FERROUS SULFATE tablet, film coated
Ferrous Sulfate by
Drug Labeling and Warnings
Ferrous Sulfate by is a Otc medication manufactured, distributed, or labeled by Central Texas Community Health Centers. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT(S)
-
INACTIVE INGREDIENTS
Other ingredients for Red Tablets: microcrystalline cellulose, dibasic calcium phosphate dihydrate, croscarmellose sodium, sodium starch glycolate, hypromellose, stearic acid, polyethylene glycol (PEG) 400, FD&C red #40 aluminum lake, magnesium stearate, titanium dioxide, polyethylene glycol (PEG) 8000, carnauba wax
Other ingredients for Green Tablets: microcrystalline cellulose, dibasic calcium phosphate dihydrate, croscarmellose sodium, sodium starch glycolate, hypromellose, stearic acid, polyethylene glycol (PEG) 400, magnesium stearate, riboflavin, FD&C bule #1 aluminum lake, titanium dioxide, FD&C blue #2 aluminum lake, polyethylene glycol (PEG) 8000, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, carnauba wax - PURPOSE
- USE(S)
-
WARNINGS
Iron may interfere with absorpiton of certain antibiotics; these products should not be taken within two hours of each other.
Occasional gastrointestinal discomfort (such as nausea) may be minimized by taking iron with meals. Iron-containing products may occassionally cause constipation or diarrhea. If pregnant or nursing consult a physicial before using this product.
- DO NOT USE
- OTHER REQUIRED WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- STORAGE
- HOW SUPPLIED
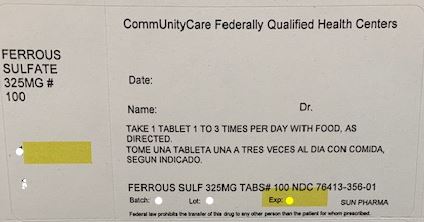
- FERROUS SULFATE TABLET, FILM COATED
-
INGREDIENTS AND APPEARANCE
FERROUS SULFATE
ferrous sulfate tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 76413-356(NDC: 57664-070) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FERROUS SULFATE (UNII: 39R4TAN1VT) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 325 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) STEARIC ACID (UNII: 4ELV7Z65AP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) FD&C RED NO. 40 (UNII: WZB9127XOA) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) CARNAUBA WAX (UNII: R12CBM0EIZ) RIBOFLAVIN (UNII: TLM2976OFR) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color GREEN Score no score Shape ROUND Size 10mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76413-356-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 07/01/2014 Labeler - Central Texas Community Health Centers (079674019) Establishment Name Address ID/FEI Business Operations Central Texas Community Health Centers 079674019 RELABEL(76413-356) , REPACK(76413-356)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.