BUPROPION HYDROCHLORIDE tablet, film coated
Bupropion Hydrochloride by
Drug Labeling and Warnings
Bupropion Hydrochloride by is a Prescription medication manufactured, distributed, or labeled by Cadila Pharmaceuticals Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use BUPROPION HYDROCHLORIDE tablets safely and effectively. See full prescribing information for BUPROPION HYDROCHLORIDE tablets.
BUPROPION HYDROCHLORIDE tablets, for oral use
Initial U.S. Approval: 1985
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
See full prescribing information for complete boxed warning.
Increased risk of suicidal thinking and behavior in children, adolescents, and young adults taking antidepressants. (5.1)
Monitor for worsening and emergence of suicidal thoughts and behaviors. (5.1)INDICATIONS AND USAGE
Bupropion hydrochloride tablets are an aminoketone antidepressant, indicated for the treatment of major depressive disorder (MDD). (1)
DOSAGE AND ADMINISTRATION
- Starting dose: 200 mg/day given as 100 mg twice daily (2.1)
- General: Increase dose gradually to reduce seizure risk. (2.1, 5.3)
- After 3 days, may increase the dose to 300 mg/day, given as 100 mg 3 times daily at an interval of at least 6 hours between doses. (2.1)
- Usual target dose: 300 mg/day as 100 mg 3 times daily. (2.1)
- Maximum dose: 450 mg/day given as 150 mg 3 times daily. (2.1)
- Periodically reassess the dose and need for maintenance treatment. (2.1)
- Moderate to severe hepatic impairment: 75 mg once daily. (2.2, 8.7)
- Mild hepatic impairment: Consider reducing the dose and/or frequency of dosing. (2.2, 8.7)
- Renal impairment: Consider reducing the dose and/or frequency. (2.3, 8.6)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
- Seizure disorder. (4, 5.3)
- Current or prior diagnosis of bulimia or anorexia nervosa. (4, 5.3)
- Abrupt discontinuation of alcohol, benzodiazepines, barbiturates, antiepileptic drugs. (4, 5.3)
- Monoamine Oxidase Inhibitors (MAOIs): Do not use MAOIs intended to treat psychiatric disorders with bupropion hydrochloride tablets or within 14 days of stopping treatment with bupropion hydrochloride tablets. Do not use bupropion hydrochloride tablets within 14 days of stopping an MAOI intended to treat psychiatric disorders. In addition, do not start bupropion hydrochloride tablets in a patient who is being treated with linezolid or intravenous methylene blue. (4, 7.6)
- Known hypersensitivity to bupropion or other ingredients of bupropion hydrochloride tablets. (4, 5.8)
WARNINGS AND PRECAUTIONS
- Neuropsychiatric adverse events during smoking cessation: Postmarketing reports of serious or clinically significant neuropsychiatric adverse events have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Observe patients attempting to quit smoking with bupropion for the occurrence of such symptoms and instruct them to discontinue bupropion and contact a healthcare provider if they experience such adverse events. (5.2)
- Seizure risk: The risk is dose-related. Can minimize risk by gradually increasing the dose and limiting daily dose to 450 mg. Discontinue if seizure occurs. (4, 5.3, 7.3)
- Hypertension: Bupropion hydrochloride tablets can increase blood pressure. Monitor blood pressure before initiating treatment and periodically during treatment. (5.4)
- Activation of mania/hypomania: Screen patients for bipolar disorder and monitor for these symptoms. (5.5)
- Psychosis and other neuropsychiatric reactions: Instruct patients to contact a healthcare professional if such reactions occur. (5.6)
- Angle-closure glaucoma: Angle-closure glaucoma has occurred in patients with untreated anatomically narrow angles treated with antidepressants. (5.7)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥5% and ≥1% more than placebo rate) are: agitation, dry mouth, constipation, headache /migraine, nausea/vomiting, dizziness, excessive sweating, tremor, insomnia, blurred vision, tachycardia, confusion, rash, hostility, cardiac arrhythmias, and auditory disturbance. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Cadila Pharmaceuticals Limited at 1-202-355-9785 (Fax 1-202-355-9784) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatchDRUG INTERACTIONS
- CYP2B6 inducers: Dose increase may be necessary if coadministered with CYP2B6 inducers (e.g., ritonavir, lopinavir, efavirenz, carbamazepine, phenobarbital, and phenytoin) based on clinical response, but should not exceed the maximum recommended dose. (7.1)
- Drugs metabolized by CYP2D6: Bupropion inhibits CYP2D6 and can increase concentrations of: antidepressants (e.g., venlafaxine, nortriptyline, imipramine, desipramine, paroxetine, fluoxetine, sertraline), antipsychotics (e.g., haloperidol, risperidone, thioridazine), beta-blockers (e.g., metoprolol), and Type 1C antiarrhythmics (e.g., propafenone, flecainide). Consider dose reduction when using with bupropion. (7.2)
- Digoxin: May decrease plasma digoxin levels. Monitor digoxin levels. (7.2)
- Drugs that lower seizure threshold: Dose bupropion hydrochloride tablets with caution. (5.3, 7.3)
- Dopaminergic drugs (levodopa and amantadine): CNS toxicity can occur when used concomitantly with bupropion hydrochloride tablets. (7.4)
- MAOIs: Increased risk of hypertensive reactions can occur when used concomitantly with bupropion hydrochloride tablets. (7.6)
- Drug-laboratory test interactions: Bupropion hydrochloride tablets can cause false-positive urine test results for amphetamines. (7.7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 General Instruction for Use
2.2 Dose Adjustment in Patients with Hepatic Impairment
2.3 Dose Adjustment in Patients with Renal Impairment
2.4 Switching a Patient to or from a Monoamine Oxidase Inhibitor (MAOI) Antidepressant
2.5 Use of Bupropion Hydrochloride Tablets with Reversible MAOIs Such as Linezolid or Methylene Blue
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNING AND PRECAUTIONS
5.1 Suicidal Thoughts and Behaviors in Children, Adolescents, and Young Adults
5.2 Neuropsychiatric Adverse Events and Suicide Risk in Smoking Cessation Treatment
5.3 Seizure
5.4 Hypertension
5.5 Activation of Mania/Hypomania
5.6 Psychosis and Other Neuropsychiatric Reactions
5.7 Angle-Closure Glaucoma
5.8 Hypersensitivity Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect Bupropion Hydrochloride Tablets
7.2 Potential for Bupropion Hydrochloride Tablets to Affect Other Drugs
7.3 Drugs that Lower Seizure Threshold
7.4 Dopaminergic Drugs (Levodopa and Amantadine)
7.5 Use With Alcohol
7.6 MAO Inhibitors
7.7 Drug -Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
10 OVERDOSAGE
10.1 Human Overdose Experience
10.2 Overdosage Management
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis,Impairment Of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
SUICIDALITY AND ANTIDEPRESSANT DRUGS
Antidepressants increased the risk of suicidal thoughts and behavior in children,
adolescents, and young adults in short-term trials. These trials did not show an increase in the risk of suicidal thoughts and behavior with antidepressant use in subjects over age 24; there was a reduction in risk with antidepressant use in subjects aged 65 and older [seeWarnings and Precautions (5.1)].
In patients of all ages who are started on antidepressant therapy, monitor closely for worsening, and for emergence of suicidal thoughts and behaviors. Advise families and caregivers of the need for close observation and communication with the prescriber [seeWarnings and Precautions (5.1)].
-
1 INDICATIONS AND USAGE
Bupropion hydrochloride tablets are indicated for the treatment of major depressive disorder (MDD), as defined by the Diagnostic and Statistical Manual (DSM).
The efficacy of bupropion hydrochloride tablets in the treatment of a major depressive episode was established in two 4-week controlled inpatient trials and one 6-week controlled outpatient trial of adult subjects with MDD [see Clinical Studies (14)].
-
2 DOSAGE AND ADMINISTRATION
2.1 General Instruction for Use
To minimize the risk of seizure, increase the dose gradually [see Warnings and Precautions (5.3)]. Increases in dose should not exceed 100 mg/day in a 3-day period. Bupropion hydrochloride tablets should be swallowed whole and not crushed, divided, or chewed. Bupropion hydrochloride tablets may be taken with or without food.
The recommended starting dose is 200 mg/day, given as 100 mg twice daily. After 3 days of dosing, the dose may be increased to 300 mg/day, given as 100 mg 3 times daily, with at least 6 hours between successive doses. Dosing above 300 mg/day may be accomplished using the 75- or 100-mg tablets.
A maximum of 450 mg/day, given in divided doses of not more than 150 mg each, may be considered for patients who show no clinical improvement after several weeks of treatment at 300 mg/day. Administer the 100-mg tablet 4 times daily to not exceed the limit of 150 mg in a single dose.
It is generally agreed that acute episodes of depression require several months or longer of antidepressant drug treatment beyond the response in the acute episode. It is unknown whether the dose of bupropion hydrochloride tablets needed for maintenance treatment is identical to the dose that provided an initial response. Periodically reassess the need for maintenance treatment and the appropriate dose for such treatment.
2.2 Dose Adjustment in Patients with Hepatic Impairment
In patients with moderate to severe hepatic impairment (Child-Pugh score: 7 to 15), the maximum dose of bupropion hydrochloride tablets is 75 mg/day. In patients with mild hepatic impairment (Child-Pugh score: 5 to 6), consider reducing the dose and/or frequency of dosing [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.3 Dose Adjustment in Patients with Renal Impairment
Consider reducing the dose and/or frequency of bupropion hydrochloride tablets in patients with renal impairment (Glomerular Filtration Rate Less than 90 mL/ min) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.4 Switching a Patient to or from a Monoamine Oxidase Inhibitor (MAOI) Antidepressant
At least 14 days should elapse between discontinuation of an MAOI intended to treat depression and initiation of therapy with bupropion hydrochloride tablets. Conversely, at least 14 days should be allowed after stopping bupropion hydrochloride tablets before starting an MAOI antidepressant [see Contraindications (4)and Drug Interactions (7.6)].
2.5 Use of Bupropion Hydrochloride Tablets with Reversible MAOIs Such as Linezolid or Methylene Blue
Do not start bupropion hydrochloride tablets in a patient who is being treated with a reversible MAOI such as Linezolid or intravenous methylene blue. Drug interactions can increase the risk of hypertensive reactions. In a patient who requires more urgent treatment of a psychiatric condition, non-pharmacological interventions, including hospitalization, should be considered [see Contraindications (4), Drug Interactions (7.6)].
In some cases, a patient already receiving therapy with bupropion hydrochloride tablets may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of hypertensive reactions in a particular patient, bupropion hydrochloride tablets should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for 2 weeks or until 24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with bupropion hydrochloride tablets may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue.
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with bupropion hydrochloride tablets is unclear. The clinician should, nevertheless, be aware of the possibility of a drug interaction with such use [see Contraindications (4), Drug Interactions (7.6)]. - 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Bupropion hydrochloride tablets are contraindicated in patients with a seizure disorder.
Bupropion hydrochloride tablets are contraindicated in patients with a current or prior diagnosis of bulimia or anorexia nervosa as a higher incidence of seizures was observed in such patients treated with bupropion hydrochloride tablets [see Warnings and Precautions (5.3)].
Bupropion hydrochloride tablets are contraindicated in patients undergoing abrupt discontinuation of alcohol, benzodiazepines, barbiturates, and antiepileptic drugs [see Warnings and Precautions (5.3), Drug Interactions (7.3)].
The use of MAOIs (intended to treat psychiatric disorders) concomitantly with bupropion hydrochloride tablets or within 14 days of discontinuing treatment with bupropion hydrochloride tablets are contraindicated. There is an increased risk of hypertensive reactions when bupropion hydrochloride tablets are used concomitantly with MAOIs. The use of bupropion hydrochloride tablets within 14 days of discontinuing treatment with an MAOI is also contraindicated. Starting bupropion hydrochloride tablets in a patient treated with reversible MAOIs such as linezolid or intravenous methylene blue is contraindicated [see Dosage and Administration (2.4, 2.5), Warnings and Precautions (5.4), Drug Interactions (7.6)].
Bupropion hydrochloride tablets are contraindicated in patients with known hypersensitivity to bupropion or other ingredients of bupropion hydrochloride tablets. Anaphylactoid/anaphylactic reactions and Stevens-Johnson syndrome have been reported [see Warnings and Precautions (5.8)].
-
5 WARNING AND PRECAUTIONS
5.1 Suicidal Thoughts and Behaviors in Children, Adolescents, and Young Adults
Patients with MDD, both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment.
Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (selective serotonin reuptake inhibitors [SSRIs] and others) show that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18 to 24 with MDD and other psychiatric disorders. Short-term clinical trials did not show an increase in the risk of suicidality with antidepressants compared with placebo in adults beyond age 24; there was a reduction with antidepressants compared with placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4,400 subjects. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 subjects. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger subjects for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs. placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1,000 subjects treated) are provided in Table 1.
Table 1. Risk Differences in the Number of Suicidality Cases by Age Group in the Pooled Placebo-Controlled Trials of Antidepressants in Pediatric and Adult Subjects
Age Range
Drug-Placebo Difference in Number of Cases of Suicidality per 1,000 Subjects Treated
Increases Compared with Placebo
<18
14 additional cases
18 to 24
5 additional cases
Decreases Compared with Placebo
25 to 64
1 fewer case
≥65
6 fewer cases
No suicides occurred in any of the pediatric trials. There were suicides in the adult trials, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer-term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes , either increases or decreases [see Boxed Warning].The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse, or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset, or were not part of the patient’s presenting symptoms.
Families and caregivers of patients being treated with antidepressants for MDD or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to healthcare providers. Such monitoring should include daily observation by families and caregivers. Prescriptions for bupropion hydrochloride tablets should be written for the smallest quantity of tablets consistent with good patient management, in order to reduce the risk of overdose.
5.2 Neuropsychiatric Adverse Events and Suicide Risk in Smoking Cessation Treatment
Bupropion hydrochloride tablets are not approved for smoking cessation treatment; however, it contains the same active ingredient as the smoking cessation medication ZYBAN. Serious neuropsychiatric adverse events have been reported in patients taking bupropion for smoking cessation. These postmarketing reports have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide [see Adverse Reactions (6.2)]. Some patients who stopped smoking may have been experiencing symptoms of nicotine withdrawal, including depressed mood. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these adverse events occurred in patients taking bupropion who continued to smoke.
Neuropsychiatric adverse events occurred in patients without and with pre-existing psychiatric disease; some patients experienced worsening of their psychiatric illnesses. Observe patients for the occurrence of neuropsychiatric adverse events. Advise patients and caregivers that the patient should stop taking bupropion hydrochloride tablets and contact a healthcare provider immediately if agitation, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of bupropion was reported. However, the symptoms persisted in some cases; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.5.3 Seizure
Bupropion hydrochloride tablets can cause seizure. The risk of seizure is dose-related. The dose should not exceed 450 mg/day. Increase the dose gradually. Discontinue Bupropion Hydrochloride Tablets and do not restart treatment if the patient experiences a seizure.
The risk of seizures is also related to patient factors, clinical situations, and concomitant medications that lower the seizure threshold. Consider these risks before initiating treatment with bupropion hydrochloride t ablets. Bupropion hydrochloride tablets are contraindicated in patients with a seizure disorder, current or prior diagnosis of anorexia nervosa or bulimia, or undergoing abrupt discontinuation of alcohol, benzodiazepines, barbiturates, and antiepileptic drugs [see Contraindications (4), Drug Interactions (7.3)]. The following conditions can also increase the risk of seizure: severe head injury; arteriovenous malformation; CNS tumor or CNS infection; severe stroke; concomitant use of other medications that lower the seizure threshold (e.g., other bupropion products, antipsychotics, tricyclic antidepressants, theophylline, and systemic corticosteroids); metabolic disorders (e.g., hypoglycemia, hyponatremia, severe hepatic impairment, and hypoxia); use of illicit drugs (e.g., cocaine); or abuse or misuse of prescription drugs such as CNS stimulants. Additional predisposing conditions include diabetes mellitus treated with oral hypoglycemic drugs or insulin; use of anorectic drugs; and excessive use of alcohol, benzodiazepines, sedative/hypnotics, or opiates.
Incidence of Seizure with Bupropion Use:
Bupropion is associated with seizures in approximately 0.4% (4/1,000) of patients treated at doses up to 450 mg/day. The estimated seizure incidence for bupropion hydrochloride tablets increases almost 10-fold between 450 and 600 mg/day.
The risk of seizure can be reduced if the dose of bupropion hydrochloride tablets does not exceed 450 mg/day, given as 150 mg 3 times daily, and the titration rate is gradual.5.4 Hypertension
Treatment with bupropion hydrochloride tablets can result in elevated blood pressure and hypertension. Assess blood pressure before initiating treatment with bupropion hydrochloride tablets, and monitor periodically during treatment. The risk of hypertension is increased if bupropion hydrochloride tablets are used concomitantly with MAOIs or other drugs that increase dopaminergic or noradrenergic activity [see Contraindications (4)].
Data from a comparative trial of the sustained-release formulation of bupropion HCl, nicotine transdermal system (NTS), the combination of sustained-release bupropion plus NTS, and placebo as an aid to smoking cessation suggest a higher incidence of treatment-emergent hypertension in patients treated with the combination of sustained-release bupropion and NTS. In this trial, 6.1% of subjects treated with the combination of sustained-release bupropion and NTS had treatment-emergent hypertension compared with 2.5%, 1.6%, and 3.1% of subjects treated with sustained-release bupropion, NTS, and placebo, respectively. The majority of these subjects had evidence of pre-existing hypertension. Three subjects (1.2%) treated with the combination of sustained-release bupropion and NTS and 1 subject (0.4%) treated with NTS had study medication discontinued due to hypertension compared with none of the subjects treated with sustained-release bupropion or placebo. Monitoring of blood pressure is recommended in patients who receive the combination of bupropion and nicotine replacement.
In a clinical trial of bupropion immediate-release in MDD subjects with stable congestive heart failure (N = 36), bupropion was associated with an exacerbation of pre-existing hypertension in 2 subjects, leading to discontinuation of bupropion treatment. There are no controlled trials assessing the safety of bupropion in patients with a recent history of myocardial infarction or unstable cardiac disease.5.5 Activation of Mania/Hypomania
Antidepressant treatment can precipitate a manic, mixed, or hypomanic manic episode. The risk appears to be increased in patients with bipolar disorder or who have risk factors for bipolar disorder. Prior to initiating bupropion hydrochloride tablets, screen patients for a history of bipolar disorder and the presence of risk factors for bipolar disorder (e.g., family history of bipolar disorder, suicide, or depression). Bupropion hydrochloride tablets are not approved for use in treating bipolar depression.
5.6 Psychosis and Other Neuropsychiatric Reactions
Depressed patients treated with bupropion hydrochloride tablets have had a variety of neuropsychiatric signs and symptoms, including delusions, hallucinations, psychosis, concentration disturbance, paranoia, and confusion. Some of these patients had a diagnosis of bipolar disorder. In some cases, these symptoms abated upon dose reduction and/or withdrawal of treatment. Instruct patients to contact a healthcare professional if such reactions occur.
5.7 Angle-Closure Glaucoma
The pupillary dilation that occurs following use of many antidepressant drugs including bupropion hydrochloride tablets may trigger an angle-closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy.
5.8 Hypersensitivity Reactions
Anaphylactoid/anaphylactic reactions have occurred during clinical trials with bupropion. Reactions have been characterized by pruritus, urticaria, angioedema, and dyspnea requiring medical treatment. In addition, there have been rare, spontaneous postmarketing reports of erythema multiforme, Stevens-Johnson syndrome, and anaphylactic shock associated with bupropion. Instruct patients to discontinue bupropion hydrochloride tablets and consult a healthcare provider if they develop an allergic or anaphylactoid/anaphylactic reaction (e.g., skin rash, pruritus, hives, chest pain, edema, and shortness of breath) during treatment.
There are reports of arthralgia, myalgia, fever with rash and other serum sickness-like symptoms suggestive of delayed hypersensitivity.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling:
Suicidal thoughts and behaviors in adolescents and young adults [see Boxed Warning,Warnings and Precautions (5.1)]Neuropsychiatric symptoms and suicide risk in smoking cessation treatment [see Warnings and Precautions (5.2)]
Seizure [see Warnings and Precautions (5.3)]
Hypertension [ see Warnings and Precautions (5.4) ]
Activation of mania or hypomania [see Warnings and Precautions (5.5)]
Psychosis and other neuropsychiatric reactions [see Warnings and Precautions (5.6)]
Angle-closure glaucoma [see Warnings and Precautions (5.7)]
Hypersensitivity reactions [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Adverse Reactions Leading to Discontinuation of Treatment
Adverse reactions were sufficiently troublesome to cause discontinuation of treatment with bupropion hydrochloride tablets in approximately 10% of the 2,400 subjects and healthy volunteers who participated in clinical trials during the product’s initial development. The more common events causing discontinuation include neuropsychiatric disturbances (3.0%), primarily agitation and abnormalities in mental status; gastrointestinal disturbances (2.1%), primarily nausea and vomiting; neurological disturbances (1.7%), primarily seizures, headaches, and sleep disturbances; and dermatologic problems (1.4%), primarily rashes. It is important to note, however, that many of these events occurred at doses that exceed the recommended daily dose.
Commonly Observed Adverse Reactions
Adverse reactions commonly encountered in subjects treated with bupropion hydrochloride tablets are agitation, dry mouth, insomnia, headache/migraine, nausea/vomiting, constipation, tremor, dizziness, excessive sweating, blurred vision, tachycardia, confusion, rash, hostility, cardiac arrhythmia, and auditory disturbance.
Table 2 summarizes the adverse reactions that occurred in placebo-controlled trials at an incidence of at least 1% of subjects receiving bupropion hydrochloride tablets and more frequently in these subjects than in the placebo group.
Table 2. Adverse Reactions Reported by at Least 1% of Subjects and at a Greater Frequency than Placebo in Controlled Clinical Trials
Adverse Reaction
Bupropion
(n=323)
%
Placebo
(n=185)
%
Cardiovascular
Cardiac arrhythmias
5.3
4.3
Dizziness
22.3
16.2
Hypertension
4.3
1.6
Hypotension
2.5
2.2
Palpitations
3.7
2.2
Syncope
1.2
0.5
Tachycardia
10.8
8.6
Dermatologic
Pruritus
2.2
0.0
Rash
8.0
6.5
Gastrointestinal
Appetite increase
3.7
2.2
Constipation
26.0
17.3
Dyspepsia
3.1
2.2
Nausea/vomiting
22.9
18.9
Genitourinary
Impotence
3.4
3.1
Menstrual complaints
4.7
1.1
Urinary frequency
2.5
2.2
Musculoskeletal
Arthritis
3.1
2.7
Neurological
Akathisia
1.5
1.1
Cutaneous temperature disturbance
1.9
1.6
Dry mouth
27.6
18.4
Excessive sweating
22.3
14.6
Headache/Migraine
25.7
22.2
Impaired sleep quality
4.0
1.6
Insomia
18.6
15.7
Sedation
19.8
19.5
Sensory disturbance
4.0
3.2
Tremor
21.1
7.6
Neuropsychiatric
Agitation
31.9
22.2
Anxiety
3.1
1.1
Confusion
8.4
4.9
Decreased libido
3.1
1.6
Delusion
1.2
1.1
Euphoria
1.2
0.5
Hostility
5.6
3.8
Nonspecific
Fever/chills
1.2
0.5
Special Senses
Auditory disturbance
5.3
3.2
Blurred vision
14.6
10.3
Gustatory disturbance
3.1
1.1
Other Adverse Reactions Observed During the Clinical Development of Bupropion Hydrochloride Tablets
The conditions and duration of exposure to bupropion hydrochloride tablets varied greatly, and a substantial proportion of the experience was gained in open and uncontrolled clinical settings. During this experience, numerous adverse events were reported; however, without appropriate controls, it is impossible to determine with certainty which events were or were not caused by bupropion hydrochloride tablets. The following enumeration is organized by organ system and describes events in terms of their relative frequency of reporting in the database.
The following definitions of frequency are used: Frequent adverse reactions are defined as those occurring in at least 1/100 subjects. Infrequent adverse reactions are those occurring in 1/100 to 1/1,000 subjects, while rare events are those occurring in less than 1/1,000 subjects.
Cardiovascular: Frequent was edema; infrequent were chest pain, electrocardiogram (ECG) abnormalities (premature beats and nonspecific ST-T changes), and shortness of breath/dyspnea; rare were flushing, and myocardial infarction.Dermatologic: Infrequent was alopecia.
Endocrine: Infrequent was gynecomastia; rare was glycosuria.
Gastrointestinal: Infrequent were dysphagia, thirst disturbance, and liver damage/jaundice; rare was intestinal perforation.
Genitourinary: Frequent was nocturia; infrequent were vaginal irritation, testicular swelling, urinary tract infection, painful erection, and retarded ejaculation; rare were enuresis, and urinary incontinence.
Neurological: Frequent were ataxia/incoordination, seizure, myoclonus, dyskinesia, and dystonia; infrequent were mydriasis, vertigo, and dysarthria; rare were electroencephalogram (EEG) abnormality, and impaired attention.
Neuropsychiatric: Frequent were mania/hypomania, increased libido, hallucinations, decrease in sexual function, and depression; infrequent were memory impairment, depersonalization, psychosis, dysphoria, mood instability, paranoia and formal thought disorder, rare was suicidal ideation.
Oral Complaints: Frequent was stomatitis; infrequent were toothache, bruxism, gum irritation, and oral edema.
Respiratory: Infrequent were bronchitis and shortness of breath/dyspnea; rare was pulmonary embolism.
Special Senses: Infrequent was visual disturbance; rare was diplopia.
Nonspecific: Frequent were flu-like symptoms; infrequent was nonspecific pain; rare was overdose.
Altered Appetite and Weight: A weight loss of greater than 5 lbs. occurred in 28% of subjects receiving bupropion hydrochloride tablets. This incidence is approximately double that seen in comparable subjects treated with tricyclics or placebo. Furthermore, while 35% of subjects receiving tricyclic antidepressants gained weight, only 9.4% of subjects treated with bupropion hydrochloride tablets did. Consequently, if weight loss is a major presenting sign of a patient’s depressive illness, the anorectic and/or weight reducing potential of bupropion hydrochloride tablets should be considered.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of bupropion hydrochloride tablets and are not described elsewhere in the label. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body (General)
Arthralgia, myalgia, and fever with rash and other symptoms suggestive of delayed hypersensitivity. These symptoms may resemble serum sickness [see Warnings and Precautions (5.8)].
Cardiovascular
Hypertension (in some cases severe), orthostatic hypotension, third degree heart block.
Endocrine
Hyponatremia, syndrome of inappropriate antidiuretic hormone secretion, hyperglycemia, hypoglycemia.
Gastrointestinal
Esophagitis, hepatitis.
Hemic and Lymphatic
Ecchymosis, leukocytosis, leukopenia, thrombocytopenia. Altered PT and/or INR, infrequently associated with hemorrhagic or thrombotic complications, were observed when bupropion was coadministered with warfarin.
Musculoskeletal
Muscle rigidity/fever/rhabdomyolysis, muscle weakness.
Nervous System
Aggression, coma, completed suicide, delirium, dream abnormalities, paranoid ideation, paresthesia, parkinsonism, restlessness, suicide attempt, unmasking of tardive dyskinesia. Skin and Appendages
Stevens-Johnson syndrome, angioedema, exfoliative dermatitis, urticaria.
Special Senses
Tinnitus, increased intraocular pressure.
-
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect Bupropion Hydrochloride Tablets
Bupropion is primarily metabolized to hydroxybupropion by CYP2B6. Therefore, the potential exists for drug interactions between bupropion hydrochloride tablets and drugs that are inhibitors or inducers of CYP2B6.
Inhibitors of CYP2B6
Ticlopidine and Clopidogrel: Concomitant treatment with these drugs can increase bupropion exposure but decrease hydroxybupropion exposure. Based on clinical response, dosage adjustment of bupropion hydrochloride tablets may be necessary when coadministered with CYP2B6 inhibitors (e.g., ticlopidine or clopidogrel) [see Clinical Pharmacology (12.3)].Inducers of CYP2B6
Ritonavir, Lopinavir, and Efavirenz: Concomitant treatment with these drugs can decrease bupropion hydrochloride tablets and hydroxybupropion exposure. Dosage increase of bupropion may be necessary when coadministered with ritonavir, lopinavir, or efavirenz [see Clinical Pharmacology (12.3)] but should not exceed the maximum recommended dose.Carbamazepine, Phenobarbital, Phenytoin: While not systematically studied, these drugs may induce the metabolism of bupropion and may decrease bupropion exposure [see Clinical Pharmacology (12.3)]. If bupropion is used concomitantly with a CYP inducer, it may be necessary to increase the dose of bupropion, but the maximum recommended dose should not be exceeded.
7.2 Potential for Bupropion Hydrochloride Tablets to Affect Other Drugs
Drugs Metabolized by CYP2D6
Bupropion and its metabolites (erythrohydrobupropion, threohydrobupropion, hydroxybupropion) are CYP2D6 inhibitors. Therefore, coadministration of bupropion hydrochloride tablets with drugs that are metabolized by CYP2D6 can increase the exposures of drugs that are substrates of CYP2D6. Such drugs include certain antidepressants (e.g., venlafaxine, nortriptyline, imipramine, desipramine, paroxetine, fluoxetine, and sertraline), antipsychotics (e.g., haloperidol, risperidone, thioridazine), beta-blockers (e.g., metoprolol), and Type 1C antiarrhythmics (e.g., propafenone and flecainide). When used concomitantly with bupropion hydrochloride tablets, it may be necessary to decrease the dose of these CYP2D6 substrates, particularly for drugs with a narrow therapeutic index.
Drugs that require metabolic activation by CYP2D6 to be effective (e.g., tamoxifen) theoretically could have reduced efficacy when administered concomitantly with inhibitors of CYP2D6 such as bupropion. Patients treated concomitantly with bupropion hydrochloride tablets and such drugs may require increased doses of the drug [see Clinical Pharmacology (12.3)].
Digoxin
Coadministration of bupropion hydrochloride tablets with digoxin may decrease plasma digoxin levels. Monitor plasma digoxin levels in patients treated concomitantly with bupropion hydrochloride tablets and digoxin [see Clinical Pharmacology (12.3)].7.3 Drugs that Lower Seizure Threshold
Use extreme caution when coadministering bupropion hydrochloride tablets with other drugs that lower seizure threshold (e.g., other bupropion products, antipsychotics, antidepressants, theophylline, or systemic corticosteroids). Use low initial doses and increase the dose gradually [see Contraindications (4), Warnings and Precautions (5.3)].
7.4 Dopaminergic Drugs (Levodopa and Amantadine)
Bupropion, levodopa, and amantadine have dopamine agonist effects. CNS toxicity has been reported when bupropion was coadministered with levodopa or amantadine. Adverse reactions have included restlessness, agitation, tremor, ataxia, gait disturbance, vertigo, and dizziness. It is presumed that the toxicity results from cumulative dopamine agonist effects. Use caution when administering bupropion hydrochloride tablets concomitantly with these drugs.
7.5 Use With Alcohol
In postmarketing experience, there have been rare reports of adverse neuropsychiatric events or reduced alcohol tolerance in patients who were drinking alcohol during treatment with bupropion hydrochloride tablets. The consumption of alcohol during treatment with bupropion hydrochloride tablets should be minimized or avoided.
7.6 MAO Inhibitors
Bupropion inhibits the reuptake of dopamine and norepinephrine. Concomitant use of MAOIs and bupropion is contraindicated because there is an increased risk of hypertensive reactions if bupropion is used concomitantly with MAOIs. Studies in animals demonstrate that the acute toxicity of bupropion is enhanced by the MAO inhibitor phenelzine. At least 14 days should elapse between discontinuation of an MAOI intended to treat depression and initiation of treatment with bupropion hydrochloride tablets. Conversely, at least 14 days should be allowed after stopping bupropion hydrochloride tablets before starting an MAOI antidepressant [see Dosage and Administration (2.4, 2.5), Contraindications (4)].
7.7 Drug -Laboratory Test Interactions
False-positive urine immunoassay screening tests for amphetamines have been reported in patients taking bupropion. This is due to lack of specificity of some screening tests. False-positive test results may result even following discontinuation of bupropion therapy.
Confirmatory tests, such as gas chromatography/mass spectrometry, will distinguish bupropion from amphetamines.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is an independent pregnancy exposure registry that monitors pregnancy outcomes in women exposed to any antidepressants during pregnancy. Healthcare providers are encouraged to register patients by calling the National Pregnancy Registry for Antidepressants at 1-844-405-6185 or visiting online at https://womensmentalhealth.org/clinical-and researchprograms/pregnancyregistry/antidepressants.
Risk Summary
Data from epidemiological studies of pregnant women exposed to bupropion in the first trimester have not identified an increased risk of congenital malformations overall (see Data). There are risks to the mother associated with untreated depression in pregnancy (see Clinical Considerations). When bupropion was administered to pregnant rats during organogenesis, there was no evidence of fetal malformations at doses up to approximately 10 times the maximum recommended human dose (MRHD) of 450 mg/day. When given to pregnant rabbits during organogenesis, non-dose–related increases in incidence of fetal malformations and skeletal variations were observed at doses approximately equal to the MRHD and greater. Decreased fetal weights were seen at doses twice the MRHD and greater (see Animal Data).
The estimated background risk for major birth defects and miscarriage is unknown for the indicated population. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and of miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk: A prospective, longitudinal study followed 201 pregnant women with a history of major depressive disorder who were euthymic and taking antidepressants during pregnancy at the beginning of pregnancy. The women who discontinued antidepressants during pregnancy were more likely to experience a relapse of major depression than women who continued antidepressants. Consider the risks to the mother of untreated depression and potential effects on the fetus when discontinuing or changing treatment with antidepressant medications during pregnancy and postpartum.
Data
Human Data: Data from the international bupropion Pregnancy Registry (675 first trimester exposures) and a retrospective cohort study using the United Healthcare database (1,213 first trimester exposures) did not show an increased risk for malformations overall. The Registry was not designed or powered to evaluate specific defects but suggested a possible increase in cardiac malformations.
No increased risk for cardiovascular malformations overall has been observed after bupropion exposure during the first trimester. The prospectively observed rate of cardiovascular malformations in pregnancies with exposure to bupropion in the first trimester from the international Pregnancy Registry was 1.3% (9 cardiovascular malformations/675 first trimester maternal bupropion exposures), which is similar to the background rate of cardiovascular malformations (approximately 1%). Data from the United Healthcare database, which had a limited number of exposed cases with cardiovascular malformations, and a case-control study (6,853 infants with cardiovascular malformations and 5,763 with non-cardiovascular malformations) of self-reported bupropion use from the National Birth Defects Prevention Study (NBDPS) did not show an increased risk for cardiovascular malformations overall after bupropion exposure during the first trimester.
Study findings on bupropion exposure during the first trimester and risk for left ventricular outflow tract obstruction (LVOTO) are inconsistent and do not allow conclusions regarding a possible association. The United Healthcare database lacked sufficient power to evaluate this association; the NBDPS found increased risk for LVOTO (n = 10; adjusted OR = 2.6; 95% CI:1.2, 5.7), and the Slone Epidemiology case control study did not find increased risk for LVOTO.
Study findings on bupropion exposure during the first trimester and risk for ventricular septal defect (VSD) are inconsistent and do not allow conclusions regarding a possible association. The Slone Epidemiology Study found an increased risk for VSD following first trimester maternal bupropion exposure (n = 17; adjusted OR = 2.5; 95% CI: 1.3, 5.0) but did not find increased risk for any other cardiovascular malformations studied (including LVOTO as above). The NBDPS and United Healthcare database study did not find an association between first trimester maternal bupropion exposure and VSD.
For the findings of LVOTO and VSD, the studies were limited by the small number of exposed cases, inconsistent findings among studies, and the potential for chance findings from multiple comparisons in case control studies.
Animal Data: In studies conducted in pregnant rats and rabbits, bupropion was administered orally during the period of organogenesis at doses of up to 450 and 150 mg/kg/day, respectively (approximately 10 and 6 times the MRHD, respectively, on a mg/m2 basis). There was no evidence of fetal malformations in rats. When given to pregnant rabbits during organogenesis, non-dose–related increases in incidence of fetal malformations and skeletal variations were observed at the lowest dose tested (25 mg/kg/day, approximately equal to the MRHD on a mg/m2 basis) and greater. Decreased fetal weights were observed at doses of 50 mg/kg/day (approximately 2 times the MRHD on a mg/m2 basis) and greater. No maternal toxicity was evident at doses of 50 mg/kg/day or less.
In a pre-and postnatal development study, bupropion administered orally to pregnant rats at doses of up to 150 mg/kg/day (approximately 3 times the MRHD on a mg/m2 basis) from embryonic implantation through lactation had no effect on pup growth or development.8.2 Lactation
Risk Summary
Data from published literature report the presence of bupropion and its metabolites in human milk (see Data). There are no data on the effects of bupropion or its metabolites on milk production. Limited data from postmarketing reports have not identified a clear association of adverse reactions in the breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for bupropion and any potential adverse effects on the breastfed child from bupropion or from the underlying maternal condition.
Data
In a lactation study of 10 women, levels of orally dosed bupropion and its active metabolites were measured in expressed milk. The average daily infant exposure (assuming 150 mL/kg daily consumption) to bupropion and its active metabolites was 2% of the maternal weight-adjusted dose. Postmarketing reports have described seizures in breastfed infants. The relationship of bupropion exposure and these seizures is unclear.
8.4 Pediatric Use
Safety and effectiveness in the pediatric population have not been established [see Boxed Warning and Warnings and Precautions (5.1)].
8.5 Geriatric Use
Of the approximately 6,000 subjects who participated in clinical trials with bupropion sustained-release tablets (depression and smoking cessation trials), 275 were aged ≥65 years and 47 were aged ≥75 years. In addition, several hundred subjects aged ≥65 years participated in clinical trials using the immediate-release formulation of bupropion (depression trials). No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Bupropion is extensively metabolized in the liver to active metabolites, which are further metabolized and excreted by the kidneys. The risk of adverse reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, it may be necessary to consider this factor in dose selection; it may be useful to monitor renal function [see Dosage and Administration (2.3), Use in Specific Populations (8.6), Clinical Pharmacology (12.3) ].
8.6 Renal Impairment
Consider a reduced dose and/or dosing frequency of bupropion hydrochloride tablets in patients with renal impairment (Glomerular Filtration Rate: less than 90 mL/min). Bupropion and its metabolites are cleared renally and may accumulate in such patients to a greater extent than usual. Monitor closely for adverse reactions that could indicate high bupropion or metabolite exposures [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
In patients with moderate to severe hepatic impairment (Child-Pugh score: 7 to 15), the maximum dose of bupropion hydrochloride tablets is 75 mg daily. In patients with mild hepatic impairment (Child-Pugh score: 5 to 6), consider reducing the dose and/or frequency of dosing [see Dosage and Administration (2.2)Clinical Pharmacology (12.3)].
-
9 DRUG ABUSE AND DEPENDENCE
9.2 Abuse
Humans
Controlled clinical trials conducted in normal volunteers, in subjects with a history of multiple drug abuse, and in depressed subjects showed some increase in motor activity and agitation/excitement, often typical of central stimulant activity.
In a population of individuals experienced with drugs of abuse, a single oral dose of 400 mg of bupropion produced mild amphetamine-like activity as compared with placebo on the Morphine-Benzedrine Subscale of the Addiction Research Center Inventories (ARCI) and a score greater than placebo but less than 15 mg of the Schedule II stimulant dextroamphetamine on the Liking Scale of the ARCI. These scales measure general feelings of euphoria and drug liking which are often associated with abuse potential.
Findings in clinical trials, however, are not known to reliably predict the abuse potential of drugs. Nonetheless, evidence from single-dose trials does suggest that the recommended daily dosage of bupropion when administered orally in divided doses is not likely to be significantly reinforcing to amphetamine or CNS stimulant abusers. However, higher doses (which could not be tested because of the risk of seizure) might be modestly attractive to those who abuse CNS stimulant drugs.
Bupropion hydrochloride tablets are intended for oral use only. The inhalation of crushed tablets or injection of dissolved bupropion has been reported. Seizures and/or cases of death have been reported when bupropion has been administered intranasally or by parenteral injection.
Animals
Studies in rodents and primates demonstrated that bupropion exhibits some pharmacologic actions common to psychostimulants. In rodents, it has been shown to increase locomotor activity, elicit a mild stereotyped behavior response, and increase rates of responding in several schedule-controlled behavior paradigms. In primate models assessing the positive-reinforcing effects of psychoactive drugs, bupropion was self-administered intravenously. In rats, bupropion produced amphetamine-like and cocaine-like discriminative stimulus effects in drug discrimination paradigms used to characterize the subjective effects of psychoactive drugs.
-
10 OVERDOSAGE
10.1 Human Overdose Experience
Overdoses of up to 30 grams or more of bupropion have been reported. Seizure was reported in approximately one-third of all cases. Other serious reactions reported with overdoses of bupropion alone included hallucinations, loss of consciousness, sinus tachycardia, and ECG changes such as conduction disturbances (including QRS prolongation) or arrhythmias. Fever, muscle rigidity, rhabdomyolysis, hypotension, stupor, coma, and respiratory failure have been reported mainly when bupropion was part of multiple drug overdoses.
Although most patients recovered without sequelae, deaths associated with overdoses of bupropion alone have been reported in patients ingesting large doses of the drug. Multiple uncontrolled seizures, bradycardia, cardiac failure, and cardiac arrest prior to death were reported in these patients.
10.2 Overdosage Management
Consult a Certified Poison Control Center for up-to-date guidance and advice. Telephone numbers for certified poison control centers are listed in the Physician’s Desk Reference (PDR). Call 1-800-222-1222 or refer to www.poison.org.
There are no known antidotes for bupropion. In case of an overdose, provide supportive care, including close medical supervision and monitoring. Consider the possibility of multiple drug overdose. Ensure an adequate airway, oxygenation, and ventilation. Monitor cardiac rhythm and vital signs. Induction of emesis is not recommended.
-
11 DESCRIPTION
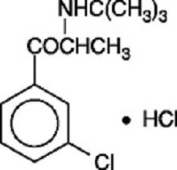
Bupropion hydrochloride, USP an antidepressant of the aminoketone class, is chemically unrelated to tricyclic, tetracyclic, selective serotonin re-uptake inhibitor, or other known antidepressant agents. Its structure closely resembles that of diethylpropion; it is related to phenylethylamines. It is designated as (±)-1-(3-chlorophenyl)-2-[(1,1-dimethylethyl)amino]-1propanone hydrochloride. The molecular weight is 276.2. The molecular formula is C13H18ClNOHCl. Bupropion hydrochloride powder is white, crystalline, and highly soluble in water. It has a bitter taste and produces the sensation of local anesthesia on the oral mucosa. The structural formula is:
Bupropion hydrochloride tablets, USP are supplied for oral administration as 75 mg (yellow) and 100 mg (maroon) film-coated tablets. Each tablet contains the labeled amount of bupropion hydrochloride, USP and the inactive ingredients: 75 mg tablet – colloidal silicone dioxide, crospovidone, D&C yellow No.10, FD&C yellow No.6, hydroxypropyl cellulose, hypromellose, microcrystalline cellulose, polyethylene glycol, talc and titanium dioxide; 100 mg tablet – colloidal silicone dioxide, crospovidone, D&C yellow No.10, FD&C red No.40, FD&C yellow No.6, hydroxypropyl cellulose, hypromellose, microcrystalline cellulose, polyethylene glycol, talc and titanium dioxide. -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The exact mechanism of the antidepressant action of bupropion is not known but is presumed to be related to noradrenergic and/or dopaminergic mechanisms. Bupropion is a relatively weak inhibitor of the neuronal reuptake of norepinephrine and dopamine and does not inhibit the reuptake of serotonin. Bupropion does not inhibit monoamine oxidase.
12.3 Pharmacokinetics
Bupropion is a racemic mixture. The pharmacological activity and pharmacokinetics of the individual enantiomers have not been studied. The mean elimination half-life (±SD) of bupropion after chronic dosing is 21 (±9) hours, and steady-state plasma concentrations of bupropion are reached within 8 days.
Absorption
The absolute bioavailability of bupropion hydrochloride tablets in humans has not been determined because an intravenous formulation for human use is not available. However, it appears likely that only a small proportion of any orally administered dose reaches the systemic circulation intact. In rat and dog studies, the bioavailability of bupropion ranged from 5% to 20%.
In humans, following oral administration of bupropion hydrochloride tablets, peak plasma bupropion concentrations are usually achieved within 2 hours. Plasma bupropion concentrations are dose-proportional following single doses of 100 to 250 mg; however, it is not known if the proportionality between dose and plasma level is maintained in chronic use.
Distribution
In vitro tests show that bupropion is 84% bound to human plasma proteins at concentrations up to 200 mcg/mL. The extent of protein binding of the hydroxybupropion metabolite is similar to that for bupropion, whereas the extent of protein binding of the threohydrobupropion metabolite is about half that seen with bupropion.
Metabolism
Bupropion is extensively metabolized in humans. Three metabolites are active: hydroxybupropion, which is formed via hydroxylation of the tert-butyl group of bupropion, and the amino-alcohol isomers threohydrobupropion and erythrohydrobupropion, which are formed via reduction of the carbonyl group. In vitro findings suggest that CYP2B6 is the principal isoenzyme involved in the formation of hydroxybupropion, while cytochrome P450 enzymes are not involved in the formation of threohydrobupropion. Oxidation of the bupropion side chain results in the formation of a glycine conjugate of meta-chlorobenzoic acid, which is then excreted as the major urinary metabolite. The potency and toxicity of the metabolites relative to bupropion have not been fully characterized. However, it has been demonstrated in an antidepressant screening test in mice that hydroxybupropion is one-half as potent as bupropion, while threohydrobupropion and erythrohydrobupropion are 5-fold less potent than bupropion. This may be of clinical importance because the plasma concentrations of the metabolites are as high as or higher than those of bupropion.
Following a single dose in humans, peak plasma concentrations of hydroxybupropion occur approximately 3 hours after administration of bupropion hydrochloride tablets and are approximately 10 times the peak level of the parent drug at steady state. The elimination half-life of hydroxybupropion is approximately 20 (±5) hours, and its AUC at steady state is about 17 times that of bupropion. The times to peak concentrations for the erythrohydrobupropion and threohydrobupropion metabolites are similar to that of the hydroxybupropion metabolite. However, their elimination half-lives are longer, 33 (±10) and 37 (±13) hours, respectively, and steady-state AUCs are 1.5 and 7 times that of bupropion, respectively.
Bupropion and its metabolites exhibit linear kinetics following chronic administration of 300 to 450 mg/day.
Elimination
Following oral administration of 200 mg of 14C-bupropion in humans, 87% and 10% of the radioactive dose were recovered in the urine and feces, respectively. Only 0.5% of the oral dose was excreted as unchanged bupropion.
Specific Populations
Factors or conditions altering metabolic capacity (e.g., liver disease, congestive heart failure [CHF], age, concomitant medications, etc.) or elimination may be expected to influence the degree and extent of accumulation of the active metabolites of bupropion. The elimination of the major metabolites of bupropion may be affected by reduced renal or hepatic function because they are moderately polar compounds and are likely to undergo further metabolism or conjugation in the liver prior to urinary excretion.
Pateint with Renal Impairment: There is limited information on the pharmacokinetics of bupropion inpatients with renal impairment. An inter-trial comparison between normal subjects and subjects with end-stage renal failure demonstrated that the parent drug Cmax and AUC values were comparable in the 2 groups, whereas the hydroxybupropion and threohydrobupropion metabolites had a 2.3- and 2.8-fold increase, respectively, in AUC for subjects with end-stage renal failure. A second trial, comparing normal subjects and subjects with moderate-to-severe renal impairment (GFR 30.9 ± 10.8 mL/min) showed that after a single 150-mg dose of sustained-release bupropion, exposure to bupropion was approximately 2-fold higher in subjects with impaired renal function, while levels of the hydroxybupropion and threo/erythrohydrobupropion (combined) metabolites were similar in the 2 groups. Bupropion is extensively metabolized in the liver to active metabolites, which are further metabolized and subsequently excreted by the kidneys. The elimination of the major metabolites of bupropion may be reduced by impaired renal function. Bupropion hydrochloride tablets should be used with caution in patients with renal impairment and a reduced frequency and/or dose should be considered [see Use in Specific Populations (8.6)].
Patient with Hepatic Impairment: The effect of hepatic impairment on the pharmacokinetics of bupropionwas characterized in 2 single-dose trials, one in subjects with alcoholic liver disease and one in subjects with mild-to-severe cirrhosis. The first trial demonstrated that the half-life of hydroxybupropion was significantly longer in 8 subjects with alcoholic liver disease than in 8 healthy volunteers (32 ± 14 hours versus 21 ± 5 hours, respectively). Although not statistically significant, the AUCs for bupropion and hydroxybupropion were more variable and tended to be greater (by 53% to 57%) in volunteers with alcoholic liver disease. The differences in half-life for bupropion and the other metabolites in the 2 groups were minimal.
The second trial demonstrated no statistically significant differences in the pharmacokinetics of bupropion and its active metabolites in 9 subjects with mild-to-moderate hepatic cirrhosis compared with 8 healthy volunteers. However, more variability was observed in some of the pharmacokinetic parameters for bupropion (AUC, Cmax, and Tmax) and its active metabolites (t½) in subjects with mild-to-moderate hepatic cirrhosis. In subjects with severe hepatic cirrhosis, significant alterations in the pharmacokinetics of bupropion and its metabolites were seen (Table 3).
Table 3. Pharmacokinetics of Bupropion and Metabolites in Patients with Severe Hepatic Cirrhosis: Ratio Relative to Healthy Matched Controls
Cmax
AUC
t 1/2
Tmaxa
Bupropion
1.69
3.12
1.43
0.5 h
Hydroxybupropion
0.31
1.28
3.88
19 h
Threo/erythrohydrobupropion amino alcohol
0.69
2.48
1.96
20 h
a Difference
Patients with Left Ventricular Dysfunction: During a chronic dosing trial with bupropion in 14 depressedsubjects with left ventricular dysfunction (history of CHF or an enlarged heart on x-ray), there was no apparent effect on the pharmacokinetics of bupropion or its metabolites, compared with healthy volunteers.
Age: The effects of age on the pharmacokinetics of bupropion and its metabolites have not beenfully characterized, but an exploration of steady-state bupropion concentrations from several depression efficacy trials involving subjects dosed in a range of 300 to 750 mg/day, on a 3 times-daily schedule, revealed no relationship between age (18 to 83 years) and plasma concentration of bupropion. A single-dose pharmacokinetic trial demonstrated that the disposition of bupropion and its metabolites in elderly subjects was similar to that of younger subjects. These data suggest there is no prominent effect of age on bupropion concentration; however, another single- and multiple-dose pharmacokinetics trial suggested that the elderly are at increased risk for accumulation of bupropion and its metabolites [seeUseinSpecificPopulations(8.5)].
Male and Female Patients:Pooled analysis of bupropion pharmacokinetic data from 90 healthy male and90 healthy female volunteers revealed no sex-related differences in the peak plasma concentrations of bupropion. The mean systemic exposure (AUC) was approximately 13% higher in male volunteers compared with female volunteers. The clinical significance of this finding is unknown.
Smokers: The effects of cigarette smoking on the pharmacokinetics of bupropion were studiedin 34 healthy male and female volunteers; 17 were chronic cigarette smokers and 17 were nonsmokers. Following oral administration of a single 150-mg dose of bupropion, there were no statistically significant differences in Cmax, half-life, Tmax, AUC, or clearance of bupropion or its active metabolites between smokers and nonsmokers.
Drug Interaction Studies
Potential for Other Drugs to Affect Bupropion Hydrochloride Tablets: In vitro studies indicate that bupropion isprimarily metabolized to hydroxybupropion by CYP2B6. Therefore, the potential exists for drug interactions between Bupropion Hydrochloride Tablets and drugs that are inhibitors or inducers of CYP2B6. In addition, in vitro studies suggest that paroxetine, sertraline, norfluoxetine, fluvoxamine, and nelfinavir inhibit the hydroxylation of bupropion.
Inhibitors of CYP2B6: Ticlopidine, Clopidogrel: In a trial in healthy male volunteers,clopidogrel 75 mg once daily or ticlopidine 250 mg twice daily increased exposures (Cmax and AUC) of bupropion by 40% and 60% for clopidogrel, and by 38% and 85% for ticlopidine, respectively. The exposures (Cmax and AUC) of hydroxybupropion were decreased 50% and 52%, respectively, by clopidogrel, and 78% and 84%, respectively, by ticlopidine. This effect is thought to be due to the inhibition of the CYP2B6-catalyzed bupropion hydroxylation.
Prasugrel: Prasugrel is a weak inhibitor of CYP2B6. In healthy subjects, prasugrel increasedbupropion Cmax and AUC values by 14% and 18%, respectively, and decreased Cmax and AUC values of hydroxybupropion, an active metabolite of bupropion, by 32% and 24%, respectively.
Cimetidine: The threohydrobupropion metabolite of bupropion does not appear to be producedby cytochrome P450 enzymes. The effects of concomitant administration of cimetidine on the pharmacokinetics of bupropion and its active metabolites were studied in 24 healthy young male volunteers. Following oral administration of bupropion 300 mg with and without cimetidine 800 mg, the pharmacokinetics of bupropion and hydroxybupropion were unaffected. However, there were 16% and 32% increases in the AUC and Cmax, respectively of the combined moieties of threohydrobupropion and erythrohydrobupropion.
Citalopram: Citalopram did not affect the pharmacokinetics of bupropion and its threemetabolites.
Inducers of CYP2B6: Ritonavir and Lopinavir: In a healthy volunteer trial, ritonavir 100 mgtwice daily reduced the AUC and Cmax of bupropion by 22% and 21%, respectively. The exposure of the hydroxybupropion metabolite was decreased by 23%, the threohydrobupropion decreased by 38%, and the erythrohydrobupropion decreased by 48%.
In a second healthy volunteer trial, ritonavir at a dose of 600 mg twice daily decreased the AUC and the Cmax of bupropion by 66% and 62%, respectively. The exposure of the hydroxybupropion metabolite was decreased by 78%, the threohydrobupropion decreased by 50%, and the erythrohydrobupropion decreased by 68%.
In another healthy volunteer trial, lopinavir 400 mg/ritonavir 100 mg twice daily decreased bupropion AUC and Cmax by 57%. The AUC and Cmax of hydroxybupropion were decreased by 50% and 31%, respectively.
Efavirenz: In a trial in healthy volunteers, efavirenz 600 mg once daily for 2 weeks reduced theAUC and Cmax of bupropion by approximately 55% and 34%, respectively. The AUC of hydroxybupropion was unchanged, whereas Cmax of hydroxybupropion was increased by 50%.
Carbamazepine, Phenobarbital, Phenytoin: While not systematically studied, these drugsmay induce the metabolism of bupropion.
Potential for Bupropion Hydrochloride Tablets to Affect Other Drugs
Animal data indicated that bupropion may be an inducer of drug-metabolizing enzymes in humans. In one trial, following chronic administration of bupropion 100 mg three times daily to 8 healthy male volunteers for 14 days, there was no evidence of induction of its own metabolism. Nevertheless, there may be potential for clinically important alterations of blood levels of co administered drugs.
Drugs Metabolized by CYP2D6: In vitro, bupropion and its metabolites(erythrohydrobupropion, threohydrobupropion, hydroxybupropion) are CYP2D6 inhibitors. In a clinical trial of 15 male subjects (ages 19 to 35 years) who were extensive metabolizers of CYP2D6, bupropion 300 mg/day followed by a single dose of 50 mg desipramine increased the Cmax, AUC, and t1/2 of desipramine by an average of approximately 2-, 5-, and 2-fold, respectively. The effect was present for at least 7 days after the last dose of bupropion.
Concomitant use of bupropion with other drugs metabolized by CYP2D6 has not been formally studied.
Citalopram: Although citalopram is not primarily metabolized by CYP2D6, in one trialbupropion increased the Cmax and AUC of citalopram by 30% and 40%, respectively.
Lamotrigine: Multiple oral doses of bupropion had no statistically significant effects on thesingle-dose pharmacokinetics of lamotrigine in 12 healthy volunteers.
Digoxin: Literature data showed that digoxin exposure was decreased when a single oral dose of 0.5-mg digoxin was administered 24 hours after a single oral dose of extended-release 150-mg bupropion in healthy volunteers. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis,Impairment Of Fertility
Lifetime carcinogenicity studies were performed in rats and mice at bupropion doses up to 300 and 150 mg/kg/day, respectively. These doses are approximately 6 and 2 times the MRHD, respectively, on a mg/m2 basis. In the rat study there was an increase in nodular proliferative lesions of the liver at doses of 100 to 300 mg/kg/day (approximately 2 to 6 times the MRHD on a mg/m2 basis); lower doses were not tested. The question of whether or not such lesions may be precursors of neoplasms of the liver is currently unresolved. Similar liver lesions were not seen in the mouse study, and no increase in malignant tumors of the liver and other organs was seen in either study.
Bupropion produced a positive response (2 to 3 times control mutation rate) in 2 of 5 strains in the Ames bacterial mutagenicity assay. Bupropion produced an increase in chromosomal aberrations in 1 of 3 in vivo rat bone marrow cytogenetic studies.
There were no effects on male and female fertility when rats were administered oral doses of bupropion up to 300 mg/kg/day (approximately 6 times the MRHD on a mg/m2 basis) to females prior to mating and either through Day 13 of gestation or through lactation, and to males for 60 days prior to and through mating. However, doses of 200 mg/kg/day (approximately 4 times the MRHD on a mg/m2 basis) or greater, caused transient ataxia or behavioral changes in adult female rats. There were also no adverse effects on fertility, reproduction, or growth and development of male or female offspring.
-
14 CLINICAL STUDIES
The efficacy of bupropion hydrochloride tablets in the treatment of major depressive disorder was established in two 4-week, placebo-controlled trials in adult inpatients with MDD (Trials 1 and 2 in Table 4) and in one 6-week, placebo-controlled trial in adult outpatients with MDD (Trial 3 in Table 4). In the first trial, the dose range of bupropion hydrochloride tablets was 300 mg to 600 mg/day administered in 3 divided doses; 78% of subjects were treated with doses of 300 mg to 450 mg/day. The trial demonstrated the efficacy of bupropion hydrochloride tablets as measured by the Hamilton Depression Rating Scale (HDRS) total score, the HDRS depressed mood item (Item 1), and the Clinical Global Impressions-severity score (CGI-S). The second trial included 2 doses of bupropion hydrochloride tablets (300 and 450 mg/day) and placebo. This trial demonstrated the effectiveness of bupropion hydrochloride tablets for only the 450-mg/day dose. The efficacy results were statistically significant for the HDRS total score and the CGI-S score, but not for HDRS Item 1. In the third trial, outpatients were treated with 300 mg/day of bupropion hydrochloride tablets. This trial demonstrated the efficacy of bupropion hydrochloride tablets as measured by the HDRS total score, the HDRS Item 1, the Montgomery-Asberg Depression Rating Scale (MADRS), the CGI-S score, and the CGI-Improvement Scale (CGI-I) score. Effectiveness of bupropion hydrochloride tablets in long-term use, that is, for more than 6 weeks, has not been systematically evaluated in controlled trials.
Table 4. Efficacy of Bupropion Hydrochloride Tablets for the Treatment of Major Depressive DisorderTrial
Number
Treatment Group
Primary Efficacy Measure: HDRS
Mean
Base line
Score (SD)
LS Mean
Score at
Endpoint Visit
(SE)
Placebo
substracted
Differencea
(95% CI)
Trial 1
Bupropion Hydrochloride Tablets300 to 600 mg/dayb
(n = 48)
28.5 (5.1)
14.9 (1.3)
-4.7 (-8.8, -0.6)
Placebo (n = 27)
29.3 (7.0)
19.6 (1.6)
--
Mean
Baseline
Score (SD)
LS Mean
Change from
Baseline (SE)
Placebo subtracted
Differencea
(95% CI)
Trial 2
Bupropion Hydrochloride Tablets 300 mg/day (n = 36)
32.4 (5.9)
-15.5 (1.7)
-4.1
Bupropion Hydrochloride Tablets 450 mg/day b (n = 34)
34.8 (4.6)
-17.4 (1.7)
-5.9 (-10.5,-1.4)
Placebo (n=39)
32.9 (5.4)
-11.5 (1.6)
--
Trial 3
Bupropion Hydrochloride Tablets 300 mg/dayb (n=110)
26.5 (4.3)
-12.0 (NA)
-3.9 (-5.7, -1.0)
Placebo (n = 106)
27.0 (3.5)
-8.7 (NA)
--
n: sample size; SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval included for doses that were demonstrated to be effective; NA: not available.
a Difference (drug minus placebo) in least-squares estimates with respect to the primary efficacy parameter. For Trial 1, it refers to the mean score at the endpoint visit; for Trials 2 and 3, it refers to the mean change from baseline to the endpoint visit.
b Doses that are demonstrated to be statistically significantly superior to placebo.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Bupropion hydrochloride tablets, 75 mg are yellow, round, film coated, biconvex tablets debossed with "C112" on one side and plain on other side. Tablets are provided as follows:
Bottle of 100 Tablets (NDC: 71209-052-05)
Bottle of 1000 Tablets (NDC: 71209-052-11)
A carton containing a total of 100 tablets (10 blister packs of 10 tablets each) (NDC: 71209-052-17)
Bupropion hydrochloride tablets, 100 mg are maroon, round, film coated, biconvex tablets debossed with "C111" on one side and plain on other side. Tablets are provided as follows:
Bottle of 100 Tablets (NDC: 71209-053-05)
Bottle of 1000 Tablets (NDC: 71209-053-11)
A carton containing a total of 100 tablets (10 blister packs of 10 tablets each) (NDC: 71209-053-17)
Store at 20º to 25ºC (68º to 77ºF); excursions permitted between 15º to 30ºC (59º to 86ºF) [See USP Controlled Room Temperature]. Protect from light and moisture. -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Suicidal Thoughts and Behaviors
Instruct patients, their families, and/or their caregivers to be alert to the emergence of anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, mania, other unusual changes in behavior, worsening of depression, and suicidal ideation, especially early during antidepressant treatment and when the dose is adjusted up or down. Advise families and caregivers of patients to observe for the emergence of such symptoms on a day-to-day basis, since changes may be abrupt. Such symptoms should be reported to the patient’s prescriber or healthcare professional, especially if they are severe, abrupt in onset, or were not part of the patient’s presenting symptoms. Symptoms such as these may be associated with an increased risk for suicidal thinking and behavior and indicate a need for very close monitoring and possibly changes in the medication.
Neuropsychiatric Adverse Events and Suicide Risk in Smoking Cessation TreatmentAlthough bupropion hydrochloride tablets are not indicated for smoking cessation treatment, it contains the same active ingredient as ZYBAN which is approved for this use. Inform patients that some patients have experienced changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation and suicide when attempting to quit smoking while taking bupropion. Instruct patients to discontinue bupropion and contact a healthcare professional if they experience such symptoms [see Warnings and Precautions (5.2), Adverse Reactions (6.2)].
Severe Allergic Reactions
Educate patients on the symptoms of hypersensitivity and to discontinue bupropion hydrochloride tablets if they have a severe allergic reaction.
Seizure
Instruct patients to discontinue and not restart bupropion hydrochloride tablets if they experience a seizure while on treatment. Advise patients that the excessive use or abrupt discontinuation of alcohol, benzodiazepines, antiepileptic drugs, or sedatives/hypnotics can increase the risk of seizure. Advise patients to minimize or avoid use of alcohol.
Angle-Closure Glaucoma
Patients should be advised that taking bupropion hydrochloride tablets can cause mild pupillary dilation, which in susceptible individuals, can lead to an episode of angle-closure glaucoma. Pre-existing glaucoma is almost always open-angle glaucoma because angle-closure glaucoma, when diagnosed, can be treated definitively with iridectomy. Open-angle glaucoma is not a risk factor for angle-closure glaucoma. Patients may wish to be examined to determine whether they are susceptible to angle closure, and have a prophylactic procedure (e.g., iridectomy), if they are susceptible [see Warnings and Precautions (5.7)].
Bupropion-Containing Products
Educate patients that bupropion hydrochloride tablets contains the same active ingredient (bupropion hydrochloride) found in ZYBAN, which is used as an aid to smoking cessation treatment, and that bupropion hydrochloride tablets should not be used in combination with ZYBAN or any other medications that contain bupropion (such as WELLBUTRIN SR®, the sustained-release formulation and WELLBUTRIN XL® or FORFIVO XL®, the extended-release formulations, and APLENZIN®, the extended-release formulation of bupropion hydrobromide). In addition, there are a number of generic bupropion HCl products for the immediate-, sustained-, and extended-release formulations.
Potential for Cognitive and Motor Impairment
Advise patients that any CNS-active drug like bupropion hydrochloride tablets may impair their ability to perform tasks requiring judgment or motor and cognitive skills. Advise patients that until they are reasonably certain that bupropion hydrochloride tablets does not adversely affect their performance, they should refrain from driving an automobile or operating complex, hazardous machinery. Bupropion hydrochloride tablets may lead to decreased alcohol tolerance.
Concomitant Medications
Counsel patients to notify their healthcare provider if they are taking or plan to take any prescription or over-the-counter drugs because bupropion hydrochloride tablets and other drugs may affect each others’ metabolisms.
Pregnancy
Advise patients to notify their healthcare provider if they become pregnant or intend to become pregnant during therapy with bupropion hydrochloride tablets. Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to bupropion hydrochloride tablets during pregnancy [see Use in Specific Populations (8.1)].
Storage Information
Instruct patients to store bupropion hydrochloride tablets at room temperature, between 59°F and 86°F (15°C to 30°C) and keep the tablets dry and out of the light.
Administration Information
Instruct patients to take bupropion hydrochloride tablets in equally divided doses 3 or 4 times a day, with doses separated by at least 6 hours to minimize the risk of seizure. Instruct patients if they miss a dose, not to take an extra tablet to make up for the missed dose and to take the next tablet at the regular time because of the dose-related risk of seizure. Instruct patients that bupropion hydrochloride tablets should be swallowed whole and not crushed, divided, or chewed. Bupropion hydrochloride tablets can be taken with or without food.
The brands listed are the registered trademarks of their respective owners.
Manufactured by:
Cadila Pharmaceuticals Limited
1389, Trasad Road, Dholka, 382225
District - Ahmedabad, Gujarat, INDIA
Revised: 11/2019 -
MEDICATION GUIDE
Bupropion Hydrochloride Tablets, USP
(bue proe' pee on hye'' droe klor' ide)
RX only
IMPORTANT: Be sure to read the three sections of this Medication Guide. The first section is about the risk of suicidal thoughts and actions with antidepressant medicines; the second section is about the risk of changes in thinking and behavior, depression and suicidal thoughts or actions with medicines used to quit smoking; and the third section is entitled “What Other Important Information Should I Know About bupropion hydrochloride tablets ?”
Antidepressant Medicines, Depression and Other Serious Mental Illnesses, and Suicidal Thoughts or Actions
This section of the Medication Guide is only about the risk of suicidal thoughts and actions with antidepressant medicines.
What is the most important information I should know about antidepressant medicines, depression and other serious mental illnesses, and suicidal thoughts or actions?
1. Antidepressant medicines may increase the risk of suicidal thoughts or actions in some children, teenagers, or young adults within the first few months of treatment.
2. Depression or other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a particularly high risk of having suicidal thoughts or actions.
These include people who have (or have a family history of) bipolar illness (also called manic-depressive illness) or suicidal thoughts or actions.
3. How can I watch for and try to prevent suicidal thoughts and actions in myself or a family member?
Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
Call your healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
Keep all follow-up visits with your healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call your healthcare provider right away if you or your family member has any of the following symptoms, especially if they are new, worse, or worry you:
- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling very agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
What else do I need to know about antidepressant medicines?
Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
It is not known if bupropion hydrochloride tablets are safe and effective in children under the age of 18.
Quitting Smoking, Quit-Smoking Medications, Changes in Thinking and Behavior, Depression, and Suicidal Thoughts or Actions
This section of the Medication Guide is only about the risk of changes in thinking and behavior, depression and suicidal thoughts or actions with drugs used to quit smoking.
Although bupropion hydrochloride tablets are not a treatment for quitting smoking, it contains the same active ingredient (bupropion hydrochloride) as ZYBAN® which is used to help patients quit smoking.
Talk to your healthcare provider or your family member’s healthcare provider about:
all risks and benefits of quit-smoking medicines.
all treatment choices for quitting smoking.
When you try to quit smoking, with or without bupropion, you may have symptoms that may be due to nicotine withdrawal, including:
urge to smoke
frustration
restlessness
depressed mood
anger
decreased heart rate
trouble sleeping
feeling anxious
increased appetite
irritability
difficulty concentrating
weight gain
Some people have even experienced suicidal thoughts when trying to quit smoking without medication.
Sometimes quitting smoking can lead to worsening of mental health problems that you already have, such as depression.
Some people have had serious side effects while taking bupropion to help them quit smoking, including:
New or worse mental health problems, such as changes in behavior or thinking, aggression, hostility, agitation, depression, or suicidal thoughts or actions. Some people had these symptoms when they began taking bupropion, and others developed them after several weeks of treatment, or after stopping bupropion. These symptoms happened more often in people who had a history of mental health problems before taking bupropion than in people without a history of mental health problems.
Stop taking bupropion hydrochloride tablets and call your healthcare provider right away if you, your family, or caregiver notice any of these symptoms. Work with your healthcare provider to decide whether you should continue to take bupropion hydrochloride tablets. In many people, these symptoms went away after stopping bupropion hydrochloride tablets, but in some people symptoms continued after stopping bupropion hydrochloride tablets. It is important for you to follow-up with your healthcare provider until your symptoms go away.
Before taking bupropion hydrochloride tablets, tell your healthcare provider if you have ever had depression or other mental health problems. You should also tell your healthcare provider about any symptoms you had during other times you tried to quit smoking, with or without bupropion.
What Other Important Information Should I Know About bupropion hydrochloride tablets?
Seizures: There is a chance of having a seizure (convulsion, fit) with bupropion hydrochloride tablets, especially in people:
With certain medical problems.
Who take certain medicines.
The chance of having seizures increases with higher doses of bupropion hydrochloride tablets. For more information, see the sections "Who should not take bupropion hydrochloride tablets?" and "What should I tell my healthcare provider before taking bupropion hydrochloride tablets?" Tell your healthcare provider about all of your medical conditions and all the medicines you take. Do not take any other medicines while you are taking bupropion hydrochloride tablets unless your healthcare provider has said it is okay to take them.
If you have a seizure while taking bupropion hydrochloride tablets, stop taking the tablets and call your healthcare provider right away. Do not take bupropion hydrochloride tablets again if you have a seizure.
High blood pressure (hypertension). Some people get high blood pressure that can be severe, while taking bupropion hydrochloride tablets. The chance of high blood pressure may be higher if you also use nicotine replacement therapy (such as a nicotine patch) to help you stop smoking. (see the section of this Medication Guide called "How should I take bupropion hydrochloride tablets?")
Manic episodes. Some people may have periods of mania while taking bupropion hydrochloride tablets, Including:
Greatly increased energy
Severe trouble sleeping
Racing thoughts
Reckless behavior
Unusually grand ideas
Excessive happiness or irritability
Talking more or faster than usualIf you have any of the above symptoms of mania, call your healthcare provider.
Unusual thoughts or behaviors. Some patients have unusual thoughts or behaviors while taking bupropion hydrochloride tablets, including delusions (believe you are someone else), hallucinations (seeing or hearing things that are not there), paranoia (feeling that people are against you), or feeling confused. If this happens to you, call your healthcare provider.
Visual problems.
eye pain
changes in vision
swelling or redness in or around the eye
Only some people are at risk for these problems. You may want to undergo an eye examination to see if you are at risk and receive preventive treatment if you are.
Severe allergic reactions. Some people can have severe allergic reactions to bupropion hydrochloride tablets. Stop taking bupropion hydrochloride tablets and call your healthcare provider right away if you get a rash, itching, hives, fever, swollen lymph glands, painful sores in the mouth or around the eyes, swelling of the lips or tongue, chest pain, or have trouble breathing. These could be signs of a serious allergic reaction.
What are bupropion hydrochloride tablets?
Bupropion hydrochloride tablets are a prescription medicine used to treat adults with a certain type of depression called major depressive disorder.
Who should not take bupropion hydrochloride tablets?
Do not take bupropion hydrochloride tablets if you
have or had a seizure disorder or epilepsy.
have or had an eating disorder such as anorexia nervosa or bulimia.
are taking any other medicines that contain bupropion, including ZYBAN (used to help people stop smoking), or WELLBUTRIN SR, WELLBUTRIN XL, APLENZIN, or FORFIVO XL. Bupropion is the same active ingredient that is in bupropion hydrochloride tablets.
drink a lot of alcohol and abruptly stop drinking, or use medicines called sedatives (these make you sleepy), benzodiazepines, or anti-seizure medicines, and you stop using them all of a sudden.
take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid.
do not take an MAOI within 2 weeks of stopping bupropion hydrochloride tablets unless directed to do so by your healthcare provider.
do not start bupropion hydrochloride tablets if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your healthcare provider.
are allergic to the active ingredient in bupropion hydrochloride tablets, bupropion, or to any of the inactive ingredients. See the end of this Medication Guide for a complete list of ingredients in bupropion hydrochloride tablets.
What should I tell my healthcare provider before taking bupropion hydrochloride tablets? Tell your healthcare provider if you have ever had depression, suicidal thoughts or actions, or other mental health problems. See "Antidepressant Medicines, Depression and Other Serious Mental Illnesses, and Suicidal Thoughts or Actions."
Tell your healthcare provider about your other medical conditions including if you:
have liver problems, especially cirrhosis of the liver.
have kidney problems.
have, or have had, an eating disorder, such as anorexia nervosa or bulimia.
have had a head injury.
have had a seizure (convulsion, fit).
have a tumor in your nervous system (brain or spine).
have had a heart attack, heart problems, or high blood pressure.
are a diabetic taking insulin or other medicines to control your blood sugar.
drink alcohol.
abuse prescription medicines or street drugs.
are pregnant or plan to become pregnant. Talk to your healthcare provider about the risk to your unborn baby if you take bupropion hydrochloride tablets during pregnancy.
o Tell your healthcare provider if you become pregnant or think you are pregnant during treatment with bupropion hydrochloride tablets.
o If you become pregnant during treatment with bupropion hydrochloride tablets, talk to your healthcare provider about registering with the National Pregnancy Registry for Antidepressants. You can register by calling 1-844-405-6185.
are breastfeeding or plan to breastfeed during treatment with bupropion hydrochloride tablets. Bupropion hydrochloride tablets passes into your milk. Talk to your healthcare provider about the best way to feed your baby during treatment with bupropion hydrochloride tablets.
Tell your healthcare provider about all the medicines you take, including prescription, over-the-counter medicines, vitamins, and herbal supplements. Many medicines increase your chances of having seizures or other serious side effects if you take them while you are taking bupropion hydrochloride tablets
How should I take bupropion hydrochloride tablets?
Take bupropion hydrochloride tablets exactly as prescribed by your healthcare provider. Do not change your dose or stop taking bupropion hydrochloride tablets without talking with your healthcare provider first.
Swallow bupropion hydrochloride tablets whole. Do not chew, cut, or crush bupropion hydrochloride tablets.
Take bupropion hydrochloride tablets at the same time each day.
Take your doses of bupropion hydrochloride tablets at least 6 hours apart.
You may take bupropion hydrochloride tablets with or without food.
If you miss a dose, do not take an extra dose to make up for the dose you missed. Wait and take your next dose at the regular time. This is very important. Too much Bupropion Hydrochloride Tablets can increase your chance of having a seizure.
If you take too much bupropion hydrochloride tablets, or overdose, call your local emergency room or poison control center right away.
Do not take any other medicines while taking bupropion hydrochloride tablets unless your healthcare provider has told you it is okay.
If you are taking bupropion hydrochloride tablets for the treatment of major depressive disorder, it may take several weeks for you to feel that bupropion hydrochloride tablets is working. Once you feel better, it is important to keep taking bupropion hydrochloride tablets exactly as directed by your healthcare provider. Call your healthcare provider if you do not feel bupropion hydrochloride tablets are working for you.
What should I avoid while taking bupropion hydrochloride tablets?
Limit or avoid using alcohol during treatment with bupropion hydrochloride tablets. If you usually drink a lot of alcohol, talk with your healthcare provider before suddenly stopping. If you suddenly stop drinking alcohol, you may increase your risk of having seizures.
Do not drive a car or use heavy machinery until you know how bupropion hydrochloride tablets affects you. Bupropion hydrochloride tablets can affect your ability to do these things safely.
What are possible side effects of bupropion hydrochloride tablets?
Bupropion hydrochloride tablets can cause serious side effects. See the sections at the begining of this Medication Guide for information about serious side effects of bupropion hydrochloride tablets.
The most common side effects of bupropion hydrochloride tablets include:- Nervousness
- Dry mouth
- Constipation
- Headache
- Nausea or vomiting
- Dizziness
- Heavy sweating
- Shakiness (tremor)
- Trouble sleeping
- Blurred vision
- Fast heartbeat
If you have nausea, take your medicine with food. If you have trouble sleeping, do not take your medicine too close to bedtime.
Tell your healthcare provider right away about any side effects that bother you.
These are not all the possible side effects of bupropion hydrochloride tablets. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Cadila Pharmaceuticals Limited at 1-202-355-9785 (Fax 1-202-355-9784).
How should I store bupropion hydrochloride tablets?
Store bupropion hydrochloride tablets at room temperature between 68°F and 77°F (20°C to 25°C).
Keep bupropion hydrochloride tablets dry and out of the light.
Keep bupropion hydrochloride tablets and all medicines out of the reach of children.
General Information about the safe and effective use of bupropion hydrochloride tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use bupropion hydrochloride tablets for a condition for which it was not prescribed. Do not give bupropion hydrochloride tablets to other people, even if they have the same symptoms you have. It may harm them.
If you take a urine drug screening test, bupropion hydrochloride tablets may make the test result positive for amphetamines. If you tell the person giving you the drug screening test that you are taking bupropion hydrochloride tablets, they can do a more specific drug screening test that should not have this problem.
This Medication Guide summarizes important information about bupropion hydrochloride tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about bupropion hydrochloride tablets that is written for healthcare professionals.
For more information about bupropion hydrochloride tablets, go to www.cadilapharma.com or call 1-202-355-9785.
What are the ingredients in Bupropion Hydrochloride Tablets?
Active ingredient: bupropion hydrochloride, USP
Inactive ingredients: 75 mg tablet – colloidal silicone dioxide, crospovidone, D&C yellow No.10, FD&C yellow No.6, hydroxypropyl cellulose, hypromellose, microcrystalline cellulose, polyethylene glycol, talc and titanium dioxide
100 mg tablet – colloidal silicone dioxide, crospovidone, D&C yellow No.10, FD&C red No.40, FD&C yellow No.6, hydroxypropyl cellulose, hypromellose, microcrystalline cellulose, polyethylene glycol, talc and titanium dioxide.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
The brands listed are the registered trademarks of their respective owners.
Manufactured by:
Cadila Pharmaceuticals Limited
1389, Trasad Road, Dholka, 382225
District - Ahmedabad, Gujarat, INDIA
Revised: 11/2019 -
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
BUPROPION HYDROCHLORIDE TABLETS, USP 75 mg - Container Label
NDC-71209-052-05
Three Times Daily
(After Initial Titration)
buPROPion
Hydrochloride
Tablets, USP
75 mgRx Only
100 Tablets

BUPROPION HYDROCHLORIDE TABLETS, USP 75 mg - Container Label
NDC-71209-052-11
Three Times Daily
(After Initial Titration)
buPROPion
Hydrochloride
Tablets, USP
75 mgRx Only
1000 Tablets

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
BUPROPION HYDROCHLORIDE TABLETS, USP 75 mg - Carton Label
NDC-71209-052-17
(10 x 10 unit-dose)
Three Times Daily
(After Initial Titration)
buPROPion
Hydrochloride
Tablets, USPRx Only

BUPROPION HYDROCHLORIDE TABLETS, USP 75 mg - Blister Label
NDC-71209-052-17
(10 x 10 unit-dose)
buPROPion
Hydrochloride
Tablets, USP

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
BUPROPION HYDROCHLORIDE TABLETS, USP 100 mg - Container Label
NDC-71209-053-05
Three Times Daily
(After Initial Titration)
buPROPion
Hydrochloride
Tablets, USP
100 mgRx Only
100 Tablets

BUPROPION HYDROCHLORIDE TABLETS, USP 100 mg - Container Label
NDC-71209-053-11
Three Times Daily
(After Initial Titration)
buPROPion
Hydrochloride
Tablets, USP
100 mgRx Only
1000 Tablets

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
BUPROPION HYDROCHLORIDE TABLETS, USP 100 mg - Carton Label
NDC-71209-053-17
(10 x 10 unit-dose)
Three Times Daily
(After Initial Titration)
buPROPion
Hydrochloride
Tablets, USPRx Only

BUPROPION HYDROCHLORIDE TABLETS, USP 100 mg - Blister Label
NDC-71209-053-17
(10 x 10 unit-dose)
buPROPion
Hydrochloride
Tablets, USP

-
INGREDIENTS AND APPEARANCE
BUPROPION HYDROCHLORIDE
bupropion hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71209-052 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BUPROPION HYDROCHLORIDE (UNII: ZG7E5POY8O) (BUPROPION - UNII:01ZG3TPX31) BUPROPION HYDROCHLORIDE 75 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSPOVIDONE (UNII: 68401960MK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) TALC (UNII: 7SEV7J4R1U) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color YELLOW Score no score Shape ROUND Size 11mm Flavor Imprint Code C112 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71209-052-05 100 in 1 BOTTLE; Type 0: Not a Combination Product 10/30/2018 2 NDC: 71209-052-11 1000 in 1 BOTTLE; Type 0: Not a Combination Product 10/30/2018 3 NDC: 71209-052-17 10 in 1 CARTON 10/30/2018 3 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208606 01/30/2017 BUPROPION HYDROCHLORIDE
bupropion hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71209-053 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BUPROPION HYDROCHLORIDE (UNII: ZG7E5POY8O) (BUPROPION - UNII:01ZG3TPX31) BUPROPION HYDROCHLORIDE 100 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSPOVIDONE (UNII: 68401960MK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) TALC (UNII: 7SEV7J4R1U) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) FD&C RED NO. 40 (UNII: WZB9127XOA) Product Characteristics Color RED (Maroon) Score no score Shape ROUND Size 12mm Flavor Imprint Code C111 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71209-053-05 100 in 1 BOTTLE; Type 0: Not a Combination Product 10/30/2018 2 NDC: 71209-053-11 1000 in 1 BOTTLE; Type 0: Not a Combination Product 10/30/2018 3 NDC: 71209-053-17 10 in 1 CARTON 10/30/2018 3 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208606 01/30/2017 Labeler - Cadila Pharmaceuticals Limited (862257719) Establishment Name Address ID/FEI Business Operations Cadila Pharmaceuticals Limited 918451696 ANALYSIS(71209-052, 71209-053) , LABEL(71209-052, 71209-053) , MANUFACTURE(71209-052, 71209-053) , PACK(71209-052, 71209-053)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.