no. 1, gelatin, sodium lauryl sulfate and titanium dioxide. The white monogramming ink contains butyl alcohol, dehydrated alcohol, isopropyl alcohol, sodium hydroxide, propylene glycol, povidone, shellac and titanium dioxide.
Memantine and donepezil hydrochlorides extended-release capsules (21 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no.10, FD&C red no. 40, gelatin, sodium lauryl sulfate and titanium dioxide. The black monogramming ink contains black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, shellac, strong ammonia solution and purified water.
Memantine and donepezil hydrochlorides extended-release capsules (28 mg/10 mg) contain the following inactive ingredients: colloidal silicon dioxide, ethyl cellulose, hydroxypropyl cellulose, hypromellose, sugar spheres (corn starch, sucrose), talc and triethyl citrate. The hard gelatin capsules contain D&C yellow no.10, FD&C red no. 40, gelatin, sodium lauryl sulfate and titanium dioxide. The black monogramming ink contains black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, purified water, shellac and strong ammonia solution.
This Patient Information has been approved by the U.S. Food and Drug Administration.
*All trademarks are the property of their respective owners.
Distributed by:
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807Rev. 04-2017-01
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MEMANTINE AND DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE
memantine and donepezil capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69238-1248 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MEMANTINE HYDROCHLORIDE (UNII: JY0WD0UA60) (MEMANTINE - UNII:W8O17SJF3T) MEMANTINE HYDROCHLORIDE 14 mg DONEPEZIL HYDROCHLORIDE (UNII: 3O2T2PJ89D) (DONEPEZIL - UNII:8SSC91326P) DONEPEZIL HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) HYDROXYPROPYL CELLULOSE, LOW SUBSTITUTED (UNII: 2165RE0K14) HYPROMELLOSES (UNII: 3NXW29V3WO) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) SODIUM HYDROXIDE (UNII: 55X04QC32I) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POVIDONE (UNII: FZ989GH94E) SHELLAC (UNII: 46N107B71O) Product Characteristics Color GREEN (green opaque) Score no score Shape CAPSULE Size 16mm Flavor Imprint Code AN;1248 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69238-1248-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 2 NDC: 69238-1248-9 90 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208328 02/01/2017 MEMANTINE AND DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE
memantine and donepezil capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69238-1247 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MEMANTINE HYDROCHLORIDE (UNII: JY0WD0UA60) (MEMANTINE - UNII:W8O17SJF3T) MEMANTINE HYDROCHLORIDE 28 mg DONEPEZIL HYDROCHLORIDE (UNII: 3O2T2PJ89D) (DONEPEZIL - UNII:8SSC91326P) DONEPEZIL HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) HYDROXYPROPYL CELLULOSE, LOW SUBSTITUTED (UNII: 2165RE0K14) HYPROMELLOSES (UNII: 3NXW29V3WO) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) FERROSOFERRIC OXIDE (UNII: XM0M87F357) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) Product Characteristics Color YELLOW (yellow opaque) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code AN;1247 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69238-1247-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 2 NDC: 69238-1247-9 90 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208328 02/01/2017 MEMANTINE AND DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE
memantine and donepezil capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69238-1552 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MEMANTINE HYDROCHLORIDE (UNII: JY0WD0UA60) (MEMANTINE - UNII:W8O17SJF3T) MEMANTINE HYDROCHLORIDE 7 mg DONEPEZIL HYDROCHLORIDE (UNII: 3O2T2PJ89D) (DONEPEZIL - UNII:8SSC91326P) DONEPEZIL HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) HYDROXYPROPYL CELLULOSE, LOW SUBSTITUTED (UNII: 2165RE0K14) HYPROMELLOSES (UNII: 3NXW29V3WO) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) SODIUM HYDROXIDE (UNII: 55X04QC32I) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) FERROSOFERRIC OXIDE (UNII: XM0M87F357) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) AMMONIA (UNII: 5138Q19F1X) WATER (UNII: 059QF0KO0R) Product Characteristics Color GREEN (green opaque) , WHITE (white to off-white pellets) Score no score Shape CAPSULE Size 16mm Flavor Imprint Code AN;1552 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69238-1552-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 2 NDC: 69238-1552-9 90 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208328 02/01/2017 MEMANTINE AND DONEPEZIL HYDROCHLORIDES EXTENDED-RELEASE
memantine and donepezil capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69238-1553 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MEMANTINE HYDROCHLORIDE (UNII: JY0WD0UA60) (MEMANTINE - UNII:W8O17SJF3T) MEMANTINE HYDROCHLORIDE 21 mg DONEPEZIL HYDROCHLORIDE (UNII: 3O2T2PJ89D) (DONEPEZIL - UNII:8SSC91326P) DONEPEZIL HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) HYDROXYPROPYL CELLULOSE, LOW SUBSTITUTED (UNII: 2165RE0K14) HYPROMELLOSES (UNII: 3NXW29V3WO) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) GELATIN (UNII: 2G86QN327L) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) SODIUM HYDROXIDE (UNII: 55X04QC32I) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) FERROSOFERRIC OXIDE (UNII: XM0M87F357) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) AMMONIA (UNII: 5138Q19F1X) WATER (UNII: 059QF0KO0R) FD&C RED NO. 40 (UNII: WZB9127XOA) Product Characteristics Color YELLOW (yellow opaque) , WHITE (white to off-white pellets) Score no score Shape CAPSULE Size 20mm Flavor Imprint Code AN;1553 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69238-1553-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 2 NDC: 69238-1553-9 90 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208328 02/01/2017 Labeler - Amneal Pharmaceuticals NY LLC (123797875)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.



