Target Corporation Allergy Relief Drug Facts
up and up allergy relief by
Drug Labeling and Warnings
up and up allergy relief by is a Otc medication manufactured, distributed, or labeled by Target Corporation. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
UP AND UP ALLERGY RELIEF- fexofenadine hydrochloride tablet, film coated
Target Corporation
----------
Target Corporation Allergy Relief Drug Facts
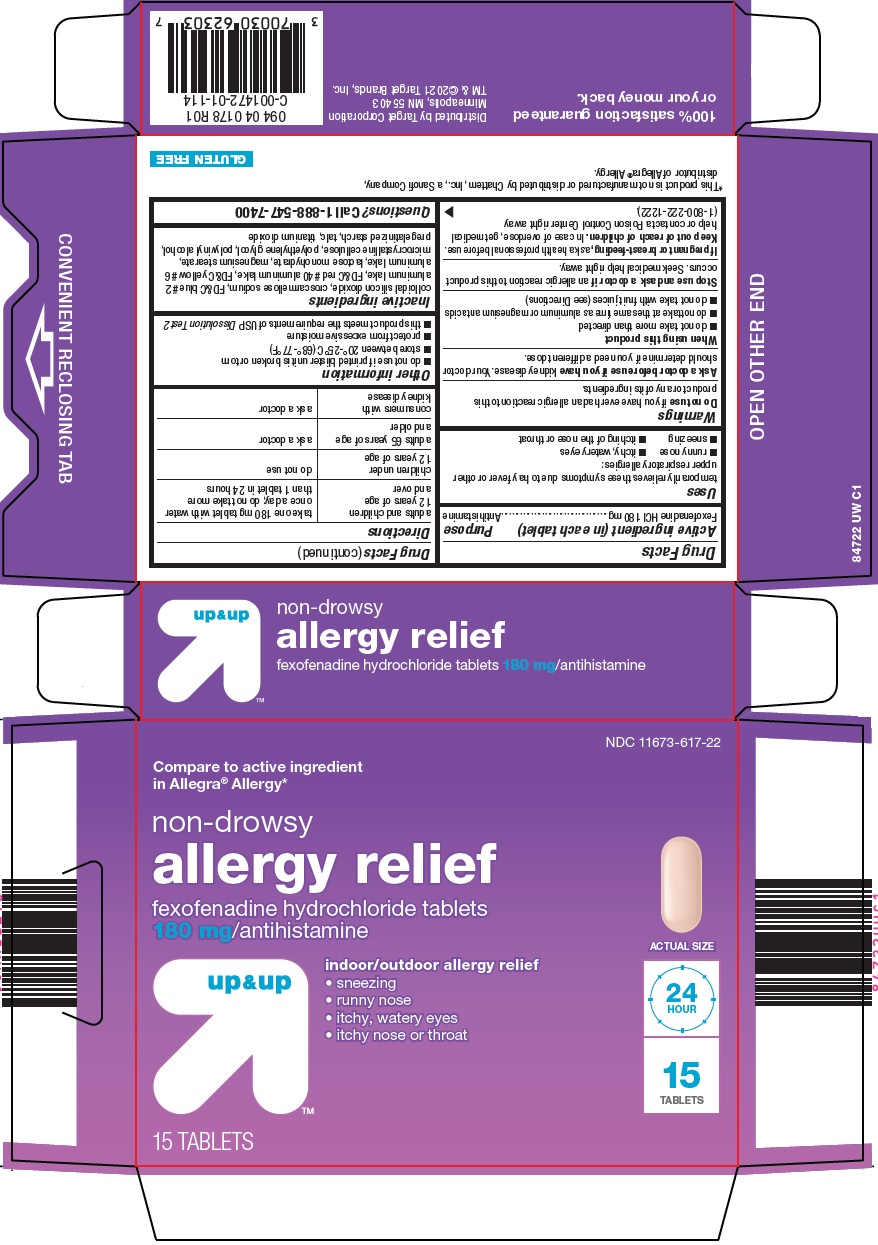
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Directions
|
adults and children 12 years of age and over |
take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours |
|
children under 12 years of age |
do not use |
|
adults 65 years of age and older |
ask a doctor |
|
consumers with kidney disease |
ask a doctor |
Other information
- do not use if printed blister unit is broken or torn
- store between 20° -25°C (68° -77°F)
- protect from excessive moisture
- this product meets the requirements of USP Dissolution Test 2
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, pregelatinized starch, talc, titanium dioxide
Package/Label Principal Display Panel
Compare to active ingredient in Allegra® Allergy
non-drowsy
allergy relief
fexofenadine hydrochloride tablets 180 mg/antihistamine
indoor/outdoor allergy relief
sneezing
runny nose
itchy, watery eyes
itchy nose or throat
ACTUAL SIZE
24 HOUR
15 TABLETS
15 TABLETS
| UP AND UP ALLERGY RELIEF
fexofenadine hydrochloride tablet, film coated |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Target Corporation (006961700) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.