Temazepam by AvKARE TEMAZEPAM capsule
Temazepam by
Drug Labeling and Warnings
Temazepam by is a Prescription medication manufactured, distributed, or labeled by AvKARE. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
Description
Temazepam is a benzodiazepine hypnotic agent. The chemical name is 7-chloro-1,3-dihydro-3-hydroxy-1-methyl-5-phenyl-2 H-1,4-benzodiazepin-2-one, and the structural formula is:
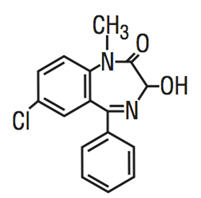
Temazepam is a white, crystalline substance, very slightly soluble in water and sparingly soluble in alcohol USP.
Temazepam capsules, 7.5 mg, 15 mg, 22.5 mg, and 30 mg, are for oral administration.
Active Ingredient: temazepam USP
Inactive Ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate. The capsule shell contains gelatin and titanium dioxide. The capsule shell for the 7.5 mg also contains FD&C Blue #1 and FD&C Red #40; the capsule shell for 15 mg and 22.5 mg also contains D&C Yellow #10, FD&C Blue #1 and FD&C Yellow #6. The imprinting ink contains: D&C Yellow #10, FD&C Blue #1, FD&C Blue #2, FD&C Red #40, iron oxide black and propylene glycol.
-
CLINICAL PHARMACOLOGY
Pharmacokinetics
In a single and multiple dose absorption, distribution, metabolism, and excretion (ADME) study, using 3H labeled drug, temazepam capsules were well absorbed and found to have minimal (8%) first pass metabolism. There were no active metabolites formed and the only significant metabolite present in blood was the O-conjugate. The unchanged drug was 96% bound to plasma proteins. The blood level decline of the parent drug was biphasic with the short half-life ranging from 0.4 to 0.6 hours and the terminal half-life from 3.5 to 18.4 hours (mean 8.8 hours), depending on the study population and method of determination. Metabolites were formed with a half-life of 10 hours and excreted with a half-life of approximately 2 hours. Thus, formation of the major metabolite is the rate limiting step in the biodisposition of temazepam. There is no accumulation of metabolites. A dose-proportional relationship has been established for the area under the plasma concentration/time curve over the 15 to 30 mg dose range.
Temazepam was completely metabolized through conjugation prior to excretion; 80% to 90% of the dose appeared in the urine. The major metabolite was the O-conjugate of temazepam (90%); the O-conjugate of N-desmethyl temazepam was a minor metabolite (7%).
Bioavailability, Induction, and Plasma Levels
Following ingestion of a 30 mg temazepam capsule, measurable plasma concentrations were achieved 10 to 20 minutes after dosing with peak plasma levels ranging from 666 to 982 ng/mL (mean 865 ng/mL) occurring approximately 1.2 to 1.6 hours (mean 1.5 hours) after dosing.
In a 7 day study, in which subjects were given a 30 mg temazepam capsule 1 hour before retiring, steady-state (as measured by the attainment of maximal trough concentrations) was achieved by the third dose. Mean plasma levels of temazepam (for days 2 to 7) were 260±210 ng/mL at 9 hours and 75±80 ng/mL at 24 hours after dosing. A slight trend toward declining 24 hour plasma levels was seen after day 4 in the study, however, the 24 hour plasma levels were quite variable.
At a dose of 30 mg once-a-day for 8 weeks, no evidence of enzyme induction was found in man.
Elimination Rate of Benzodiazepine Hypnotics and Profile of Common Untoward Effects
The type and duration of hypnotic effects and the profile of unwanted effects during administration of benzodiazepine hypnotics may be influenced by the biologic half-life of the administered drug and for some hypnotics, the half-life of any active metabolites formed. Benzodiazepine hypnotics have a spectrum of half-lives from short (<4 hours) to long (>20 hours). When half-lives are long, drug (and for some drugs their active metabolites) may accumulate during periods of nightly administration and be associated with impairments of cognitive and/or motor performance during waking hours; the possibility of interaction with other psychoactive drugs or alcohol will be enhanced. In contrast, if half-lives are shorter, drug (and, where appropriate, its active metabolites) will be cleared before the next dose is ingested, and carry-over effects related to excessive sedation or CNS depression should be minimal or absent. However, during nightly use for an extended period, pharmacodynamic tolerance or adaptation to some effects of benzodiazepine hypnotics may develop. If the drug has a short elimination half-life, it is possible that a relative deficiency of the drug, or, if appropriate, its active metabolites (i.e., in relationship to the receptor site) may occur at some point in the interval between each night's use. This sequence of events may account for 2 clinical findings reported to occur after several weeks of nightly use of rapidly eliminated benzodiazepine hypnotics, namely, increased wakefulness during the last third of the night, and the appearance of increased signs of daytime anxiety.
Controlled Trials Supporting Efficacy
Temazepam capsules improved sleep parameters in clinical studies. Residual medication effects ("hangover") were essentially absent. Early morning awakening, a particular problem in the geriatric patient, was significantly reduced.
Patients with chronic insomnia were evaluated in 2 week, placebo controlled sleep laboratory studies with temazepam capsules at doses of 7.5 mg, 15 mg, and 30 mg, given 30 minutes prior to bedtime. There was a linear dose-response improvement in total sleep time and sleep latency, with significant drug-placebo differences at 2 weeks occurring only for total sleep time at the 2 higher doses, and for sleep latency only at the highest dose.
In these sleep laboratory studies, REM sleep was essentially unchanged and slow wave sleep was decreased. No measurable effects on daytime alertness or performance occurred following temazepam capsules treatment or during the withdrawal period, even though a transient sleep disturbance in some sleep parameters was observed following withdrawal of the higher doses. There was no evidence of tolerance development in the sleep laboratory parameters when patients were given temazepam capsules nightly for at least 2 weeks.
In addition, normal subjects with transient insomnia associated with first night adaptation to the sleep laboratory were evaluated in 24 hour, placebo controlled sleep laboratory studies with temazepam capsules at doses of 7.5 mg, 15 mg, and 30 mg, given 30 minutes prior to bedtime. There was a linear dose-response improvement in total sleep time, sleep latency and number of awakenings, with significant drug-placebo differences occurring for sleep latency at all doses, for total sleep time at the 2 higher doses and for number of awakenings only at the 30 mg dose.
-
INDICATIONS AND USAGE
Temazepam capsules are indicated for the short-term treatment of insomnia (generally 7 to 10 days).
For patients with short-term insomnia, instructions in the prescription should indicate that temazepam capsules should be used for short periods of time (7 to 10 days).
The clinical trials performed in support of efficacy were 2 weeks in duration with the final formal assessment of sleep latency performed at the end of treatment.
-
CONTRAINDICATIONS
Benzodiazepines may cause fetal harm when administered to a pregnant woman. An increased risk of congenital malformations associated with the use of diazepam and chlordiazepoxide during the first trimester of pregnancy has been suggested in several studies. Transplacental distribution has resulted in neonatal CNS depression following the ingestion of therapeutic doses of a benzodiazepine hypnotic during the last weeks of pregnancy.
Reproduction studies in animals with temazepam were performed in rats and rabbits. In a perinatal- postnatal study in rats, oral doses of 60 mg/kg/day resulted in increasing nursling mortality. Teratology studies in rats demonstrated increased fetal resorptions at doses of 30 and 120 mg/kg in one study and increased occurrence of rudimentary ribs, which are considered skeletal variants, in a second study at doses of 240 mg/kg or higher. In rabbits, occasional abnormalities such as exencephaly and fusion or asymmetry of ribs were reported without dose relationship. Although these abnormalities were not found in the concurrent control group, they have been reported to occur randomly in historical controls. At doses of 40 mg/kg or higher, there was an increased incidence of the 13th rib variant when compared to the incidence in concurrent and historical controls.
Temazepam capsules are contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus. Patients should be instructed to discontinue the drug prior to becoming pregnant. The possibility that a woman of childbearing potential may be pregnant at the time of institution of therapy should be considered.
-
WARNINGS
Sleep disturbance may be the presenting manifestation of an underlying physical and/or psychiatric disorder. Consequently, a decision to initiate symptomatic treatment of insomnia should only be made after the patient has been carefully evaluated. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. Worsening of insomnia may be the consequence of an unrecognized psychiatric or physical disorder as may the emergence of new abnormalities of thinking or behavior. Such abnormalities have also been reported to occur in association with the use of drugs with central nervous system depressant activity, including those of the benzodiazepine class. Because some of the worrisome adverse effects of benzodiazepines, including temazepam capsules, appear to be dose related ( see PRECAUTIONS and DOSAGE AND ADMINISTRATION), it is important to use the lowest possible effective dose. Elderly patients are especially at risk.
Some of these changes may be characterized by decreased inhibition, e.g., aggressiveness and extroversion that seem out of character, similar to that seen with alcohol. Other kinds of behavioral changes can also occur, for example, bizarre behavior, agitation, hallucinations, and depersonalization. Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake after ingestion of a sedative-hypnotic, with amnesia for the event) have been reported. These events can occur in sedative-hypnotic-naïve as well as in sedative-hypnotic-experienced persons. Although behaviors such as sleep-driving may occur with temazepam capsules alone at therapeutic doses, the use of alcohol and other CNS depressants with temazepam capsules appears to increase the risk of such behaviors, as does the use of temazepam capsules at doses exceeding the maximum recommended dose. Due to the risk to the patient and the community, discontinuation of temazepam capsules should be strongly considered for patients who report a "sleep-driving" episode. Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with sleep-driving, patients usually do not remember these events. Amnesia and other neuro-psychiatric symptoms may occur unpredictably. In primarily depressed patients, worsening of depression, including suicidal thinking has been reported in association with the use of sedative/hypnotics.
It can rarely be determined with certainty whether a particular instance of the abnormal behaviors listed above is drug induced, spontaneous in origin, or a result of an underlying psychiatric or physical disorder. Nonetheless, the emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
Withdrawal symptoms (of the barbiturate type) have occurred after the abrupt discontinuation of benzodiazepines ( see DRUG ABUSE AND DEPENDENCE).
Severe Anaphylactic and Anaphylactoid Reactions
Rare cases of angioedema involving the tongue, glottis or larynx have been reported in patients after taking the first or subsequent doses of sedative-hypnotics, including temazepam capsules. Some patients have had additional symptoms such as dyspnea, throat closing, or nausea and vomiting that suggest anaphylaxis. Some patients have required medical therapy in the emergency department. If angioedema involves the tongue, glottis or larynx, airway obstruction may occur and be fatal. Patients who develop angioedema after treatment with temazepam capsules should not be rechallenged with the drug.
-
PRECAUTIONS
General
Since the risk of the development of oversedation, dizziness, confusion, and/or ataxia increases substantially with larger doses of benzodiazepines in elderly and debilitated patients, 7.5 mg of temazepam capsules is recommended as the initial dosage for such patients.
Temazepam capsules should be administered with caution in severely depressed patients or those in whom there is any evidence of latent depression; it should be recognized that suicidal tendencies may be present and protective measures may be necessary.
The usual precautions should be observed in patients with impaired renal or hepatic function and in patients with chronic pulmonary insufficiency.
If temazepam capsules are to be combined with other drugs having known hypnotic properties or CNS-depressant effects, consideration should be given to potential additive effects.
The possibility of a synergistic effect exists with the co-administration of temazepam capsules and diphenhydramine. One case of stillbirth at term has been reported 8 hours after a pregnant patient received temazepam capsules and diphenhydramine. A cause and effect relationship has not yet been determined ( see CONTRAINDICATIONS).
Information for Patients
The text of a patient Medication Guide is printed at the end of this insert. To assure safe and effective use of temazepam capsules, the information and instructions provided in this patient Medication Guide should be discussed with patients.
Special Concerns
"Sleep-Driving" and Other Complex Behaviors – There have been reports of people getting out of bed after taking a sedative-hypnotic and driving their cars while not fully awake, often with no memory of the event. If a patient experiences such an episode, it should be reported to his or her doctor immediately, since "sleep-driving" can be dangerous. This behavior is more likely to occur when temazepam capsules are taken with alcohol or other central nervous system depressants ( see WARNINGS). Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with sleep-driving, patients usually do not remember these events.
Laboratory Tests
The usual precautions should be observed in patients with impaired renal or hepatic function and in patients with chronic pulmonary insufficiency. Abnormal liver function tests as well as blood dyscrasias have been reported with benzodiazepines.
Drug Interactions
The pharmacokinetic profile of temazepam does not appear to be altered by orally administered cimetidine dosed according to labeling.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies were conducted in rats at dietary temazepam doses up to 160 mg/kg/day for 24 months and in mice at dietary doses of 160 mg/kg/day for 18 months. No evidence of carcinogenicity was observed although hyperplastic liver nodules were observed in female mice exposed to the highest dose. The clinical significance of this finding is not known.
Fertility in male and female rats was not adversely affected by temazepam capsules.
No mutagenicity tests have been done with temazepam.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when temazepam capsules are administered to a nursing woman.
Geriatric Use
Clinical studies of temazepam capsules did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in response between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy commonly observed in this population. Temazepam capsules 7.5 mg is recommended as the initial dosage for patients aged 65 and over since the risk of the development of oversedation, dizziness, confusion, ataxia and/or falls increases substantially with larger doses of benzodiazepines in elderly and debilitated patients.
-
ADVERSE REACTIONS
During controlled clinical studies in which 1076 patients received temazepam capsules at bedtime, the drug was well tolerated. Side effects were usually mild and transient. Adverse reactions occurring in 1% or more of patients are presented in the following table:
Temazepam capsules Placebo % Incidence % Incidence (n= 1076) (n=783) Drowsiness 9.1 5.6 Headache 8.5 9.1 Fatigue 4.8 4.7 Nervousness 4.6 8.2 Lethargy 4.5 3.4 Dizziness 4.5 3.3 Nausea 3.1 3.8 Hangover 2.5 1.1 Anxiety 2.0 1.5 Depression 1.7 1.8 Dry Mouth 1.7 2.2 Diarrhea 1.7 1.1 Abdominal Discomfort 1.5 1.9 Euphoria 1.5 0.4 Weakness 1.4 0.9 Confusion 1.3 0.5 Blurred Vision 1.3 1.3 Nightmares 1.2 1.7 Vertigo 1.2 0.8 The following adverse events have been reported less frequently (0.5% to 0.9%):
Central Nervous System – anorexia, ataxia, equilibrium loss, tremor, increased dreaming
Cardiovascular – dyspnea, palpitations
Gastrointestinal – vomiting
Musculoskeletal – backache
Special Senses – hyperhidrosis, burning eyes
Amnesia, hallucinations, horizontal nystagmus, and paradoxical reactions including restlessness, overstimulation and agitation were rare (less than 0.5%).
To report SUSPECTED ADVERSE REACTIONS contact AvKARE, Inc. at 1-855-361-3993; email drugsafety@avkare.com; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DRUG ABUSE AND DEPENDENCE
Abuse and addiction are separate and distinct from physical dependence and tolerance. Abuse is characterized by misuse of the drug for non-medical purposes, often in combination with other psychoactive substances. Physical dependence is a state of adaptation that is manifested by a specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug and/or administration of an antagonist. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug's effects over time. Tolerance may occur to both the desired and undesired effects of drugs and may develop at different rates for different effects.
Addiction is a primary, chronic, neurobiological disease with genetic, psychosocial, and environmental factors influencing its development and manifestations. It is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, utilizing a multidisciplinary approach, but relapse is common.
Abuse and Dependence
Withdrawal symptoms, similar in character to those noted with barbiturates and alcohol (convulsions, tremor, abdominal, and muscle cramps, vomiting, and sweating), have occurred following abrupt discontinuance of benzodiazepines. The more severe withdrawal symptoms have usually been limited to those patients who received excessive doses over an extended period of time. Generally milder withdrawal symptoms (e.g., dysphoria and insomnia) have been reported following abrupt discontinuance of benzodiazepines taken continuously at therapeutic levels for several months. Consequently, after extended therapy at doses higher than 15 mg, abrupt discontinuation should generally be avoided and a gradual dosage tapering schedule followed. As with any hypnotic, caution must be exercised in administering temazepam capsules to individuals known to be addiction-prone or to those whose history suggests they may increase the dosage on their own initiative. It is desirable to limit repeated prescriptions without adequate medical supervision.
-
OVERDOSAGE
Manifestations of acute overdosage of temazepam capsules can be expected to reflect the CNS effects of the drug and include somnolence, confusion, and coma, with reduced or absent reflexes, respiratory depression, and hypotension. The oral LD 50 of temazepam capsules was 1963 mg/kg in mice, 1833 mg/kg in rats, and >2400 mg/kg in rabbits.
Treatment
If the patient is conscious, vomiting should be induced mechanically or with emetics. Gastric lavage should be employed utilizing concurrently a cuffed endotracheal tube if the patient is unconscious to prevent aspiration and pulmonary complications. Maintenance of adequate pulmonary ventilation is essential. The use of pressor agents intravenously may be necessary to combat hypotension. Fluids should be administered intravenously to encourage diuresis. The value of dialysis has not been determined. If excitation occurs, barbiturates should not be used. It should be borne in mind that multiple agents may have been ingested. Flumazenil (Romazicon ®) 1, a specific benzodiazepine receptor antagonist, is indicated for the complete or partial reversal of the sedative effects of benzodiazepines and may be used in situations when an overdose with a benzodiazepine is known or suspected. Prior to the administration of flumazenil, necessary measures should be instituted to secure airway, ventilation, and intravenous access. Flumazenil is intended as an adjunct to, not as a substitute for, proper management of benzodiazepine overdose. Patients treated with flumazenil should be monitored for re-sedation, respiratory depression, and other residual benzodiazepine effects for an appropriate period after treatment. The prescriber should be aware of a risk of seizure in association with flumazenil treatment, particularly in long-term benzodiazepine users and in cyclic antidepressant overdose. The complete flumazenil package insert including CONTRAINDICATIONS, WARNINGS, and PRECAUTIONS should be consulted prior to use.
Up-to-date information about the treatment of overdose can often be obtained from a certified Regional Poison Control Center. Telephone numbers of certified Regional Poison Control Centers are listed in the Physicians' Desk Reference ®2.
- 1 Romazicon is the registered trademark of Hoffman-LaRoche Inc.
- 2 Trademark of Medical Economics Company, Inc.
-
DOSAGE AND ADMINISTRATION
While the recommended usual adult dose is 15 mg before retiring, 7.5 mg may be sufficient for some patients, and others may need 30 mg. In transient insomnia, a 7.5 mg dose may be sufficient to improve sleep latency. In elderly or debilitated patients, it is recommended that therapy be initiated with 7.5 mg until individual responses are determined.
-
HOW SUPPLIED
Temazepam capsules USP
7.5 mg
White opaque/turquoise opaque capsules imprinted "Mutual 866".
Bottles of 100 NDC: 42291-797-01 15 mg
White opaque/green opaque capsules imprinted "Mutual 867".
22.5 mg
White opaque/light green opaque capsules imprinted "Mutual 868".
30 mg
White opaque/white opaque capsules imprinted "Mutual 869".
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
TEMAZEPAM CAPSULES USP C-IV
Read the Medication Guide that comes with temazepam capsules before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your doctor about your medical condition or treatment.
What is the most important information I should know about temazepam capsules?
After taking temazepam capsules, you may get up out of bed while not being fully awake and do an activity that you do not know you are doing. The next morning, you may not remember that you did anything during the night. You have a higher chance for doing these activities if you drink alcohol or take other medicines that make you sleepy with temazepam capsules. Reported activities include:
- driving a car ("sleep-driving")
- making and eating food
- talking on the phone
- having sex
- sleep-walking
Call your doctor right away if you find out that you have done any of the above activities after taking temazepam capsules.
Important:
1. Take temazepam capsules exactly as prescribed
- Do not take more temazepam capsules than prescribed.
- Take temazepam capsules right before you get in bed, not sooner.
2. Do not take temazepam capsules if you:
- drink alcohol
- take other medicines that can make you sleepy. Talk to your doctor about all of your medicines. Your doctor will tell you if you can take temazepam capsules with your other medicines
- cannot get a full night's sleep
What are temazepam capsules?
Temazepam capsules are a sedative-hypnotic (sleep) medicine. Temazepam capsules are used in adults for the short-term (usually 7 to 10 days) treatment of a sleep problem called insomnia. Symptoms of insomnia include:
- trouble falling asleep
- waking up often during the night
Temazepam capsules are not for children.
Temazepam capsules are a federally controlled substance (C-IV) because it can be abused or lead to dependence. Keep temazepam capsules in a safe place to prevent misuse and abuse. Selling or giving away temazepam capsules may harm others, and is against the law. Tell your doctor if you have ever abused or been dependent on alcohol, prescription medicines or street drugs. Who should not take temazepam capsules?
Do not take temazepam capsules if you are pregnant or planning to become pregnant. Temazepam capsules may cause birth defects or harm a fetus (unborn baby).
Temazepam capsules may not be right for you. Before starting temazepam capsules, tell your doctor about all of your health conditions, including if you:
- have a history of depression, mental illness, or suicidal thoughts
- have a history of drug or alcohol abuse or addiction
- have kidney or liver disease
- have a lung disease or breathing problems
- are breastfeeding
Tell your doctor about all of the medicines you take including prescription and nonprescription medicines, vitamins and herbal supplements. Medicines can interact with each other, sometimes causing serious side effects. Do not take temazepam capsules with other medicines that can make you sleepy.
Know the medicines you take. Keep a list of your medicines with you to show your doctor and pharmacist each time you get a new medicine.
How should I take temazepam capsules?
- Take temazepam capsules exactly as prescribed. Do not take more temazepam capsules than prescribed for you.
- Take temazepam capsules right before you get into bed.
- Do not take temazepam capsules unless you are able to get a full night's sleep before you must be active again.
- Call your doctor if your insomnia worsens or is not better within 7 to 10 days. This may mean that there is another condition causing your sleep problems.
- If you take too many temazepam capsules or overdose, call your doctor or poison control center right away, or get emergency treatment.
What are the possible side effects of temazepam capsules?
Possible serious side effects of temazepam capsules include:
- getting out of bed while not being fully awake and do an activity that you do not know you are doing. (See " What is the most important information I should know about temazepam capsules?")
- abnormal thoughts and behavior. Symptoms include more outgoing or aggressive behavior than normal, confusion, agitation, hallucinations, worsening of depression, and suicidal thoughts.
- memory loss
- anxiety
- severe allergic reactions. Symptoms include swelling of the tongue or throat, trouble breathing, and nausea and vomiting. Get emergency medical help if you get these symptoms after taking temazepam capsules.
Call your doctor right away if you have any of the above side effects or any other side effects that worry you while using temazepam capsules.
The most common side effects of temazepam capsules are:
- drowsiness
- headache
- tiredness
- nervousness
- dizziness
- nausea
- "hangover" feeling the day after taking temazepam capsules
- You may still feel drowsy the next day after taking temazepam capsules. Do not drive or do other dangerous activities after taking temazepam capsules until you feel fully awake.
- You may have withdrawal symptoms if you stop taking temazepam capsules suddenly. Withdrawal symptoms can be serious and include seizures. Mild withdrawal symptoms include a depressed mood and trouble sleeping. Talk to your doctor to check if you need to stop temazepam capsules slowly.
These are not all the side effects of temazepam capsules. Ask your doctor or pharmacist for more information.
How should I store temazepam capsules?
- Store temazepam capsules at room temperature, 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]
- Keep temazepam capsules and all medicines out of reach of children.
General Information about temazepam capsules
- Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide.
- Do not use temazepam capsules for a condition for which it was not prescribed.
- Do not share temazepam capsules with other people, even if you think they have the same symptoms that you have. It may harm them and it is against the law.
This Medication Guide summarizes the most important information about temazepam capsules. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about temazepam capsules that is written for healthcare professionals.
What are the ingredients in temazepam capsules?
Active Ingredient: temazepam USP
Inactive Ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate. The capsule shell contains gelatin and titanium dioxide. The capsule shell for the 7.5 mg also contains FD&C Blue #1 and FD&C Red #40; the capsule shell for 15 mg and 22.5 mg also contains D&C Yellow #10, FD&C Blue #1 and FD&C Yellow #6. The imprinting ink contains: D&C Yellow #10, FD&C Blue #1, FD&C Blue #2, FD&C Red #40, iron oxide black and propylene glycol.
Rx only
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
- SPL UNCLASSIFIED SECTION
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
AvKARE
NDC 42291-797-01
Temazepam Capsules USP CIV
7.5mg
100 Capsules Rx Only
PHARMACIST: PLEASE DISPENSE WITH MEDICATION GUIDE ATTACHED.
Each Capsule Contains:
Temazepam, USP ........................ 7.5 mg
USUAL DOSAGE: Read package outsert for full prescribing information.Store at 20º to 25ºC (68º to 77ºF). [See USP Controlled Room Temperature.]
DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER.
Capsule Imprinted: Mutual 866Keep out of the reach of children.
Manufactured for:
AvKARE, Inc.
Pulaski, TN 38478
Mfg. Rev. 02, 07/14 AV 08/15 (P)N3 42291 79701 7

-
INGREDIENTS AND APPEARANCE
TEMAZEPAM
temazepam capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 42291-797(NDC:53489-648) Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMAZEPAM (UNII: CHB1QD2QSS) (TEMAZEPAM - UNII:CHB1QD2QSS) TEMAZEPAM 7.5 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) FERROSOFERRIC OXIDE (UNII: XM0M87F357) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white (opaque) , turquoise (opaque) Score no score Shape CAPSULE Size 14mm Flavor Imprint Code Mutual;866 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 42291-797-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 08/25/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078581 08/25/2015 Labeler - AvKARE, Inc. (796560394)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.