CARBAMAZEPINE tablet, extended release CARBAMAZEPIN- carbamazepine tablet, extended release
Carbamazepine by
Drug Labeling and Warnings
Carbamazepine by is a Prescription medication manufactured, distributed, or labeled by Upsher-Smith Laboratories, LLC, CSPC Ouyi Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNINGS
SERIOUS DERMATOLOGIC REACTIONS AND HLA-B*1502 ALLELE
SERIOUS AND SOMETIMES FATAL DERMATOLOGIC REACTIONS, INCLUDING TOXIC EPIDERMAL NECROLYSIS (TEN) AND STEVENS-JOHNSON SYNDROME (SJS), HAVE BEEN REPORTED DURING TREATMENT WITH CARBAMAZEPINE. THESE REACTIONS ARE ESTIMATED TO OCCUR IN 1 TO 6 PER 10,000 NEW USERS IN COUNTRIES WITH MAINLY CAUCASIAN POPULATIONS, BUT THE RISK IN SOME ASIAN COUNTRIES IS ESTIMATED TO BE ABOUT 10 TIMES HIGHER. STUDIES IN PATIENTS OF CHINESE ANCESTRY HAVE FOUND A STRONG ASSOCIATION BETWEEN THE RISK OF DEVELOPING SJS/TEN AND THE PRESENCE OF HLA-B*1502, AN INHERITED ALLELIC VARIANT OF THE HLA-B GENE. HLA-B*1502 IS FOUND ALMOST EXCLUSIVELY IN PATIENTS WITH ANCESTRY ACROSS BROAD AREAS OF ASIA. PATIENTS WITH ANCESTRY IN GENETICALLY AT-RISK POPULATIONS SHOULD BE SCREENED FOR THE PRESENCE OF HLA-B*1502 PRIOR TO INITIATING TREATMENT WITH CARBAMAZEPINE. PATIENTS TESTING POSITIVE FOR THE ALLELE SHOULD NOT BE TREATED WITH CARBAMAZEPINE UNLESS THE BENEFIT CLEARLY OUTWEIGHS THE RISK [SEE WARNINGS AND PRECAUTIONS, LABORATORY TESTS].
APLASTIC ANEMIA AND AGRANULOCYTOSIS
APLASTIC ANEMIA AND AGRANULOCYTOSIS HAVE BEEN REPORTED IN ASSOCIATION WITH THE USE OF CARBAMAZEPINE. DATA FROM A POPULATION-BASED CASE CONTROL STUDY DEMONSTRATE THAT THE RISK OF DEVELOPING THESE REACTIONS IS 5 TIMES TO 8 TIMES GREATER THAN IN THE GENERAL POPULATION. HOWEVER, THE OVERALL RISK OF THESE REACTIONS IN THE UNTREATED GENERAL POPULATION IS LOW, APPROXIMATELY SIX PATIENTS PER ONE MILLION POPULATION PER YEAR FOR AGRANULOCYTOSIS AND TWO PATIENTS PER ONE MILLION POPULATION PER YEAR FOR APLASTIC ANEMIA.
ALTHOUGH REPORTS OF TRANSIENT OR PERSISTENT DECREASED PLATELET OR WHITE BLOOD CELL COUNTS ARE NOT UNCOMMON IN ASSOCIATION WITH THE USE OF CARBAMAZEPINE, DATA ARE NOT AVAILABLE TO ESTIMATE ACCURATELY THEIR INCIDENCE OR OUTCOME. HOWEVER, THE VAST MAJORITY OF THE CASES OF LEUKOPENIA HAVE NOT PROGRESSED TO THE MORE SERIOUS CONDITIONS OF APLASTIC ANEMIA OR AGRANULOCYTOSIS.
BECAUSE OF THE VERY LOW INCIDENCE OF AGRANULOCYTOSIS AND APLASTIC ANEMIA, THE VAST MAJORITY OF MINOR HEMATOLOGIC CHANGES OBSERVED IN MONITORING OF PATIENTS ON CARBAMAZEPINE ARE UNLIKELY TO SIGNAL THE OCCURRENCE OF EITHER ABNORMALITY. NONETHELESS, COMPLETE PRETREATMENT HEMATOLOGICAL TESTING SHOULD BE OBTAINED AS A BASELINE. IF A PATIENT IN THE COURSE OF TREATMENT EXHIBITS LOW OR DECREASED WHITE BLOOD CELL OR PLATELET COUNTS, THE PATIENT SHOULD BE MONITORED CLOSELY. DISCONTINUATION OF THE DRUG SHOULD BE CONSIDERED IF ANY EVIDENCE OF SIGNIFICANT BONE MARROW DEPRESSION DEVELOPS.
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Carbamazepine, USP is an anticonvulsant and specific analgesic for trigeminal neuralgia, available for oral administration as extended-release tablets of 100 mg, 200 mg, and 400 mg. Its chemical name is 5H-dibenz[b,f]azepine-5-carboxamide, and its structural formula is:
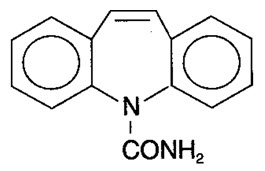
Carbamazepine, USP is a white to off-white powder, practically insoluble in water and soluble in alcohol and in acetone. Its molecular weight is 236.27.
Inactive Ingredients
Carbamazepine extended-release tablets, USP: cellulose acetate, dextrates, ferric oxide red, hydroxyethyl cellulose, hypromellose, magnesium stearate, mannitol, polyethylene glycol, sodium lauryl sulfate, titanium dioxide, ammonium hydroxide, ferrosoferric oxide, propylene glycol and shellac glaze.
FDA approved dissolution specification differs from that of the USP.
-
CLINICAL PHARMACOLOGY
In controlled clinical trials, carbamazepine has been shown to be effective in the treatment of psychomotor and grand mal seizures, as well as trigeminal neuralgia.
Mechanism of Action
Carbamazepine has demonstrated anticonvulsant properties in rats and mice with electrically and chemically induced seizures. It appears to act by reducing polysynaptic responses and blocking the post-tetanic potentiation. Carbamazepine greatly reduces or abolishes pain induced by stimulation of the infraorbital nerve in cats and rats. It depresses thalamic potential and bulbar and polysynaptic reflexes, including the linguomandibular reflex in cats. Carbamazepine is chemically unrelated to other anticonvulsants or other drugs used to control the pain of trigeminal neuralgia. The mechanism of action remains unknown.
The principal metabolite of carbamazepine, carbamazepine-10,11-epoxide, has anticonvulsant activity as demonstrated in several in vivo animal models of seizures. Though clinical activity for the epoxide has been postulated, the significance of its activity with respect to the safety and efficacy of carbamazepine has not been established.
Pharmacokinetics
In clinical studies, carbamazepine suspension, conventional tablets, and extended-release tablets delivered equivalent amounts of drug to the systemic circulation. However, the suspension was absorbed somewhat faster, and the extended-release tablet slightly slower, than the conventional tablet. The bioavailability of the extended-release tablet was 89% compared to suspension. Following a twice a day dosage regimen, the suspension provides higher peak levels and lower trough levels than those obtained from the conventional tablet for the same dosage regimen. On the other hand, following a three times a day dosage regimen, carbamazepine suspension affords steady-state plasma levels comparable to carbamazepine tablets given twice a day when administered at the same total mg daily dose. Following a twice a day dosage regimen, carbamazepine extended-release tablets afford steady-state plasma levels comparable to conventional carbamazepine tablets given four times a day, when administered at the same total mg daily dose. Carbamazepine in blood is 76% bound to plasma proteins. Plasma levels of carbamazepine are variable and may range from 0.5 mcg/mL to 25 mcg/mL, with no apparent relationship to the daily intake of the drug. Usual adult therapeutic levels are between 4 mcg/mL and 12 mcg/mL. In polytherapy, the concentration of carbamazepine and concomitant drugs may be increased or decreased during therapy, and drug effects may be altered [see PRECAUTIONS, Drug Interactions]. Following chronic oral administration of suspension, plasma levels peak at approximately 1.5 hours compared to 4 hours to 5 hours after administration of conventional carbamazepine tablets, and 3 hours to 12 hours after administration of carbamazepine extended-release tablets. The CSF/serum ratio is 0.22, similar to the 24% unbound carbamazepine in serum. Because carbamazepine induces its own metabolism, the half-life is also variable. Autoinduction is completed after 3 weeks to 5 weeks of a fixed dosing regimen. Initial half-life values range from 25 hours to 65 hours, decreasing to 12 hours to 17 hours on repeated doses. Carbamazepine is metabolized in the liver. Cytochrome P450 3A4 was identified as the major isoform responsible for the formation of carbamazepine-10,11-epoxide from carbamazepine. Human microsomal epoxide hydrolase has been identified as the enzyme responsible for the formation of the 10,11-transdiol derivative from carbamazepine-10,11 epoxide. After oral administration of 14C-carbamazepine, 72% of the administered radioactivity was found in the urine and 28% in the feces. This urinary radioactivity was composed largely of hydroxylated and conjugated metabolites, with only 3% of unchanged carbamazepine.
The pharmacokinetic parameters of carbamazepine disposition are similar in children and in adults. However, there is a poor correlation between plasma concentrations of carbamazepine and carbamazepine dose in children. Carbamazepine is more rapidly metabolized to carbamazepine-10,11-epoxide (a metabolite shown to be equipotent to carbamazepine as an anticonvulsant in animal screens) in the younger age groups than in adults. In children below the age of 15, there is an inverse relationship between CBZ-E/CBZ ratio and increasing age (in one report from 0.44 in children below the age of 1 year to 0.18 in children between 10 years to 15 years of age).
The effects of race and gender on carbamazepine pharmacokinetics have not been systematically evaluated.
-
INDICATIONS AND USAGE
Epilepsy
Carbamazepine is indicated for use as an anticonvulsant drug. Evidence supporting efficacy of carbamazepine as an anticonvulsant was derived from active drug-controlled studies that enrolled patients with the following seizure types:
- Partial seizures with complex symptomatology (psychomotor, temporal lobe). Patients with these seizures appear to show greater improvement than those with other types.
- Generalized tonic-clonic seizures (grand mal).
- Mixed seizure patterns which include the above, or other partial or generalized seizures. Absence seizures (petit mal) do not appear to be controlled by carbamazepine [see PRECAUTIONS, General].
-
CONTRAINDICATIONS
Carbamazepine should not be used in patients with a history of previous bone marrow depression, hypersensitivity to the drug, or known sensitivity to any of the tricyclic compounds, such as amitriptyline, desipramine, imipramine, protriptyline, nortriptyline, etc. Likewise, on theoretical grounds its use with monoamine oxidase (MAO) inhibitors is not recommended. Before administration of carbamazepine, MAO inhibitors should be discontinued for a minimum of 14 days, or longer if the clinical situation permits.
Coadministration of carbamazepine and nefazodone may result in insufficient plasma concentrations of nefazodone and its active metabolite to achieve a therapeutic effect. Coadministration of carbamazepine with nefazodone is contraindicated.
-
WARNINGS
Serious Dermatologic Reactions
Serious and sometimes fatal dermatologic reactions, including toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome (SJS), have been reported with carbamazepine treatment. The risk of these events is estimated to be about 1 to 6 per 10,000 new users in countries with mainly Caucasian populations. However, the risk in some Asian countries is estimated to be about 10 times higher. Carbamazepine should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered.
SJS/TEN and HLA-B*1502 Allele
Retrospective case-control studies have found that in patients of Chinese ancestry there is a strong association between the risk of developing SJS/TEN with carbamazepine treatment and the presence of an inherited variant of the HLA-B gene, HLA-B*1502. The occurrence of higher rates of these reactions in countries with higher frequencies of this allele suggests that the risk may be increased in allele-positive individuals of any ethnicity.
Across Asian populations, notable variation exists in the prevalence of HLA-B*1502. Greater than 15% of the population is reported positive in Hong Kong, Thailand, Malaysia, and parts of the Philippines, compared to about 10% in Taiwan and 4% in North China. South Asians, including Indians, appear to have intermediate prevalence of HLA-B*1502, averaging 2% to 4%, but higher in some groups. HLA-B*1502 is present in less than 1% of the population in Japan and Korea.
HLA-B*1502 is largely absent in individuals not of Asian origin (e.g., Caucasians, African-Americans, Hispanics, and Native Americans).
Prior to initiating carbamazepine therapy, testing for HLA-B*1502 should be performed in patients with ancestry in populations in which HLA-B*1502 may be present. In deciding which patients to screen, the rates provided above for the prevalence of HLA-B*1502 may offer a rough guide, keeping in mind the limitations of these figures due to wide variability in rates even within ethnic groups, the difficulty in ascertaining ethnic ancestry, and the likelihood of mixed ancestry. Carbamazepine should not be used in patients positive for HLA-B*1502 unless the benefits clearly outweigh the risks. Tested patients who are found to be negative for the allele are thought to have a low risk of SJS/TEN [see BOXED WARNING and PRECAUTIONS, Laboratory Tests].
Over 90% of carbamazepine treated patients who will experience SJS/TEN have this reaction within the first few months of treatment. This information may be taken into consideration in determining the need for screening of genetically at-risk patients currently on carbamazepine.
The HLA-B*1502 allele has not been found to predict risk of less severe adverse cutaneous reactions from carbamazepine, such as maculopapular eruption (MPE) or to predict Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS).
Limited evidence suggests that HLA-B*1502 may be a risk factor for the development of SJS/TEN in patients of Chinese ancestry taking other antiepileptic drugs associated with SJS/TEN, including phenytoin. Consideration should be given to avoiding use of other drugs associated with SJS/TEN in HLA-B*1502 positive patients, when alternative therapies are otherwise equally acceptable.
Hypersensitivity Reactions and HLA-A*3101 Allele
Retrospective case-control studies in patients of European, Korean, and Japanese ancestry have found a moderate association between the risk of developing hypersensitivity reactions and the presence of HLA-A*3101, an inherited allelic variant of the HLA-A gene, in patients using carbamazepine. These hypersensitivity reactions include SJS/TEN, maculopapular eruptions, and Drug Reaction with Eosinophilia and Systemic Symptoms [see DRESS/Multiorgan hypersensitivity below].
HLA-A*3101 is expected to be carried by more than 15% of patients of Japanese, Native American, Southern Indian (for example, Tamil Nadu), and some Arabic ancestry; up to about 10% in patients of Han Chinese, Korean, European, Latin American, and other Indian ancestry; and up to about 5% in African-Americans and patients of Thai, Taiwanese, and Chinese (Hong Kong) ancestry.
The risks and benefits of carbamazepine therapy should be weighed before considering carbamazepine in patients known to be positive for HLA-A*3101.
Application of HLA genotyping as a screening tool has important limitations and must never substitute for appropriate clinical vigilance and patient management. Many HLA-B*1502-positive and HLA-A*3101-positive patients treated with carbamazepine will not develop SJS/TEN or other hypersensitivity reactions, and these reactions can still occur infrequently in HLA-B*1502-negative and HLA-A*3101-negative patients of any ethnicity. The role of other possible factors in the development of, and morbidity from, SJS/TEN and other hypersensitivity reactions, such as antiepileptic drug (AED) dose, compliance, concomitant medications, comorbidities, and the level of dermatologic monitoring, have not been studied.
Aplastic Anemia and Agranulocytosis
Aplastic anemia and agranulocytosis have been reported in association with the use of carbamazepine [see BOXED WARNING]. Patients with a history of adverse hematologic reaction to any drug may be particularly at risk of bone marrow depression.
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as multiorgan hypersensitivity, has occurred with carbamazepine. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematologic abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. This disorder is variable in its expression, and other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity (e.g., fever, lymphadenopathy) may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Carbamazepine should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
Hypersensitivity
Hypersensitivity reactions to carbamazepine have been reported in patients who previously experienced this reaction to anticonvulsants including phenytoin, primidone, and phenobarbital. If such history is present, benefits and risks should be carefully considered and, if carbamazepine is initiated, the signs and symptoms of hypersensitivity should be carefully monitored.
Patients should be informed that about a third of patients who have had hypersensitivity reactions to carbamazepine also experience hypersensitivity reactions with oxcarbazepine (Trileptal®).
Anaphylaxis and Angioedema
Rare cases of anaphylaxis and angioedema involving the larynx, glottis, lips, and eyelids have been reported in patients after taking the first or subsequent doses of carbamazepine. Angioedema associated with laryngeal edema can be fatal. If a patient develops any of these reactions after treatment with carbamazepine, the drug should be discontinued, and an alternative treatment started. These patients should not be re-challenged with the drug.
Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including carbamazepine, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 years to 100 years) in the clinical trials analyzed. Table 1 shows absolute and relative risk by indication for all evaluated AEDs.
Table 1 Risk by Indication for Antiepileptic Drugs in the Pooled Analysis Indication Placebo Patients with Events Per 1,000 Patients Drug Patients with Events Per 1,000 Patients Relative Risk:
Incidence of Events in Drug Patients/Incidence in Placebo PatientsRisk Difference:
Additional Drug Patients with Events Per 1,000 PatientsEpilepsy 1.0 3.4 3.5 2.4 Psychiatric 5.7 8.5 1.5 2.9 Other 1.0 1.8 1.9 0.9 Total 2.4 4.3 1.8 1.9 The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing carbamazepine, or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
General
Carbamazepine has shown mild anticholinergic activity that may be associated with increased intraocular pressure; therefore, patients with increased intraocular pressure should be closely observed during therapy.
Because of the relationship of the drug to other tricyclic compounds, the possibility of activation of a latent psychosis and, in elderly patients, of confusion or agitation should be borne in mind.
The use of carbamazepine should be avoided in patients with a history of hepatic porphyria (e.g., acute intermittent porphyria, variegate porphyria, porphyria cutanea tarda). Acute attacks have been reported in such patients receiving carbamazepine therapy. Carbamazepine administration has also been demonstrated to increase porphyrin precursors in rodents, a presumed mechanism for the induction of acute attacks of porphyria.
As with all antiepileptic drugs, carbamazepine should be withdrawn gradually to minimize the potential of increased seizure frequency.
Hyponatremia can occur as a result of treatment with carbamazepine. In many cases, the hyponatremia appears to be caused by the syndrome of inappropriate antidiuretic hormone secretion (SIADH). The risk of developing SIADH with carbamazepine treatment appears to be dose-related. Elderly patients and patients treated with diuretics are at greater risk of developing hyponatremia. Signs and symptoms of hyponatremia include headache, new or increased seizure frequency, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which can lead to falls. Consider discontinuing carbamazepine in patients with symptomatic hyponatremia.
Usage in Pregnancy
Carbamazepine can cause fetal harm when administered to a pregnant woman.
Epidemiological data suggest that there may be an association between the use of carbamazepine during pregnancy and congenital malformations, including spina bifida. There have also been reports that associate carbamazepine with developmental disorders and congenital anomalies (e.g., craniofacial defects, cardiovascular malformations, and anomalies involving various body systems). Developmental delays based on neurobehavioral assessments have been reported. When treating or counseling women of childbearing potential, the prescribing physician will wish to weigh the benefits of therapy against the risks. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Retrospective case reviews suggest that, compared with monotherapy, there may be a higher prevalence of teratogenic effects associated with the use of anticonvulsants in combination therapy. Therefore, if therapy is to be continued, monotherapy may be preferable for pregnant women.
In humans, transplacental passage of carbamazepine is rapid (30 minutes to 60 minutes), and the drug is accumulated in the fetal tissues, with higher levels found in liver and kidney than in brain and lung.
Carbamazepine has been shown to have adverse effects in reproduction studies in rats when given orally in dosages 10 times to 25 times the maximum human daily dosage (MHDD) of 1,200 mg on a mg/kg basis or 1.5 times to 4 times the MHDD on a mg/m2 basis. In rat teratology studies, 2 of 135 offspring showed kinked ribs at 250 mg/kg and 4 of 119 offspring at 650 mg/kg showed other anomalies (cleft palate, 1; talipes, 1; anophthalmos, 2). In reproduction studies in rats, nursing offspring demonstrated a lack of weight gain and an unkempt appearance at a maternal dosage level of 200 mg/kg.
Antiepileptic drugs should not be discontinued abruptly in patients in whom the drug is administered to prevent major seizures because of the strong possibility of precipitating status epilepticus with attendant hypoxia and threat to life. In individual cases where the severity and frequency of the seizure disorder are such that removal of medication does not pose a serious threat to the patient, discontinuation of the drug may be considered prior to and during pregnancy, although it cannot be said with any confidence that even minor seizures do not pose some hazard to the developing embryo or fetus.
Tests to detect defects using currently accepted procedures should be considered a part of routine prenatal care in childbearing women receiving carbamazepine.
There have been a few cases of neonatal seizures and/or respiratory depression associated with maternal carbamazepine and other concomitant anticonvulsant drug use. A few cases of neonatal vomiting, diarrhea, and/or decreased feeding have also been reported in association with maternal carbamazepine use. These symptoms may represent a neonatal withdrawal syndrome.
To provide information regarding the effects of in utero exposure to carbamazepine, physicians are advised to recommend that pregnant patients taking carbamazepine enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry. This can be done by calling the toll-free number 1-888-233-2334, and must be done by patients themselves. Information on the registry can also be found at the website http://www.aedpregnancyregistry.org/.
-
PRECAUTIONS
General
Before initiating therapy, a detailed history and physical examination should be made.
Carbamazepine should be used with caution in patients with a mixed seizure disorder that includes atypical absence seizures, since in these patient's carbamazepine has been associated with increased frequency of generalized convulsions [see INDICATIONS AND USAGE].
Therapy should be prescribed only after critical benefit-to-risk appraisal in patients with a history of cardiac conduction disturbance, including second- and third-degree AV heart block; cardiac, hepatic, or renal damage; adverse hematologic or hypersensitivity reaction to other drugs, including reactions to other anticonvulsants; or interrupted courses of therapy with carbamazepine.
AV heart block, including second- and third-degree block, have been reported following carbamazepine treatment. This occurred generally, but not solely, in patients with underlying EKG abnormalities or risk factors for conduction disturbances.
Hepatic effects, ranging from slight elevations in liver enzymes to rare cases of hepatic failure have been reported [see ADVERSE REACTIONS and PRECAUTIONS, Laboratory Tests]. In some cases, hepatic effects may progress despite discontinuation of the drug. In addition, rare instances of vanishing bile duct syndrome have been reported. This syndrome consists of a cholestatic process with a variable clinical course ranging from fulminant to indolent, involving the destruction and disappearance of the intrahepatic bile ducts. Some, but not all, cases are associated with features that overlap with other immunoallergenic syndromes, such as multiorgan hypersensitivity (DRESS syndrome) and serious dermatologic reactions. As an example, there has been a report of vanishing bile duct syndrome associated with Stevens-Johnson syndrome and in another case an association with fever and eosinophilia.
Information for Patients
Patients should be informed of the availability of a Medication Guide, and they should be instructed to read the Medication Guide before taking carbamazepine.
Patients should be made aware of the early toxic signs and symptoms of a potential hematologic problem, as well as dermatologic, hypersensitivity or hepatic reactions. These symptoms may include, but are not limited to, fever, sore throat, rash, ulcers in the mouth, easy bruising, lymphadenopathy and petechial or purpuric hemorrhage, and in the case of liver reactions, anorexia, nausea/vomiting, or jaundice. The patient should be advised that, because these signs and symptoms may signal a serious reaction, that they must report any occurrence immediately to a physician. In addition, the patient should be advised that these signs and symptoms should be reported even if mild or when occurring after extended use.
Patients should be advised that serious skin reactions have been reported in association with carbamazepine. In the event a skin reaction should occur while taking carbamazepine, patients should consult with their physician immediately [see WARNINGS].
Patients should be advised that anaphylactic reactions and angioedema may occur during treatment with carbamazepine [see WARNINGS]. Advise patients to immediately report signs and symptoms suggesting angioedema (swelling of the face, eyes, lips, or tongue, or difficulty in swallowing or breathing) and to stop taking the drug until they have consulted with their healthcare provider.
Patients, their caregivers, and families should be counseled that AEDs, including carbamazepine, may increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
Carbamazepine may interact with some drugs. Therefore, patients should be advised to report to their doctors the use of any other prescription or nonprescription medications or herbal products.
Caution should be exercised if alcohol is taken in combination with carbamazepine therapy, due to a possible additive sedative effect.
Since dizziness and drowsiness may occur, patients should be cautioned about the hazards of operating machinery or automobiles or engaging in other potentially dangerous tasks.
Patients should be encouraged to enroll in the NAAED Pregnancy Registry if they become pregnant. This registry is collecting information about the safety of antiepileptic drugs during pregnancy. To enroll, patients can call the toll-free number 1-888-233-2334 [see WARNINGS, Usage in Pregnancy subsection].
Laboratory Tests
For genetically at-risk patients [see WARNINGS], high-resolution 'HLA-B*1502 typing' is recommended. The test is positive if either one or two HLA-B*1502 alleles are detected and negative if no HLA-B*1502 alleles are detected.
Complete pretreatment blood counts, including platelets and possibly reticulocytes and serum iron, should be obtained as a baseline. If a patient in the course of treatment exhibits low or decreased white blood cell or platelet counts, the patient should be monitored closely. Discontinuation of the drug should be considered if any evidence of significant bone marrow depression develops.
Baseline and periodic evaluations of liver function, particularly in patients with a history of liver disease, must be performed during treatment with this drug since liver damage may occur [see PRECAUTIONS, General and ADVERSE REACTIONS]. Carbamazepine should be discontinued, based on clinical judgment, if indicated by newly occurring or worsening clinical or laboratory evidence of liver dysfunction or hepatic damage, or in the case of active liver disease.
Baseline and periodic eye examinations, including slit-lamp, funduscopy, and tonometry, are recommended since many phenothiazines and related drugs have been shown to cause eye changes.
Baseline and periodic complete urinalysis and BUN determinations are recommended for patients treated with this agent because of observed renal dysfunction.
Monitoring of blood levels [see CLINICAL PHARMACOLOGY] has increased the efficacy and safety of anticonvulsants. This monitoring may be particularly useful in cases of dramatic increase in seizure frequency and for verification of compliance. In addition, measurement of drug serum levels may aid in determining the cause of toxicity when more than one medication is being used.
Thyroid function tests have been reported to show decreased values with carbamazepine administered alone.
Interference with some pregnancy tests has been reported.
Drug Interactions
Clinically meaningful drug interactions have occurred with concomitant medications and include (but are not limited to) the following:
Agents That May Affect Carbamazepine Plasma Levels
When carbamazepine is given with drugs that can increase or decrease carbamazepine levels, close monitoring of carbamazepine levels is indicated, and dosage adjustment may be required.
Agents That Increase Carbamazepine Levels
CYP3A4 inhibitors inhibit carbamazepine metabolism and can thus increase plasma carbamazepine levels. Drugs that have been shown, or would be expected, to increase plasma carbamazepine levels include aprepitant, cimetidine, ciprofloxacin, danazol, diltiazem, macrolides (e.g., erythromycin, clarithromycin), fluoxetine, fluvoxamine, trazodone, omeprazole, oxybutynin, isoniazid, niacinamide (nicotinamide), azoles (e.g., ketaconazole, itraconazole, fluconazole, voriconazole), acetazolamide, verapamil, ticlopidine, grapefruit juice, and protease inhibitors.
Human microsomal epoxide hydrolase has been identified as the enzyme responsible for the formation of the 10,11-transdiol derivative from carbamazepine-10,11 epoxide. Coadministration of inhibitors of human microsomal epoxide hydrolase may result in increased carbamazepine-10,11 epoxide plasma concentrations. Accordingly, the dosage of carbamazepine should be adjusted and/or the plasma levels monitored when used concomitantly with loxapine, quetiapine, valproic acid, or brivaracetam.
Agents That Decrease Carbamazepine Levels
CYP3A4 inducers can increase the rate of carbamazepine metabolism. Drugs that have been shown, or that would be expected, to decrease plasma carbamazepine levels include cisplatin, doxorubicin HCl, felbamate, fosphenytoin, rifampin, phenobarbital, phenytoin, primidone, methsuximide, theophylline, aminophylline.
Effect of Carbamazepine on Plasma Levels of Concomitant Agents
Decreased Levels of Concomitant Medications
Carbamazepine is a potent inducer of hepatic 3A4 and is also known to be an inducer of CYP1A2, 2B6, 2C8/9/19, and may therefore reduce plasma concentrations of co-medications mainly metabolized by CYP 1A2, 2B6, 2C8/9/19, and 3A4, through induction of their metabolism. When used concomitantly with carbamazepine, monitoring of concentrations or dosage adjustment of these agents may be necessary:
- When carbamazepine is added to aripiprazole, the aripiprazole dose should be doubled. Additional dose increases should be based on clinical evaluation. If carbamazepine is later withdrawn, the aripiprazole dose should be reduced.
- When carbamazepine is used with tacrolimus, monitoring of tacrolimus blood concentrations and appropriate dosage adjustments are recommended.
- The use of concomitant strong CYP3A4 inducers, such as carbamazepine should be avoided with temsirolimus. If patients must be coadministered carbamazepine with temsirolimus, an adjustment of temsirolimus dosage should be considered.
- The use of carbamazepine with lapatinib should generally be avoided. If carbamazepine is started in a patient already taking lapatinib, the dose of lapatinib should be gradually titrated up. If carbamazepine is discontinued, the lapatinib dose should be reduced.
- Concomitant use of carbamazepine with nefazodone results in plasma concentrations of nefazodone and its active metabolite insufficient to achieve a therapeutic effect. Coadministration of carbamazepine with nefazodone is contraindicated [see CONTRAINDICATIONS].
- Monitor concentrations of valproate when carbamazepine is introduced or withdrawn in patients using valproic acid.
In addition, carbamazepine causes, or would be expected to cause, decreased levels of the following drugs, for which monitoring of concentrations or dosage adjustment may be necessary: acetaminophen, albendazole, alprazolam, aprepitant, buprenorphone, bupropion, citalopram, clonazepam, clozapine, corticosteroids (e.g., prednisolone, dexamethasone), cyclosporine, dicumarol, dihydropyridine calcium channel blockers (e.g., felodipine), doxycycline, eslicarbazepine, ethosuximide, everolimus, haloperidol, imatinib, itraconazole, lamotrigine, levothyroxine, methadone, methsuximide, mianserin, midazolam, olanzapine, oral and other hormonal contraceptives, oxcarbazepine, paliperidone, phensuximide, phenytoin, praziquantel, protease inhibitors, risperidone, sertraline, sirolimus, tadalafil, theophylline, tiagabine, topiramate, tramadol, trazodone, tricyclic antidepressants (e.g., imipramine, amitriptyline, nortriptyline), valproate, warfarin, ziprasidone, zonisamide.
Other Drug Interactions
- Cyclophosphamide is an inactive prodrug and is converted to its active metabolite in part by CYP3A. The rate of metabolism and the leukopenic activity of cyclophosphamide are reportedly increased by chronic coadministration of CYP3A4 inducers. There is a potential for increased cyclophosphamide toxicity when coadministered with carbamazepine.
- Concomitant administration of carbamazepine and lithium may increase the risk of neurotoxic side effects.
- Concomitant use of carbamazepine with olanzapine, dantrolene, or ibuprofen may increase plasma carbamazepine levels.
- Concomitant use of carbamazepine and isoniazid has been reported to increase isoniazid-induced hepatotoxicity.
- Alterations of thyroid function have been reported in combination therapy with other anticonvulsant medications.
- Concomitant use of carbamazepine with hormonal contraceptive products (e.g., oral, and levonorgestrel subdermal implant contraceptives) may render the contraceptives less effective because the plasma concentrations of the hormones may be decreased. Breakthrough bleeding and unintended pregnancies have been reported. Alternative or back-up methods of contraception should be considered.
- Resistance to the neuromuscular blocking action of the nondepolarizing neuromuscular blocking agents pancuronium, vecuronium, rocuronium and cisatracurium has occurred in patients chronically administered carbamazepine. Whether or not carbamazepine has the same effect on other non-depolarizing agents is unknown. Patients should be monitored closely for more rapid recovery from neuromuscular blockade than expected, and infusion rate requirements may be higher.
- Concomitant use of carbamazepine with rivaroxaban, apixaban, dabigatran, and edoxaban (direct acting oral anticoagulants) is expected to result in decreased plasma concentrations of these anticoagulants that may be insufficient to achieve the intended therapeutic effect. In general, coadministration of carbamazepine with rivaroxaban, apixaban, dabigatran, and edoxaban should be avoided.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carbamazepine, when administered to Sprague-Dawley rats for two years in the diet at doses of 25 mg/kg/day, 75 mg/kg/day, and 250 mg/kg/day, resulted in a dose-related increase in the incidence of hepatocellular tumors in females and of benign interstitial cell adenomas in the testes of males.
Carbamazepine must, therefore, be considered to be carcinogenic in Sprague-Dawley rats. Bacterial and mammalian mutagenicity studies using carbamazepine produced negative results. The significance of these findings relative to the use of carbamazepine in humans is, at present, unknown.
Nursing Mothers
Carbamazepine and its epoxide metabolite are transferred to breast milk. The ratio of the concentration in breast milk to that in maternal plasma is about 0.4 for carbamazepine and about 0.5 for the epoxide. The estimated doses given to the newborn during breastfeeding are in the range of 2 mg to 5 mg daily for carbamazepine and 1 mg to 2 mg daily for the epoxide.
Because of the potential for serious adverse reactions in nursing infants from carbamazepine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Substantial evidence of carbamazepine's effectiveness for use in the management of children with epilepsy [see INDICATIONS AND USAGE for specific seizure types] is derived from clinical investigations performed in adults and from studies in several in vitro systems which support the conclusion that (1) the pathogenetic mechanisms underlying seizure propagation are essentially identical in adults and children, and (2) the mechanism of action of carbamazepine in treating seizures is essentially identical in adults and children.
Taken as a whole, this information supports a conclusion that the generally accepted therapeutic range of total carbamazepine in plasma (i.e., 4 mcg/mL to 12 mcg/mL) is the same in children and adults.
The evidence assembled was primarily obtained from short-term use of carbamazepine. The safety of carbamazepine in children has been systematically studied up to 6 months. No longer-term data from clinical trials is available.
-
ADVERSE REACTIONS
If adverse reactions are of such severity that the drug must be discontinued, the physician must be aware that abrupt discontinuation of any anticonvulsant drug in a responsive epileptic patient may lead to seizures or even status epilepticus with its life-threatening hazards.
The most severe adverse reactions have been observed in the hemopoietic system and skin [see BOXED WARNING], the liver, and the cardiovascular system.
The most frequently observed adverse reactions, particularly during the initial phases of therapy, are dizziness, drowsiness, unsteadiness, nausea, and vomiting. To minimize the possibility of such reactions, therapy should be initiated at the lowest dosage recommended.
The following additional adverse reactions have been reported:
Hemopoietic System: Aplastic anemia, agranulocytosis, pancytopenia, bone marrow depression, thrombocytopenia, leukopenia, leukocytosis, eosinophilia, acute intermittent porphyria, variegate porphyria, porphyria cutanea tarda.
Skin: Toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome (SJS) [see BOXED WARNING], Acute Generalized Exanthematous Pustulosis (AGEP), pruritic and erythematous rashes, urticaria, photosensitivity reactions, alterations in skin pigmentation, exfoliative dermatitis, erythema multiforme and nodosum, purpura, aggravation of disseminated lupus erythematosus, alopecia, diaphoresis, onychomadesis and hirsutism. In certain cases, discontinuation of therapy may be necessary.
Cardiovascular System: Congestive heart failure, edema, aggravation of hypertension, hypotension, syncope and collapse, aggravation of coronary artery disease, arrhythmias and AV block, thrombophlebitis, thromboembolism (e.g., pulmonary embolism), and adenopathy or lymphadenopathy.
Some of these cardiovascular complications have resulted in fatalities. Myocardial infarction has been associated with other tricyclic compounds.
Liver: Abnormalities in liver function tests, cholestatic and hepatocellular jaundice, hepatitis, very rare cases of hepatic failure.
Pancreatic: Pancreatitis.
Respiratory System: Pulmonary hypersensitivity characterized by fever, dyspnea, pneumonitis, or pneumonia.
Genitourinary System: Urinary frequency, acute urinary retention, oliguria with elevated blood pressure, azotemia, renal failure, and impotence. Albuminuria, glycosuria, elevated BUN, and microscopic deposits in the urine have also been reported. There have been rare reports of impaired male fertility and/or abnormal spermatogenesis.
Testicular atrophy occurred in rats receiving carbamazepine orally from 4 weeks to 52 weeks at dosage levels of 50 mg/kg/day to 400 mg/kg/day. Additionally, rats receiving carbamazepine in the diet for 2 years at dosage levels of 25 mg/kg/day, 75 mg/kg/day, and 250 mg/kg/day had a dose-related incidence of testicular atrophy and aspermatogenesis. In dogs, it produced a brownish discoloration, presumably a metabolite, in the urinary bladder at dosage levels of 50 mg/kg and higher. Relevance of these findings to humans is unknown.
Nervous System: Dizziness, drowsiness, disturbances of coordination, confusion, headache, fatigue, blurred vision, visual hallucinations, transient diplopia, oculomotor disturbances, nystagmus, speech disturbances, abnormal involuntary movements, peripheral neuritis and paresthesias, depression with agitation, talkativeness, tinnitus, hyperacusis, neuroleptic malignant syndrome.
There have been reports of associated paralysis and other symptoms of cerebral arterial insufficiency, but the exact relationship of these reactions to the drug has not been established.
Isolated cases of neuroleptic malignant syndrome have been reported both with and without concomitant use of psychotropic drugs.
Digestive System: Nausea, vomiting, gastric distress and abdominal pain, diarrhea, constipation, anorexia, and dryness of the mouth and pharynx, including glossitis and stomatitis.
Eyes: Scattered punctate cortical lens opacities, increased intraocular pressure [see WARNINGS, General] as well as conjunctivitis, have been reported. Although a direct causal relationship has not been established, many phenothiazines and related drugs have been shown to cause eye changes.
Musculoskeletal System: Aching joints and muscles, and leg cramps.
Metabolism: Fever and chills. Hyponatremia [see WARNINGS, General]. Decreased levels of plasma calcium have been reported. Osteoporosis has been reported.
Isolated cases of a lupus erythematosus-like syndrome have been reported. There have been occasional reports of elevated levels of cholesterol, HDL cholesterol, and triglycerides in patients taking anticonvulsants.
A case of aseptic meningitis, accompanied by myoclonus and peripheral eosinophilia, has been reported in a patient taking carbamazepine in combination with other medications. The patient was successfully de-challenged, and the meningitis reappeared upon re-challenge with carbamazepine.
- DRUG ABUSE AND DEPENDENCE
-
OVERDOSAGE
Acute Toxicity
Lowest known lethal dose: adults, 3.2 g (a 24-year-old woman died of a cardiac arrest and a 24-year-old man died of pneumonia and hypoxic encephalopathy); children, 4 g (a 14-year-old girl died of a cardiac arrest), 1.6 g (a 3-year-old girl died of aspiration pneumonia).
Oral LD50 in animals (mg/kg): mice, 1,100 to 3,750; rats, 3,850 to 4,025; rabbits, 1,500 to 2,680; guinea pigs, 920.
Signs and Symptoms
The first signs and symptoms appear after 1 hour to 3 hours. Neuromuscular disturbances are the most prominent. Cardiovascular disorders are generally milder, and severe cardiac complications occur only when very high doses (greater than 60 g) have been ingested.
Nervous System and Muscles
Impairment of consciousness ranging in severity to deep coma. Convulsions, especially in small children. Motor restlessness, muscular twitching, tremor, athetoid movements, opisthotonos, ataxia, drowsiness, dizziness, mydriasis, nystagmus, adiadochokinesia, ballism, psychomotor disturbances, dysmetria. Initial hyperreflexia followed by hyporeflexia.
Treatment
The prognosis in cases of severe poisoning is critically dependent upon prompt elimination of the drug, which may be achieved by inducing vomiting, irrigating the stomach, and by taking appropriate steps to diminish absorption. If these measures cannot be implemented without risk on the spot, the patient should be transferred at once to a hospital, while ensuring that vital functions are safeguarded. There is no specific antidote.
Measures to Accelerate Elimination
Forced diuresis.
Dialysis is indicated only in severe poisoning associated with renal failure. Replacement transfusion is indicated in severe poisoning in small children.
Respiratory Depression
Keep the airways free; resort, if necessary, to endotracheal intubation, artificial respiration, and administration of oxygen.
Hypotension, Shock
Keep the patient's legs raised and administer a plasma expander. If blood pressure fails to rise despite measures taken to increase plasma volume, use of vasoactive substances should be considered.
Warning
Diazepam or barbiturates may aggravate respiratory depression (especially in children), hypotension, and coma. However, barbiturates should not be used if drugs that inhibit monoamine oxidase have also been taken by the patient either in overdosage or in recent therapy (within 1 week).
Surveillance
Respiration, cardiac function (ECG monitoring), blood pressure, body temperature, pupillary reflexes, and kidney and bladder function should be monitored for several days.
Treatment of Blood Count Abnormalities
If evidence of significant bone marrow depression develops, the following recommendations are suggested: (1) stop the drug, (2) perform daily CBC, platelet, and reticulocyte counts, (3) do a bone marrow aspiration and trephine biopsy immediately and repeat with sufficient frequency to monitor recovery.
Special periodic studies might be helpful as follows: (1) white cell and platelet antibodies, (2) 59Fe-ferrokinetic studies, (3) peripheral blood cell typing, (4) cytogenetic studies on marrow and peripheral blood, (5) bone marrow culture studies for colony-forming units, (6) hemoglobin electrophoresis for A2 and F hemoglobin, and (7) serum folic acid and B12 levels.
A fully developed aplastic anemia will require appropriate, intensive monitoring and therapy, for which specialized consultation should be sought.
-
DOSAGE AND ADMINISTRATION
(SEE TABLE BELOW)
Monitoring of blood levels has increased the efficacy and safety of anticonvulsants [see PRECAUTIONS, Laboratory Tests]. Dosage should be adjusted to the needs of the individual patient. A low initial daily dosage with a gradual increase is advised. As soon as adequate control is achieved, the dosage may be reduced very gradually to the minimum effective level. Medication should be taken with meals.
Conversion of patients from oral carbamazepine tablets to carbamazepine suspension: Patients should be converted by administering the same number of mg per day in smaller, more frequent doses (i.e., twice a day tablets to three times a day suspension).
Carbamazepine extended-release tablets are an extended-release formulation for twice a day administration. When converting patients from carbamazepine conventional tablets to carbamazepine extended-release tablets, the same total daily mg dose of carbamazepine extended-release tablets should be administered. Carbamazepine extended-release tablets must be swallowed whole and never crushed or chewed. Carbamazepine extended-release tablets should be inspected for chips or cracks. Damaged tablets, or tablets without a release portal, should not be consumed. Carbamazepine extended-release tablet coating is not absorbed and is excreted in the feces; these coatings may be noticeable in the stool.
Epilepsy
[see INDICATIONS AND USAGE]
Adults and children over 12 years of age
Initial
200 mg twice a day for carbamazepine extended-release tablets (400 mg/day). Increase at weekly intervals by adding up to 200 mg/day using a twice a day regimen of carbamazepine extended-release tablets or three times a day or four times a day regimen of the other formulations until the optimal response is obtained. Dosage generally should not exceed 1,000 mg daily in children 12 years to 15 years of age, and 1,200 mg daily in patients above 15 years of age. Doses up to 1,600 mg daily have been used in adults in rare instances.
Children 6 years to 12 years of age
Initial
100 mg twice a day for carbamazepine extended-release tablets (200 mg/day). Increase at weekly intervals by adding up to 100 mg/day using a twice a day regimen of carbamazepine extended-release tablets or three times a day or four times a day regimen of the other formulations until the optimal response is obtained. Dosage generally should not exceed 1,000 mg daily.
Children under 6 years of age
Initial
10 to 20 mg/kg/day twice a day or three times a day as tablets, or four times a day as suspension. Increase weekly to achieve optimal clinical response administered three times a day or four times a day.
Maintenance
Ordinarily, optimal clinical response is achieved at daily doses below 35 mg/kg. If satisfactory clinical response has not been achieved, plasma levels should be measured to determine whether or not they are in the therapeutic range. No recommendation regarding the safety of carbamazepine for use at doses above 35 mg/kg/24 hours can be made.
Combination Therapy
Carbamazepine may be used alone or with other anticonvulsants. When added to existing anticonvulsant therapy, the drug should be added gradually while the other anticonvulsants are maintained or gradually decreased, except phenytoin, which may have to be increased [see PRECAUTIONS, Drug Interactions, and Pregnancy].
Trigeminal Neuralgia
[see INDICATIONS AND USAGE]
Initial
On the first day, 100 mg twice a day for carbamazepine extended-release tablets, for a total daily dose of 200 mg. This daily dose may be increased by up to 200 mg/day using increments of 100 mg every 12 hours for carbamazepine extended-release tablets, only as needed to achieve freedom from pain. Do not exceed 1,200 mg daily.
Maintenance
Control of pain can be maintained in most patients with 400 mg to 800 mg daily. However, some patients may be maintained on as little as 200 mg daily, while others may require as much as 1,200 mg daily. At least once every 3 months throughout the treatment period, attempts should be made to reduce the dose to the minimum effective level or even to discontinue the drug.
Dosage Information Initial Dose Subsequent Dose Maximum Daily Dose Indication Carbamazepine Extended-Release Tablets Carbamazepine Extended-Release Tablets Carbamazepine Extended-Release Tablets Epilepsy
Under 6 yr.-- -- -- 6 yr. to 12 yr. 100 mg twice a day
(200 mg/day)Add 100 mg/day at weekly intervals, twice a day 1,000 mg/24 hr. Over 12 yr. 200 mg twice a day
(400 mg/day)Add up to 200 mg/day at weekly intervals, twice a day 1,000 mg/24 hr. (12 yr. to 15 yr.)
1,200 mg/24 hr. (˃ 15 yr.)
1,600 mg/24 hr. (adults, in rare instances)Trigeminal Neuralgia 100 mg twice a day
(200 mg/day)Add up to 200 mg/day in increments of 100 mg every 12 hr. 1,200 mg/24 hr. -
HOW SUPPLIED
Carbamazepine extended-release tablets, USP, 100 mg are available as pinkish white, round, coated tablets, imprinted "B28" and a release portal on one side. They are supplied as follows:
Bottles of 100 with a child-resistant closure, NDC: 0832-6022-11
Carbamazepine extended-release tablets, USP, 200 mg are available as pinkish white, round, coated tablets, imprinted "B29" and a release portal on one side. They are supplied as follows:
Bottles of 100 with a child-resistant closure, NDC: 0832-6023-11
Carbamazepine extended-release tablets, USP, 400 mg are available as pinkish white, round, coated tablets, imprinted "B30" and a release portal on one side. They are supplied as follows:
Bottles of 100 with a child-resistant closure, NDC: 0832-6024-11
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
Carbamazepine Extended-Release Tablets, USP
(karʺ ba mazʹ e peen), for oral useRead this Medication Guide before you start taking carbamazepine extended-release tablets and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is the most important information I should know about carbamazepine extended-release tablets?
Do not stop taking carbamazepine extended-release tablets without first talking to your healthcare provider.
Stopping carbamazepine extended-release tablets suddenly can cause serious problems.
Carbamazepine extended-release tablets can cause serious side effects, including:
- 1.
Carbamazepine extended-release tablets may cause rare but serious skin rashes that may lead to death. These serious skin reactions are more likely to happen when you begin taking carbamazepine extended-release tablets within the first four months of treatment but may occur at later times. These reactions can happen in anyone, but are more likely in people of Asian descent. If you are of Asian descent, you may need a genetic blood test before you take carbamazepine extended-release tablets to see if you are at a higher risk for serious skin reactions with this medicine. Symptoms may include:
- skin rash
- hives
- sores in your mouth
- blistering or peeling of the skin
- 2. Carbamazepine extended-release tablets may cause rare but serious blood problems. Symptoms may include:
- fever, sore throat, or other infections that come and go or do not go away
- easy bruising
- red or purple spots on your body
- bleeding gums or nose bleeds
- severe fatigue or weakness
- 3. Carbamazepine extended-release tablets may cause allergic reactions or serious problems, which may affect organs and other parts of your body like the liver or blood cells. You may or may not have a rash with these types of reactions.
Call your healthcare provider right away if you have any of the following:- swelling of your face, eyes, lips, or tongue
- a skin rash
- painful sores in the mouth or around your eyes
- unusual bruising or bleeding
- frequent infections or infections that do not go away
- fever, swollen glands, or sore throat that do not go away or come and go
- trouble swallowing or breathing
- hives
- yellowing of your skin or eyes
- severe fatigue or weakness
- severe muscle pain
- 4. Like other antiepileptic drugs, carbamazepine extended-release tablets may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call your healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
How can I watch for early symptoms of suicidal thoughts and actions?
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- Keep all follow-up visits with your healthcare provider as scheduled.
Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
Do not stop carbamazepine extended-release tablets without first talking to a healthcare provider.
Stopping carbamazepine extended-release tablets suddenly can cause serious problems. You should talk to your healthcare provider before stopping.
Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
What are carbamazepine extended-release tablets?
Carbamazepine extended-release tablets are a prescription medicine used to treat:
- certain types of seizures (partial, tonic-clonic, mixed)
- certain types of nerve pain (trigeminal and glossopharyngeal neuralgia)
Carbamazepine extended-release tablets are not a regular pain medicine and should not be used for aches or pains.
Who should not take carbamazepine extended-release tablets?
Do not take carbamazepine extended-release tablets if you:
- have a history of bone marrow depression.
- are allergic to carbamazepine or any of the ingredients in carbamazepine extended-release tablets. See the end of this Medication Guide for a complete list of ingredients in carbamazepine extended-release tablets.
- take nefazodone.
- are allergic to medicines called tricyclic antidepressants (TCAs). Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure.
- have taken a medicine called a Monoamine Oxidase Inhibitor (MAOI) in the last 14 days. Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure.
What should I tell my healthcare provider before taking carbamazepine extended-release tablets?
Before you take carbamazepine extended-release tablets, tell your healthcare provider if you:
- have or have had suicidal thoughts or actions, depression, or mood problems
- have or ever had heart problems
- have or ever had blood problems
- have or ever had liver problems
- have or ever had kidney problems
- have or ever had allergic reactions to medicines
- have or ever had increased pressure in your eye
- have any other medical conditions
- drink grapefruit juice or eat grapefruit
- use birth control. Carbamazepine extended-release tablets may make your birth control less effective. Tell your healthcare provider if your menstrual bleeding changes while you take birth control and carbamazepine extended-release tablets.
- are pregnant or plan to become pregnant. Carbamazepine may harm your unborn baby. Tell your healthcare provider right away if you become pregnant while taking carbamazepine extended-release tablets. You and your healthcare provider should decide if you should take carbamazepine extended-release tablets while you are pregnant.
- If you become pregnant while taking carbamazepine extended-release tablets, talk to your healthcare provider about registering with the North American Antiepileptic Drug (NAAED) Pregnancy Registry. The purpose of this registry is to collect information about the safety of antiepileptic medicine during pregnancy. You can enroll in this registry by calling 1-888-233-2334.
- are breastfeeding or plan to breastfeed. Carbamazepine passes into breast milk. You and your healthcare provider should discuss whether you should take carbamazepine extended-release tablets or breastfeed; you should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Taking carbamazepine extended-release tablets with certain other medicines may cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take carbamazepine extended-release tablets?
- Do not stop taking carbamazepine extended-release tablets without first talking to your healthcare provider. Stopping carbamazepine extended-release tablets suddenly can cause serious problems. Stopping seizure medicine suddenly in a patient who has epilepsy may cause seizures that will not stop (status epilepticus).
- Take carbamazepine extended-release tablets exactly as prescribed. Your healthcare provider will tell you how much carbamazepine extended-release tablets to take.
- Your healthcare provider may change your dose. Do not change your dose of carbamazepine extended-release tablets without talking to your healthcare provider.
- Take carbamazepine extended-release tablets with food.
- Do not crush, chew, or break carbamazepine extended-release tablets.
- Tell your healthcare provider if you cannot swallow carbamazepine extended-release tablets whole.
- If you take too much carbamazepine extended-release tablets, call your healthcare provider or local Poison Control Center right away.
What should I avoid while taking carbamazepine extended-release tablets?
- Do not drink alcohol or take other drugs that make you sleepy or dizzy while taking carbamazepine extended-release tablets until you talk to your healthcare provider. Carbamazepine extended-release tablets taken with alcohol or drugs that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how carbamazepine extended-release tablets affects you. Carbamazepine extended-release tablets may slow your thinking and motor skills.
What are the possible side effects of carbamazepine extended-release tablets?
See "What is the most important information I should know about carbamazepine extended-release tablets?"
Carbamazepine extended-release tablets may cause other serious side effects. These include:
- Irregular heartbeat – symptoms include:
- Fast, slow, or pounding heartbeat
- Shortness of breath
- Feeling lightheaded
- Fainting
- Liver problems – symptoms include:
- yellowing of your skin or the whites of your eyes
- dark urine
- pain on the right side of your stomach area (abdominal pain)
- easy bruising
- loss of appetite
- nausea or vomiting
Get medical help right away if you have any of the symptoms listed above or listed in "What is the most important information I should know about carbamazepine extended-release tablets?"
The most common side effects of carbamazepine extended-release tablets include:
- dizziness
- drowsiness
- problems with walking and coordination (unsteadiness)
- nausea
- vomiting
These are not all the possible side effects of carbamazepine extended-release tablets. For more information, ask your healthcare provider or pharmacist.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store carbamazepine extended-release tablets?
Store carbamazepine extended-release tablets at 25°C (77°F); excursions permitted from 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
- Keep carbamazepine extended-release tablets dry.
Keep carbamazepine extended-release tablets and all medicines out of the reach of children.
General Information about carbamazepine extended-release tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use carbamazepine extended-release tablets for a condition for which it was not prescribed. Do not give carbamazepine extended-release tablets to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about carbamazepine extended-release tablets. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for the full prescribing information about carbamazepine extended-release tablets that is written for health professionals.
What are the ingredients in carbamazepine extended-release tablets?
Active ingredient: carbamazepine, USP
Inactive ingredients: cellulose acetate, dextrates, ferric oxide red, hydroxyethyl cellulose, hypromellose, magnesium stearate, mannitol, polyethylene glycol, sodium lauryl sulfate, titanium dioxide, ammonium hydroxide, ferrosoferric oxide, propylene glycol and shellac glaze.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
For Medication Guides, please visit www.upsher-smith.com or call 1-888-650-3789.
Made in China
Distributed by
UPSHER-SMITH LABORATORIES. LLC
Maple Grove, MN 55369Revised: 10/2023
- 1.
Carbamazepine extended-release tablets may cause rare but serious skin rashes that may lead to death. These serious skin reactions are more likely to happen when you begin taking carbamazepine extended-release tablets within the first four months of treatment but may occur at later times. These reactions can happen in anyone, but are more likely in people of Asian descent. If you are of Asian descent, you may need a genetic blood test before you take carbamazepine extended-release tablets to see if you are at a higher risk for serious skin reactions with this medicine. Symptoms may include:
-
PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label
NDC: 0832-6022-11
Carbamazepine Extended-Release
Tablets, USP100 mg
Carbamazepine extended-release tablets must be
swallowed whole and never crushed or chewed.PHARMACIST: Dispense the Medication
Guide provided separately to each patient.100 Tablets
Rx onlyUPSHER-SMITH
CSPC

-
PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label
NDC: 0832-6023-11
Carbamazepine Extended-Release
Tablets, USP200 mg
Carbamazepine extended-release tablets must be
swallowed whole and never crushed or chewed.PHARMACIST: Dispense the Medication
Guide provided separately to each patient.100 Tablets
Rx onlyUPSHER-SMITH
CSPC

-
PRINCIPAL DISPLAY PANEL - 400 mg Tablet Bottle Label
NDC: 0832-6024-11
Carbamazepine Extended-Release
Tablets, USP400 mg
Carbamazepine extended-release tablets must be swallowed whole
and never crushed or chewed.PHARMACIST: Dispense the Medication
Guide provided separately to each patient.100 Tablets
Rx onlyUPSHER-SMITH
CSPC

-
INGREDIENTS AND APPEARANCE
CARBAMAZEPINE
carbamazepine tablet, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0832-6022 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Carbamazepine (UNII: 33CM23913M) (Carbamazepine - UNII:33CM23913M) Carbamazepine 100 mg Inactive Ingredients Ingredient Name Strength cellulose acetate (UNII: 3J2P07GVB6) dextrates (UNII: G263MI44RU) ferric oxide red (UNII: 1K09F3G675) hydroxyethyl cellulose, unspecified (UNII: T4V6TWG28D) hypromellose, unspecified (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) mannitol (UNII: 3OWL53L36A) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) sodium lauryl sulfate (UNII: 368GB5141J) titanium dioxide (UNII: 15FIX9V2JP) ammonia (UNII: 5138Q19F1X) ferrosoferric oxide (UNII: XM0M87F357) propylene glycol (UNII: 6DC9Q167V3) shellac (UNII: 46N107B71O) Product Characteristics Color WHITE (pinkish white) Score no score Shape ROUND Size 8mm Flavor Imprint Code B28 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0832-6022-11 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/13/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213311 04/13/2021 CARBAMAZEPIN
carbamazepine tablet, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0832-6023 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Carbamazepine (UNII: 33CM23913M) (Carbamazepine - UNII:33CM23913M) Carbamazepine 200 mg Inactive Ingredients Ingredient Name Strength cellulose acetate (UNII: 3J2P07GVB6) dextrates (UNII: G263MI44RU) ferric oxide red (UNII: 1K09F3G675) hydroxyethyl cellulose, unspecified (UNII: T4V6TWG28D) hypromellose, unspecified (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) mannitol (UNII: 3OWL53L36A) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) sodium lauryl sulfate (UNII: 368GB5141J) titanium dioxide (UNII: 15FIX9V2JP) ammonia (UNII: 5138Q19F1X) ferrosoferric oxide (UNII: XM0M87F357) propylene glycol (UNII: 6DC9Q167V3) shellac (UNII: 46N107B71O) Product Characteristics Color WHITE (pinkish white) Score no score Shape ROUND Size 10mm Flavor Imprint Code B29 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0832-6023-11 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/13/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213311 04/13/2021 CARBAMAZEPINE
carbamazepine tablet, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0832-6024 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Carbamazepine (UNII: 33CM23913M) (Carbamazepine - UNII:33CM23913M) Carbamazepine 400 mg Inactive Ingredients Ingredient Name Strength cellulose acetate (UNII: 3J2P07GVB6) dextrates (UNII: G263MI44RU) ferric oxide red (UNII: 1K09F3G675) hydroxyethyl cellulose, unspecified (UNII: T4V6TWG28D) hypromellose, unspecified (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) mannitol (UNII: 3OWL53L36A) polyethylene glycol, unspecified (UNII: 3WJQ0SDW1A) sodium lauryl sulfate (UNII: 368GB5141J) titanium dioxide (UNII: 15FIX9V2JP) ammonia (UNII: 5138Q19F1X) ferrosoferric oxide (UNII: XM0M87F357) propylene glycol (UNII: 6DC9Q167V3) shellac (UNII: 46N107B71O) Product Characteristics Color WHITE (pinkish white) Score no score Shape ROUND Size 13mm Flavor Imprint Code B30 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0832-6024-11 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/13/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213311 04/13/2021 Labeler - Upsher-Smith Laboratories, LLC (047251004) Establishment Name Address ID/FEI Business Operations CSPC Ouyi Pharmaceutical Co., Ltd 421303775 manufacture(0832-6022, 0832-6023, 0832-6024) , analysis(0832-6022, 0832-6023, 0832-6024) , pack(0832-6022, 0832-6023, 0832-6024)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.