ADULT TUSSIN DM COUGH PLUS CHEST CONGESTION DM- dextromethorphan hbr, guaifenesin liquid
ADULT TUSSIN DM COUGH PLUS CHEST CONGESTION DM by
Drug Labeling and Warnings
ADULT TUSSIN DM COUGH PLUS CHEST CONGESTION DM by is a Otc medication manufactured, distributed, or labeled by Marc Glassman, Inc., Aurohealth LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredients (in each 10 mL)
- Purposes
- Uses
- Warnings
-
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- Ask a doctor before use if you have
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
- do not take more than 6 doses (12 tsp) in any 24-hour period
- keep dosing cup with product
- measure only with dosing cup provided
- this adult product is not intended for use in children under 12 years of age
- mL = milliliter
Age
Dose
adults and children
12 years and over
10 mL
every 4 hours
children under 12 years
do not use
- Other information
- Inactive ingredients
- QUESTIONS
-
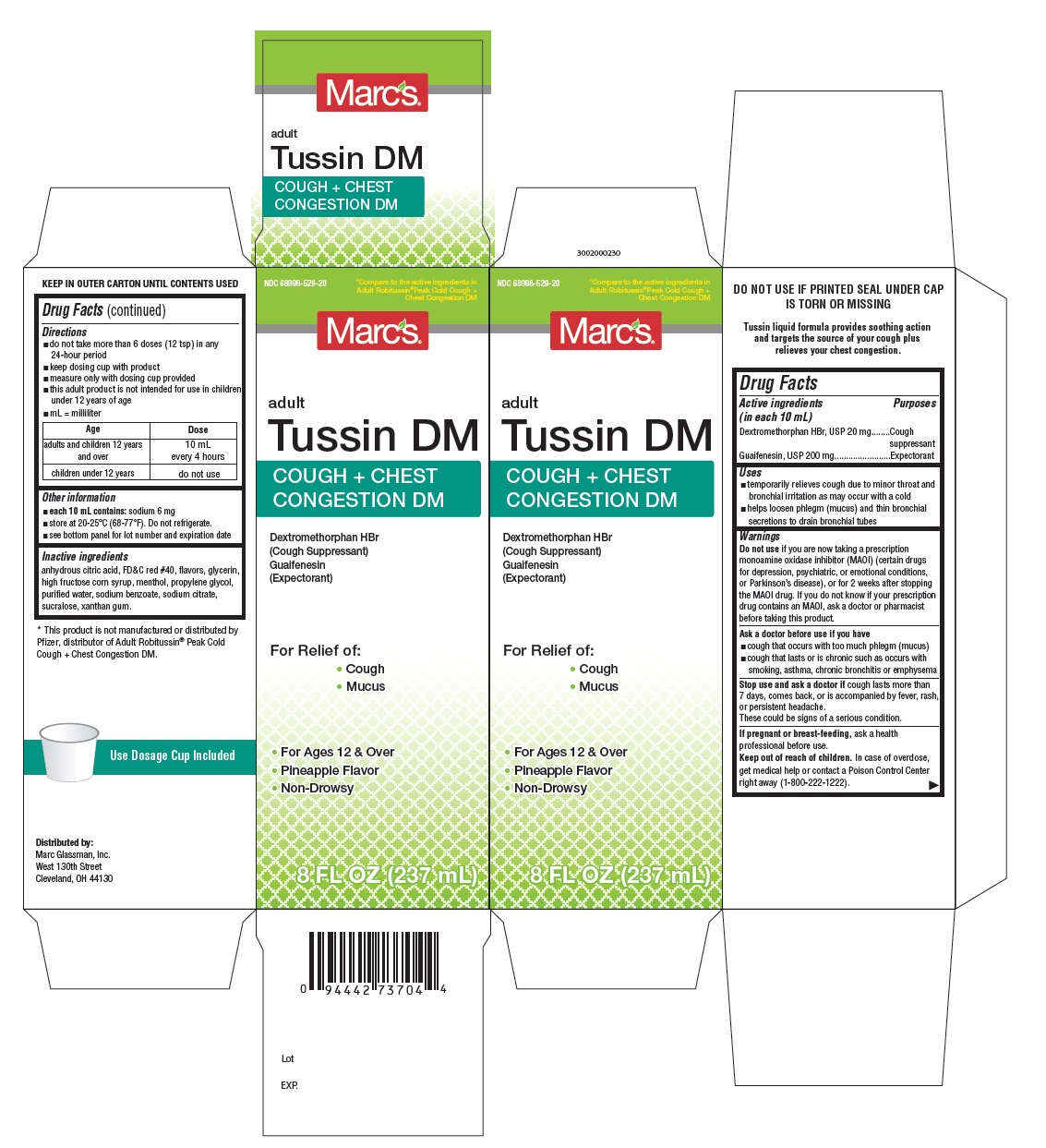
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 8 FL OZ (237 mL Bottle)
NDC: 68996-529-20
*Compare to th active ingredients in
Adult Robitussin® Peak Cold Cough+
Chest Congestion DM
Macrs®
adult
Tussin DM
COUGH + CHEST
CONGESTION DM
Dextromethorphan HBr
(Cough Suppressant)
Guaifenesin
(Expectorant)
For Relief of:- Cough
- Mucus
- For Ages 12 & Over
- Pineapple Flavor
- Non-Drowsy
8 FLOZ (237 mL)
-
INGREDIENTS AND APPEARANCE
ADULT TUSSIN DM COUGH PLUS CHEST CONGESTION DM
dextromethorphan hbr, guaifenesin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68998-529 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 20 mg in 10 mL GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 200 mg in 10 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) FD&C RED NO. 40 (UNII: WZB9127XOA) ORANGE (UNII: 5EVU04N5QU) PINEAPPLE (UNII: 2A88ZO081O) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color RED (a clear red colored liquid) Score Shape Size Flavor ORANGE, PINEAPPLE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68998-529-20 1 in 1 CARTON 07/14/2017 1 237 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part341 07/14/2017 Labeler - Marc Glassman, Inc. (094487477) Registrant - Aurohealth LLC (078728447) Establishment Name Address ID/FEI Business Operations Aurohealth LLC 078728447 MANUFACTURE(68998-529)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.