Leader Childrens Cold and Cough 577
Childrens Cold and Cough by
Drug Labeling and Warnings
Childrens Cold and Cough by is a Otc medication manufactured, distributed, or labeled by CARDINAL HEALTH 110, LLC. DBA LEADER, Guardian Drug Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CHILDRENS COLD AND COUGH - brompheniramine maleate, dextromethorphan, phenylephrine hcl liquid
CARDINAL HEALTH 110, LLC. DBA LEADER
----------
Leader Childrens Cold and Cough 577
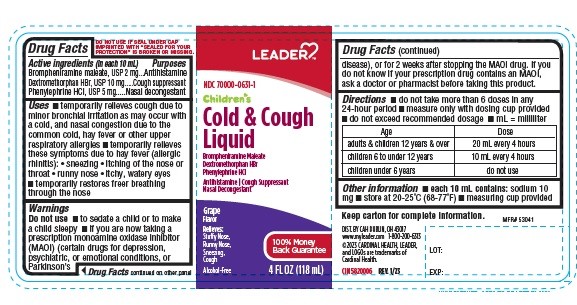
ACTIVE INGRADIENTS (in each 10 mL)
Brompheniramine maleate, USP 2 mg
Dextromethorphan HBr, USP 10 mg
Phenylephrine HCl, USP 5 mg
USES
- temporarily relieves cough due to minor bronchial irritation as may occur with a cold, and nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves these symptoms due to hay fever (allergic rhinitis):
- sneezing
- Itching of the nose or throat
- runny nose
- itchy, watery eyes
- temporarily restores freer breathing through the nose
DO NOT USE
- to sedate a child or to make a child sleepy
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, or psychiatric, for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
ASK A DOCTOR BEFORE USE IF YOU HAVE
- heart disease
- glaucoma
- diabetes
- thyroid disease
- high blood pressure
- difficulty in urination due to an enlarged prostate gland
- cough that occurs with too much phlegm (mucus)
- a breathing problem or persistent or chronic cough that lasts such as occurs with smoking, asthma, chronic bronchitis or emphysema
ASK A DOCTOR OR PHARMACIST BEFORE USE IF YOU ARE
- taking any other oral nasal decongestant or stimulant
- taking sedative or tranquilizers
WHEN USING THIS PRODUCT
- do not take more than directed
- may cause drowsiness
- alcohol, sedatives, and tranquillizers may increase drowsiness
- avoid alcoholic beverages
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
STOP USE AND ASK A DOCTOR IF
- nervousness, dizziness or sleeplessness occur
- symptoms do not improve within 7 days or are accompanied by fever
- cough lasts more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. These could be signs of a serious condition.
KEEP OUT OF REACH OF CHILDREN.
In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
DIRECTIONS
- do not take more than 6 doses in any 24-hour period
- measure only with dosing cup provided
- do not exceed recommended dosage
- mL = milliliter
| Age | Dose |
| adults & children 12 years & over | 20 mL every 4 hours |
| children 6 to under 12 years | 10 mL every 4 hours |
| children under 6 years | do not use |
OTHER INFORMATION
- each 10 mL contains: sodium 10 mg
- store at 20-25ºC (68-77ºF)
- measuring cup provided
INACTIVE INGREDIENTS
anhydrous citric acid, artificial flavor, FD&C blue #1, FD&C Red #40, flavor, glycerin, propylene glycol, propyl gallate, purified water, sodium benzoate, sodium citrate, sorbitol, sucralose
PRINCIPAL DISPLAY PANEL
LEADER
NDC: 70000-0631-1
Children's Cold & Cough Liquid
Brompheniramine Maleate
Dextromethorphan Hydrobromide
Phenylephrine Hydrochloride
Antihistamine
Cough Suppressant
Nasal Decongestant
Grape
Flavor
Relieves:
Stuffy Nose,
Runny Nose,
Sneezing,
Cough
Alcohol-Free
COMPARE TO CHILDREN'S DIMETAPP®
COLD & COUGH
active ingredients*
4 FL OZ (118 mL)

| CHILDRENS COLD AND COUGH
brompheniramine maleate, dextromethorphan, phenylephrine hcl liquid |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - CARDINAL HEALTH 110, LLC. DBA LEADER (063997360) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Guardian Drug Company | 119210276 | MANUFACTURE(70000-0631) | |