SOLE SERUM- lidocaine spray
SOLE SERUM by
Drug Labeling and Warnings
SOLE SERUM by is a Otc medication manufactured, distributed, or labeled by THE COMFORT LAB LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only. If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a physician. Do not use in large quantities, particularly over raw surfaces or blistered areas. Avoid contact with the eyes. Rinse with water to remove. - KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
-
INACTIVE INGREDIENT
Inactive Ingredients
Acrylates/C10-30 Alkyl Acrylates Crosspolymer, Acrylates/Dimethicone Copolymer, Alkyl Benzoate, Diazolidinyl Urea, Ethoxydiglycol, Ethylenediaminetetraacetic acid, Disodium EDTA, Ethylhexyl Palmitate, Glyceryl Stearate, Peg 100 Stearate, Isododecane, Behenyl
Methacrylate/t-Butyl Methacrylate Copolymer, Isopropyl Palmitate, Lavandula Angustifolia (Lavender) Oil, Lecithin, Malaleuca Alternifolia {Tea
Tree) Leaf Oil, Mentha Piperita (Peppermint) Oil, Menthol, Peg-8 Dimethicone, Phenoxyethanol,Benzoic Acid, Ethylhexylglycerin, Glycereth -2 Cocoate, Poloxamer 407, Potassium Cetyl Phosphate, Potassium Sorbate, Propylene Glycol, Sorbic Acid, Water - OTHER SAFETY INFORMATION
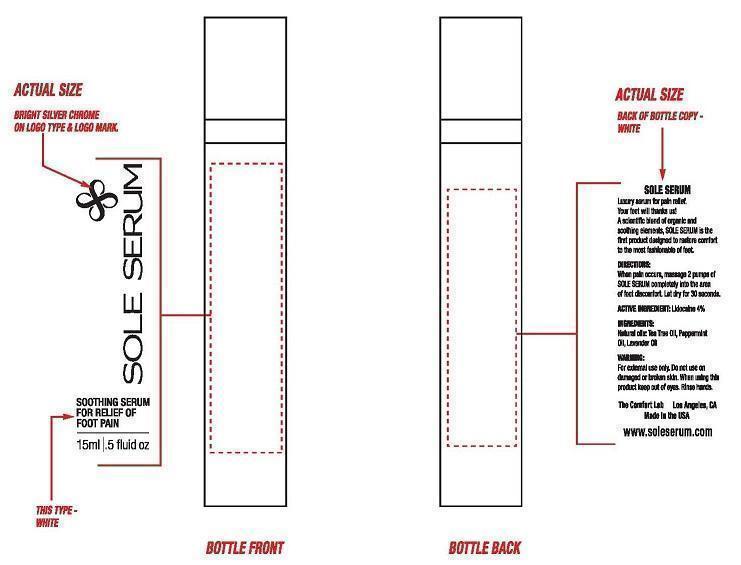
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SOLE SERUM
lidocaine sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 61867-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 4 g in 100 mL Inactive Ingredients Ingredient Name Strength CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) DIETHYLENE GLYCOL MONOETHYL ETHER (UNII: A1A1I8X02B) EDETIC ACID (UNII: 9G34HU7RV0) EDETATE DISODIUM (UNII: 7FLD91C86K) Ethylhexyl Palmitate (UNII: 2865993309) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) ISODODECANE (UNII: A8289P68Y2) TERT-BUTYL METHACRYLATE (UNII: F9029V5818) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) LAVENDER OIL (UNII: ZBP1YXW0H8) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) TEA TREE OIL (UNII: VIF565UC2G) PEPPERMINT OIL (UNII: AV092KU4JH) MENTHOL (UNII: L7T10EIP3A) PEG-8 DIMETHICONE (UNII: GIA7T764OD) PHENOXYETHANOL (UNII: HIE492ZZ3T) BENZOIC ACID (UNII: 8SKN0B0MIM) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERETH-2 COCOATE (UNII: JWM00VS7HC) POLOXAMER 407 (UNII: TUF2IVW3M2) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SORBIC ACID (UNII: X045WJ989B) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 61867-101-51 1 in 1 BOX 1 NDC: 61867-101-11 15 mL in 1 BOTTLE 2 NDC: 61867-101-52 1 in 1 BOX 2 NDC: 61867-101-12 30 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 02/24/2014 Labeler - THE COMFORT LAB LLC (079250438) Registrant - THE COMFORT LAB LLC (079250438)
Trademark Results [SOLE SERUM]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SOLE SERUM 86581851 4805776 Live/Registered |
INNOVATIVE BRANDS LLC 2015-03-31 |
 SOLE SERUM 86015562 not registered Dead/Abandoned |
THE COMFORT LAB, LLC 2013-07-19 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.