ACCRUFER- ferric maltol capsule
ACCRUFER by
Drug Labeling and Warnings
ACCRUFER by is a Prescription medication manufactured, distributed, or labeled by Shield TX (UK) Ltd, Patheon France S.A.S, Butterworth Laboratories Ltd, Eurofins Amatsi Analytics, Sterling Pharma Solutions Limited, ACM Pharma. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ACCRUFER ® safely and effectively. See full prescribing information for ACCRUFER.
ACCRUFER (ferric maltol) capsules, for oral use
Initial U.S. Approval: 2019RECENT MAJOR CHANGES
Indications and Usage ( 1) 12/2025
INDICATIONS AND USAGE
ACCRUFER is an iron replacement product indicated for the treatment of iron deficiency in adult and pediatric patients 10 years of age and older. ( 1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Capsules: 30 mg ( 3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
Most common adverse reactions (incidence > 1%) are flatulence, diarrhea, constipation, feces discolored, abdominal pain, nausea, vomiting and abdominal discomfort/distension. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Shield Therapeutics Inc at 1-888-963-6267 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 12/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Increased Risk of Inflammatory Bowel Disease (IBD) Flare
5.2 Iron Overload
5.3 Risk of Overdosage in Children Due to Accidental Ingestion
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on ACCRUFER
7.2 Effect of ACCRUFER on Other Drugs
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 Maltol Phamacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Patients with Inflammatory Bowel Disease (IBD)
14.2 Patients with Chronic Kidney Disease (CKD)
14.3 Pediatric Patients with Iron Deficiency Anemia
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
Adults and Children Aged 10 Years and Above
The recommended dosage of ACCRUFER is 30 mg orally twice daily, on an empty stomach 1 hour before or 2 hours after meals.
Swallow capsules whole. Do not open, break, or chew capsules.
Treatment duration will depend on the severity of iron deficiency but generally at least 12 weeks of treatment is required. The treatment should be continued as long as necessary until ferritin levels are within the normal range.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
ACCRUFER is contraindicated in patients with a history of:
- Hypersensitivity to the active substance or to any of the excipients [see Description ( 11)] . Reactions could include shock, clinically significant hypotension, loss of consciousness, and/or collapse.
- Hemochromatosis and other iron overload syndromes [see Warnings and Precautions ( 5.1)] . Use may result in iron overdose [see Overdosage ( 10)].
- Receiving repeated blood transfusions. Use may result in iron overload [see Warnings and Precautions ( 5.2) and Overdosage ( 10)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Increased Risk of Inflammatory Bowel Disease (IBD) Flare
Avoid use of ACCRUFER in patients with an active inflammatory bowel disease (IBD) flare, as there is potential risk of increased inflammation in the gastrointestinal tract.
5.2 Iron Overload
Excessive therapy with iron products can lead to excess storage of iron with the possibility of iatrogenic hemosiderosis. Do not administer ACCRUFER to patients with evidence of iron overload or patients receiving intravenous iron [see Contraindications( 4)] . Assess iron parameters prior to initiating ACCRUFER and monitor iron parameters while on therapy [see Overdosage ( 10) and Clinical Pharmacology ( 12.2)] .
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Increased Risk of Inflammatory Bowel Disease Flare [see Warnings and Precautions ( 5.1)]
- Iron Overload [see Warnings and Precautions ( 5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to ACCRUFER in 175 adult patients in the placebo-controlled phase of three randomized studies conducted in patients with anemia and quiescent inflammatory bowel disease (IBD) (Studies AEGIS 1 & 2) or non-dialysis dependent chronic kidney disease (CKD) (AEGIS 3). The pooled patient population had a mean age of 58 years, 67.4% were female (n=118), and 81.7% (n=143) were Caucasian.
Table 1 presents all adverse reactions occurring in the placebo-controlled period of the pooled randomized studies [see Clinical Studies ( 14)] occurring at a rate of > 1% in the treated group, and for which the rate for ACCRUFER exceeds the rate for placebo.
Table 1. Adverse Reactions Reported by ≥1% of Patients Treated with ACCRUFER During Placebo-Controlled Period of Pooled Studies (Studies AEGIS1/2 and AEGIS 3) ACCRUFER
30 mg Twice Daily
(N = 175)Placebo
(N = 120)Body System
Adverse Reaction Gastrointestinal Flatulence 4.6% 0.0% Diarrhea 4.0% 1.7% Constipation 4.0% 0.8% Feces discolored 4.0% 0.8% Abdominal pain 2.9% 2.5% Nausea 1.7% 0.8% Vomiting 1.7% 0.0% Abdominal discomfort 1.1% 0.0% Abdominal distension 1.1% 0.0% The proportion of patients who discontinued treatment due to adverse reactions during the double-blind, placebo-controlled portion of studies was 4.6% for patients taking ACCRUFER. The most common adverse reaction leading to discontinuation of ACCRUFER in these studies was abdominal pain (1.7% of patients).
Pediatric Patients with Iron Deficiency Anemia
The safety profile of ACCRUFER in pediatric patients was assessed in 24 patients aged 10 to <18 years of age enrolled to the FORTIS trial and treated with ACCRUFER.
Overall, the safety profile reported in pediatric patients was consistent with the safety profile reported in adult patients with iron deficiency anemia.
-
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on ACCRUFER
Oral Medications
There are no empirical data on avoiding drug interactions between ACCRUFER and concomitant oral medications. Concomitant use of some drugs may reduce the bioavailability of iron after administration of ACCRUFER. Separate the administration of ACCRUFER from these drugs. The duration of separation may depend on the absorption characteristics of the medication concomitantly administered, such as time to peak concentration or whether the drug is an immediate or extended release product. Monitor clinical response to ACCRUFER.
7.2 Effect of ACCRUFER on Other Drugs
Dimercaprol
Concomitant use of iron products with dimercaprol may increase the risk of nephrotoxicity. Avoid concomitant use of ACCRUFER with dimercaprol.
Oral Medications
Concomitant use of ACCRUFER may decrease the bioavailability of some drugs, including mycophenolate, ethinyl estradiol, ciprofloxacin and doxycycline [see Clinical Pharmacology ( 12.3)] . For oral drugs where reductions in bioavailability may cause clinically significant effects on its safety or efficacy, separate the administration of ACCRUFER by at least 4 hours. Monitor clinical responses to concomitant drugs as appropriate.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
ACCRUFER is not absorbed systemically as an intact complex following oral administration, and maternal use is not expected to result in fetal exposure to the drug [see Clinical Pharmacology ( 12.3)] .
In animal reproduction studies, oral administration of ferric or ferrous compounds to gravid CD1-mice and Wistar-rats during organogenesis at doses 13 to 32 times the recommended human dose resulted in no adverse developmental outcomes. An overdose of iron in pregnant women may carry a risk for spontaneous abortion, gestational diabetes and fetal malformation (see Data).
In animal reproduction studies, oral administration of maltol to pregnant Crl: COBS-CD (SD) BR rats during organogenesis at doses 6 times the recommended human dose resulted in no adverse developmental outcomes (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Untreated iron deficiency anemia (IDA) in pregnancy is associated with adverse maternal outcomes such as post-partum anemia. Adverse pregnancy outcomes associated with IDA include increased risk for preterm delivery and low birth weight.
Data
Animal Data
In embryofetal development studies in mice and rats, pregnant animals received oral doses of ferric or ferrous compounds (ferrous sulfate or ferric sodium pyrophosphate) of up to 160 mg/kg/day in mice, or up to 200 mg/kg/day in rats, during the period of organogenesis. Administration of ferric or ferrous compounds at doses 13 times (in mice) or 32 times (in rats) the recommended human dose resulted in no maternal toxicity and no adverse developmental outcomes.
In a multigeneration reproductive and developmental study in rats, pregnant animals received oral doses of maltol of 100, 200, and 400 mg/kg/day, during the period of organogenesis. Administration of maltol at doses 6 times the recommended human dose resulted in no maternal toxicity and no adverse developmental outcomes.
8.2 Lactation
Risk Summary
There are no data on the presence of ACCRUFER in human milk, the effects on the breastfed child, or the effects on milk production. ACCRUFER is not absorbed systemically as an intact complex by the mother following oral administration, and breastfeeding is not expected to result in exposure of the child to ACCRUFER.8.4 Pediatric Use
The safety and effectiveness of ACCRUFER for the treatment of iron deficiency have been established in pediatric patients 10 years and older. Use of ACCRUFER for this indication is supported by evidence from adequate and well-controlled studies of ACCRUFER in adults with additional data from an adequate and well-controlled study conducted in pediatric patients aged 10 years and older [see Clinical Studies ( 14.3)].
Safety and effectiveness of ACCRUFER have not been established in pediatric patients under 10 years.
-
10 OVERDOSAGE
No data are available regarding overdose of ACCRUFER in patients. Acute iron ingestion of 20 mg/kg elemental iron is potentially toxic and 200- 250 mg/kg is potentially fatal. Early signs and symptoms of iron overdose may include nausea, vomiting, abdominal pain and diarrhea. In more serious cases there may be evidence of hypoperfusion, metabolic acidosis and systemic toxicity.
Dosages of ACCRUFER in excess of iron needs may lead to accumulation of iron in storage sites leading to hemosiderosis. Periodic monitoring of iron parameters such as serum ferritin and transferrin saturation may assist in recognizing iron accumulation. Do not administer ACCRUFER to patients with iron overload [see Contraindications ( 4) ].
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
-
11 DESCRIPTION
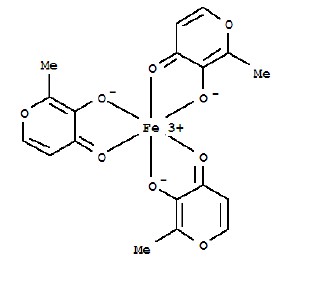
ACCRUFER (ferric maltol) capsules, an iron replacement product for oral administration, contain 30 mg iron and 201.5 mg maltol. Ferric maltol contains iron in a stable ferric state as a complex with a trimaltol ligand. Ferric maltol is 3-hydroxy-2-methyl-4H-pyrane-4-one iron (III) complex (3:1) and has the molecular formula (C 6H 5O 3) 3Fe and a molecular mass of 431.2.
Each red capsule, printed with “30”, contains colloidal anhydrous silica, crospovidone (Type A), lactose monohydrate, magnesium stearate and sodium lauryl sulfate as inactive ingredients. In addition, the capsule shell contains FD&C Blue No. 1, FD&C Red No. 40, FD&C Yellow No.6, hypromellose and titanium dioxide. The ink used for printing the marking contains Shellac, ammonium hydroxide, ethanol, iron oxide black and propylene glycol.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
ACCRUFER delivers iron for uptake across the intestinal wall and transfer to transferrin and ferritin.
12.2 Pharmacodynamics
ACCRUFER has been shown to increase serum iron parameters, including ferritin and transferrin saturation (TSAT) in adults and pediatric patients 10 years and older.
12.3 Pharmacokinetics
Adults
The pharmacokinetic properties of serum iron after administration of ACCRUFER was assessed in subjects with iron deficiency (with or without anemia) following a single dose and at steady state (after 1 week) of ACCRUFER 30 mg, 60 mg, or 90 mg twice daily (1 to 3 times the approved recommended dosage). Total serum iron concentrations increase in a less than dose proportional manner with increasing ACCRUFER doses. Total serum iron peak values were reached 1.5 to 3 hours after administration of ACCRUFER, and were comparable between Day 1 and Day 8.
Pediatric Population
In a pharmacokinetic substudy of the FORTIS trial [see Clinical Studies ( 14.3)] in pediatric patients with iron-deficiency anemia, ferric maltol 15 mg or 30 mg was administered orally twice daily, for 7-10 days in children aged 10 to <18 years. After a single dose of 30 mg on Day 1, baseline-corrected serum iron reached peak concentration (C max = 119 µg/dL) at 2 hours, with a AUC 0-t of 374 h·µg/dL in children aged 12 to <18 years. After a single dose of 15 mg on Day 1, baseline-corrected serum iron reached peak concentration (C max = 40 µg/dL) at 2.1 hours, with a AUC 0-t of 60.1 h·µg/dL in children aged 10 to 11 years. No accumulation of maltol glucuronide was observed.
Absorption
ACCRUFER dissociates upon uptake from the gastrointestinal tract allowing iron and maltol to be absorbed separately.
Effect of Food
Food has been shown to decrease the bioavailability of iron after administration of ferric maltol capsules.
Drug Interaction Studies
In vitro
Of the drugs screened for an interaction with ferric maltol in vitro at pH 1.2, 4.5 and 6.8, only mycophenolate and ethinyl estradiol showed any potential for interaction. Mycophenolate recovery was reduced by up to 16% at pH 1.2 but there was no interaction at pH 4.5; due to solubility issues data are not available for pH 6.8. Ethinyl estradiol recovery was reduced by up to 35% at pH 4.5; due to solubility issues data are not available for pH 1.2 and pH 6.8. These potential oral interactions can be avoided by spacing the administration of those drugs and ACCRUFER [see Drug Interactions ( 7.2)] .
Lisinopril, metoprolol and warfarin showed no interaction at any of the 3 pH conditions and can be taken with ACCRUFER.
No interaction with ferric maltol was observed for atorvastatin (pH 6.8), and norgestimate (pH 1.2) (data were not obtainable at the other pH conditions due to solubility issues).
In vivo
No clinical studies evaluating the drug interaction potential of ACCRUFER have been conducted.
Iron-containing preparations may decrease ciprofloxacin absorption into the bloodstream, resulting in lower serum and urine levels and reduced effectiveness.
Absorption of tetracyclines including doxycycline is reported to be impaired by iron-containing preparations.
12.6 Maltol Phamacokinetics
Maltol is metabolized through glucuronidation (UGT1A6) and sulphation in vitro. Of the total maltol ingested, a mean of between 39.8% and 60% was excreted in the urine as maltol glucuronide. There was no clinically meaningful change in exposure of maltol or maltol glucuronide in subjects with non-dialysis dependent chronic kidney disease (eGFR of >15 mL/min/1.73m 2and <60 mL/min/1.73m 2).
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Ferric Maltol
ACCRUFER is not absorbed systemically as an intact complex.
Carcinogenicity studies have not been conducted with ferric maltol.
Ferric maltol was mutagenic in vitro in reverse bacterial mutation (Ames) assays. Ferric maltol increased revertant frequency in the absence and presence of metabolic activation.
Fertility studies have not been conducted with ferric maltol.
Maltol
The carcinogenic potential of maltol has been evaluated in long-term animal toxicity studies in two species: CD-1 mice and Sprague-Dawley rats. Maltol was not carcinogenic in a 18-month study in mice at doses up to 400 mg/kg (approximately 5.6 times the human daily dose). Maltol was not carcinogenic in a 2-year study in rats at doses up to 400 mg/kg (approximately 11.2 times the human daily dose).
Maltol was mutagenic in vitro in reverse bacterial mutation (Ames) assays. Maltol increased revertant frequency in the absence and presence of metabolic activation. Maltol was clastogenic in vivo in a mouse micronucleus assay (increase in polychromatic erythrocytes) at intraperitoneal doses of 774 mg/kg. Absorbed maltol is rapidly conjugated with glucuronic acid. It is therefore unlikely that the mutagenic activity of maltol would be expressed under the conditions of oral human intake.
In a multi-generation animal reproduction study in male and female rats, there were no effects on mating, fertility, or early embryonic development at doses up to 400 mg/kg/day (approximately 11.2 times the human daily dose).
-
14 CLINICAL STUDIES
14.1 Patients with Inflammatory Bowel Disease (IBD)
The safety and efficacy of ACCRUFER for the treatment of iron deficiency anemia was studied in two randomized, placebo-controlled trials: AEGIS 1 ( NCT01252221) and AEGIS 2 ( NCT01340872). These trials enrolled 128 patients (age range 18-76 years; 45 males and 83 females) with quiescent IBD (58 patients with Ulcerative Colitis [UC] and 70 patients with Crohns disease [CD]) and baseline Hb concentrations between 9.5 g/dL and 12 /13 g/dL for females / males and ferritin < 30 mcg/L. All patients had discontinued prior oral ferrous product treatment due to lack of efficacy or inability to tolerate oral iron replacement products. Subjects were randomized 1:1 to receive either 30 mg ACCRUFER twice daily or a matched placebo control for 12 weeks.) and AEGIS 2 ( NCT01340872). These trials enrolled 128 patients (age range 18-76 years; 45 males and 83 females) with quiescent IBD (58 patients with Ulcerative Colitis [UC] and 70 patients with Crohns disease [CD]) and baseline Hb concentrations between 9.5 g/dL and 12 /13 g/dL for females / males and ferritin < 30 mcg/L. All patients had discontinued prior oral ferrous product treatment due to lack of efficacy or inability to tolerate oral iron replacement products. Subjects were randomized 1:1 to receive either 30 mg ACCRUFER twice daily or a matched placebo control for 12 weeks.
The major efficacy outcome was the mean difference in Hb concentration from baseline to week 12 between ACCRUFER and placebo. The Least Square [LS] mean difference from baseline was 2.18 g/dL (p<0.0001)(see Table 2).
Table 2. Summary of Hemoglobin Concentration (g/dL) and Change from Baseline to Week 12 AEGIS 1 & 2 - Analysis Using Multiple Imputation - Full Analysis Set Population Visit (Week)
StatisticACCRUFER
(N = 64)Placebo
(N =64)Baseline Mean (SD) 11.0 (1.03) 11.10 (0.85) Mean change from baseline to Week 12 LS Mean (SE) 2.25 (0.12) 0.06 (0.13) Treatment Comparison Difference in Change from Baseline LSM Difference (SE)
ACCRUFER Placebo)1-sided lower
97.5%CIp-value ACCRUFER versus placebo 2.18 (0.19) (1.81) <0.0001 Note: Multiple imputation was based on treatment, gender, disease [UC or CD], and Hb concentration at baseline, Week 4, and 8. For each imputed dataset, the change from baseline to Week 12 was analyzed using an ANCOVA model with treatment as the factor and gender, disease, baseline Hb concentration as covariates. The LS mean difference in change from baseline Hb to Week 4 and 8 between ACCRUFER and placebo were 1.04 g/dl and 1.73 g/dl, respectively.
The mean ferritin (mcg/L) levels in ACCRUFER subjects at baseline were 8.6 mcg/L [SD 6.77]) and the mean ferritin (mcg/L) levels at Week 12 were 26.0 mcg/L [SD 30.57] with a mean overall improvement of 17.3 mcg/L.
Following completion of the 12-week placebo-controlled phase of the studies, eligible patients transitioned to ACCRUFER 30 mg twice daily open-label treatment for an additional 52 weeks.
During the open-label phase with ACCRUFER, the mean change in Hb concentration from baseline to Week 64 was 3.1 g/dL [SD 1.46 g/dL, n = 35] and the ferritin value demonstrated a mean of 68.9 mcg/L [SD 96.24] at 64 weeks, with a mean overall improvement of 60.4 mcg/L.
14.2 Patients with Chronic Kidney Disease (CKD)
The safety and efficacy of ACCRUFER for the treatment of iron deficiency anemia was studied in AEGIS 3 ( NCT02968368), a trial that enrolled 167 patients (mean age 67.4 years, range 30-90 years; 50 males and 117 females) with non-dialysis dependent chronic kidney disease (CKD) and baseline hemoglobin (Hb) concentrations between 8g/dL and 11 g/dL and ferritin < 250 mcg/L with a Transferrin saturation (TSAT) <25% or ferritin < 500 mcg/L with a TSAT <15%. ACCRUFER was administered at a dose of 30 mg twice daily. Subjects were randomized 2:1 to receive either 30 mg ACCRUFER twice daily or a matched placebo control for 16 weeks.
The major efficacy outcome was the mean difference in Hb concentration from baseline to Week 16 between ACCRUFER and placebo. The LS mean difference was 0.52 g/dL (p= 0.0149) (see Table 3).
Table 3. Summary of Hemoglobin Concentration (g/dL) and Change from Baseline to Week 16 Analysis Using Multiple Imputation Intent-to-Treat Population Visit (Week)
StatisticACCRUFER
(N = 111)Placebo
(N = 56)Baseline Mean (SD) 10.06 (0.77) 10.03 (0.82) Mean change from baseline to Week 16 LS Mean (SE) 0.50 (0.12) -0.02 (0.16) Treatment Comparison Difference in Change from Baseline LSM Difference (SE)
ACCRUFER – Placebo95% CI p-value ACCRUFER versus placebo 0.52 (0.21) (0.10, 0.93) 0.0149 Note: Multiple imputation was based on treatment, gender, eGFR at baseline, and Hb concentration at baseline, Week 4 and 8. For each imputed dataset, the change from baseline to Week 16 was analyzed using an ANCOVA model with treatment as the factor and baseline Hb concentration, baseline eGFR as covariates. The LS mean difference in change from baseline Hb to Week 4 and 8 between ACCRUFER and placebo were 0.13 g/dl and 0.46 g/dl, respectively.
The mean change in ferritin concentration from baseline to Week 16 was 49.3 mcg/L for the ACCRUFER group and 6.3 mcg/L for the placebo group. The mean difference for ACCRUFER versus placebo was 43.0 mcg/L.
14.3 Pediatric Patients with Iron Deficiency Anemia
The efficacy of ACCRUFER for the treatment of iron deficiency was evaluated in 24 patients aged 10 to <18 (age range 10-17 years; 4 males and 20 females; 13 White, 8 Black or African American, 2 Asian, 1 Other; 10 Hispanic or Latino, 14 Not Hispanic or Latino) with iron deficiency anemia who received aged-based dosing of ACCRUFER twice daily in the FORTIS study. Patients aged 10-11 years received 15 mg twice daily while patients 12 to <18 years received 30 mg twice daily.
Efficacy was assessed based on mean change from baseline to week 12 in Hb (g/dL) (descriptive statistics). A clinically significant increase in Hb was observed in children 10 years and above receiving ACCRUFER orally twice daily for 12 weeks (see Table 4). The results are presented for the pooled population who received either 15 mg twice daily or 30 mg orally twice daily (based on age). The 15 mg twice daily dose is not approved and not recommended for use. The recommended dosage of ACCRUFER in pediatric patients aged 10 years and over is 30 mg orally twice daily.
Table 4. Summary of Hemoglobin Concentration (g/dL) and Change from Baseline to Week 12 in Pediatric Patients with Iron Deficiency Anemia in the FORTIS Study a Endpoint ACCRUFER
10 to <18 years
(N=24)Hb concentrations (g/dL) Baseline (SD) 10.73 (0.89) Mean change from baseline to week 12 b 1.10 (1.06) a Subset of patients in the 10 to <18 subgroup
b Modified-Intent to Treat (mITT) population: all subjects who received at least 1 dose of study drug
The mean change in Hb at week 12 in the subgroup (N=21) who received the recommended dose of ACCRUFER 30 mg orally twice daily was 1.23 g/dL [SD 1.00].
The mean ferritin levels in ACCRUFER subjects aged 10 to <18 years (N=24) at baseline were 11.4 mcg/L [SD 10.06] and the mean ferritin levels at Week 12 were 20 mcg/L [SD 13.51] with a mean overall improvement of 8.6 mcg/L [SD 10.29]. -
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
ACCRUFER (ferric maltol) 30 mg iron capsules are supplied as 60 red capsules, printed with “30”, in HDPE bottles with a child-proof polypropylene push-lock.
1 Bottle of 60-count 30 mg ferric iron capsules (NDC: 73059-001-60).
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Administration
- Inform patients to take ACCRUFER as directed on an empty stomach 1 hour before or 2 hours after meals. Instruct patients on concomitant medications that should be dosed apart from ACCRUFER [see Dosage and Administration ( 2.1) and Drug Interactions ( 7.2)] .
- Advise patients to swallow the capsules whole and not to open, break, or chew capsules.
Adverse Reactions
Advise patients that ACCRUFER may cause, flatulence, diarrhea, constipation, discolored feces, abdominal pain, nausea, vomiting or abdominal bloating or discomfort. Advise patients to report severe or persistent gastrointestinal symptoms or any allergic reactions to their physician [see Adverse Reactions ( 6.1)].
Increased Risk of IBD Flare
Advise patients that they should not use ACCRUFER if they are experiencing an IBD flare [see Warnings and Precautions ( 5.1)] .
Iron Overload and Risk of Accidental Overdose in Children
Inform patients to keep this product out of reach of children as accidental overdose of iron products is a leading cause of fatal poisonings in children. In case of accidental overdose, advise them to call a doctor or poison control center immediately [see Warnings and Precautions ( 5.2,5.3)].
Distributed by Shield Therapeutics Inc, 7-57 Wells Avenue Newton, MA, 02459
-
Patient Information
ACCRUFER ®(ak-roo-fer)
(ferric maltol)
capsulesWhat is ACCRUFER?
ACCRUFER is a prescription medicine used in adults and children 10 years of age and older to treat low iron stores in your body.
It is not known if ACCRUFER is safe and effective in children under 10 years of age.
Do not take ACCRUFER if you:
- are allergic to ferric maltol or any of the ingredients in ACCRUFER. See the end of this leaflet for a complete list of ingredients in ACCRUFER.
- have any illness that causes you to store too much iron in your body or if you have a problem with how your body uses iron.
- are receiving repeated blood transfusions.
Before taking ACCRUFER, tell your healthcare provider about all your medical conditions, including if you:
- have inflammatory bowel disease (IBD).
- are pregnant or plan to become pregnant. It is not known if ACCRUFER will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if ACCRUFER passes into your breast milk and may harm your baby. Talk to your healthcare provider about the best way to feed your baby during treatment with ACCRUFER.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Taking ACCRUFER with certain other medicines may affect each other causing serious side effects.
You may need to take ACCRUFER at least 4 hours before or 4 hours after you take certain medicines. Ask your healthcare provider for a list of these medicines if you are not sure if you take one of these medicines.
Especially tell your healthcare provider if you take:
- dimercaprol
- other oral iron tablets or health supplements containing iron
Ask your healthcare provider if you are not sure if you take one of these medicines.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I take ACCRUFER?
- Take ACCRUFER exactly as your healthcare provider tells you to.
- Take ACCRUFER 2 times a day on an empty stomach 1 hour before or 2 hours after meals.
- Swallow ACCRUFER capsules whole. Do not open, break, or chew ACCRUFER capsules.
- In case of accidental overdose, call your healthcare provider or Poison Help line at 1-800-222-1222 or go to the nearest hospital emergency room right away.
What are the possible side effects of ACCRUFER?
ACCRUFER may cause serious side effects, including:
- Increased risk of inflammatory bowel disease (IBD) flare.You should avoid taking ACCRUFER if you have inflammatory bowel disease (IBD) and are experiencing a flare.
- Too much iron stored in your body (iron overload).Your healthcare provider should check the iron level in your blood before you start and during treatment with ACCRUFER.
- Risk of overdose in children due to accidental swallowing.Accidental overdose of iron-containing products is a leading cause of death from poisoning in children under 6. Keep ACCRUFER in a safe place and out of the reach of children.
The most common side effects of ACCRUFER include:
- gas
- diarrhea
- constipation
- discolored stools
- stomach pain
- nausea
- vomiting
- stomach area discomfort or bloating
These are not all the possible side effects of ACCRUFER.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ACCRUFER?
- Store ACCRUFER at room temperature between 68°F to 77°F (20°C to 25°C).
- ACCRUFER comes in a child-resistant bottle.
Keep ACCRUFER and all medicines out of reach of children.
General information about the safe and effective use of ACCRUFER.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ACCRUFER for a condition for which it was not prescribed. Do not give ACCRUFER to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ACCRUFER that is written for health professionals.
What are the ingredients in ACCRUFER?
Active ingredient: ferric maltol
Inactive ingredients:
Capsule: colloidal anhydrous silica, crospovidone (Type A), lactose monohydrate, magnesium stearate, sodium lauryl sulfate
Capsule Shell: FD&C Blue No. 1 FD&C Red No. 40, FD&C Yellow 6, hypromellose, titanium dioxide.
Ink: Shellac, ammonium hydroxide, ethanol, iron oxide black, propylene glycol
Distributed by Shield Therapeutics Inc, 7-57 Wells Avenue Newton, MA, 02459
US Patents 7459569, 9248148, 9802973, 10179120
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: December 2025 -
PRINCIPAL DISPLAY PANEL - 60 Capsules
Carton Label - ACCRUFER ®(ferric maltol) capsules 30 mg (60 Capsules)
NDC: 73059-001-60
ACCRUFeR ®
(ferric maltol*)
capsules
30 mgFor oral
administration only60 capsules
Distributed by
Shield Therapeutics IncSHIELD
THERAPEUTICSRx only

Bottle Label - ACCRUFER ®(ferric maltol) capsules 30 mg
NDC73059-001-60
ACCRUFeR ®
(ferric maltol*)
capsules
30 mg
For oral administration only60 capsules
SHIELD
THERAPEUTICSRx only
*Each capsule contains 30 mg iron (as
ferric maltol)WARNING:Accidental overdose of
iron-containing products is a leading
cause of fatal poisoning in children
under 6. Keep this product out of reach
of children. In case of accidental
overdose, call a doctor or poison control
center immediately.Store at 20°C to 25°C (68°F to 77°F);
excursions permitted to 15°C to 30°C
(59°F to 86°F). (See USP controlled room
temperature).
Usual Dosage: 1 capsule twice daily on an
empty stomach. See prescribing information.
Distributed by Shield Therapeutics Inc.
-
PRINCIPAL DISPLAY PANEL - 10 Capsules
Carton Label - ACCRUFER ® (ferric maltol) capsules 30 mg (10 Capsules)
NDC: 73059-001-10
ACCRUFeR®
(ferric maltol*)capsules
30 mg
For oral
administration only10 capsules
Professional Sample -
Not For SaleSHIELD
THERAPEUTICSRx only
Bottle Label - ACCRUFER ® (ferric maltol) capsules 30 mg (10 Capsules)
NDC 73059-001-10
ACCRUFeR®
(ferric maltol*) 30 mg capsules
For oral administration
only10 capsules
Sample-Not for Sale
SHIELD
THERAPEUTICSRx only
*Each capsule contains 30 mg iron (as ferric maltol).
Store at 20°C to 25°C (68°F to 77°F);
Usual Dosage: 1 capsule twice daily on an
empty stomach. See Prescribing Information.Distributed by Shield Therapeutics Inc.

-
INGREDIENTS AND APPEARANCE
ACCRUFER
ferric maltol capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73059-001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FERRIC MALTOL (UNII: MA10QYF1Z0) (FERRIC CATION - UNII:91O4LML611) FERRIC CATION 30 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) SODIUM LAURYL SULFATE (UNII: 368GB5141J) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSPOVIDONE (UNII: 2S7830E561) HYPROMELLOSES (UNII: 3NXW29V3WO) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color red Score no score Shape CAPSULE Size 19mm Flavor Imprint Code 30 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73059-001-60 1 in 1 CARTON 07/01/2021 1 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC: 73059-001-06 1 in 1 CARTON 11/01/2021 07/21/2023 2 6 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC: 73059-001-10 1 in 1 CARTON 04/01/2022 3 10 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA212320 07/01/2021 Labeler - Shield TX (UK) Ltd (211237588) Registrant - Patheon France S.A.S (543127229) Establishment Name Address ID/FEI Business Operations Eurofins Amatsi Analytics 270324185 analysis(73059-001) Establishment Name Address ID/FEI Business Operations Sterling Pharma Solutions Limited 349157623 api manufacture(73059-001) Establishment Name Address ID/FEI Business Operations ACM Pharma 384719527 analysis(73059-001) Establishment Name Address ID/FEI Business Operations Patheon France S.A.S 543127229 manufacture(73059-001) , analysis(73059-001) , pack(73059-001) , label(73059-001)
Trademark Results [ACCRUFER]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ACCRUFER 88212183 5894686 Live/Registered |
Shield TX (UK) Limited 2018-11-30 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.