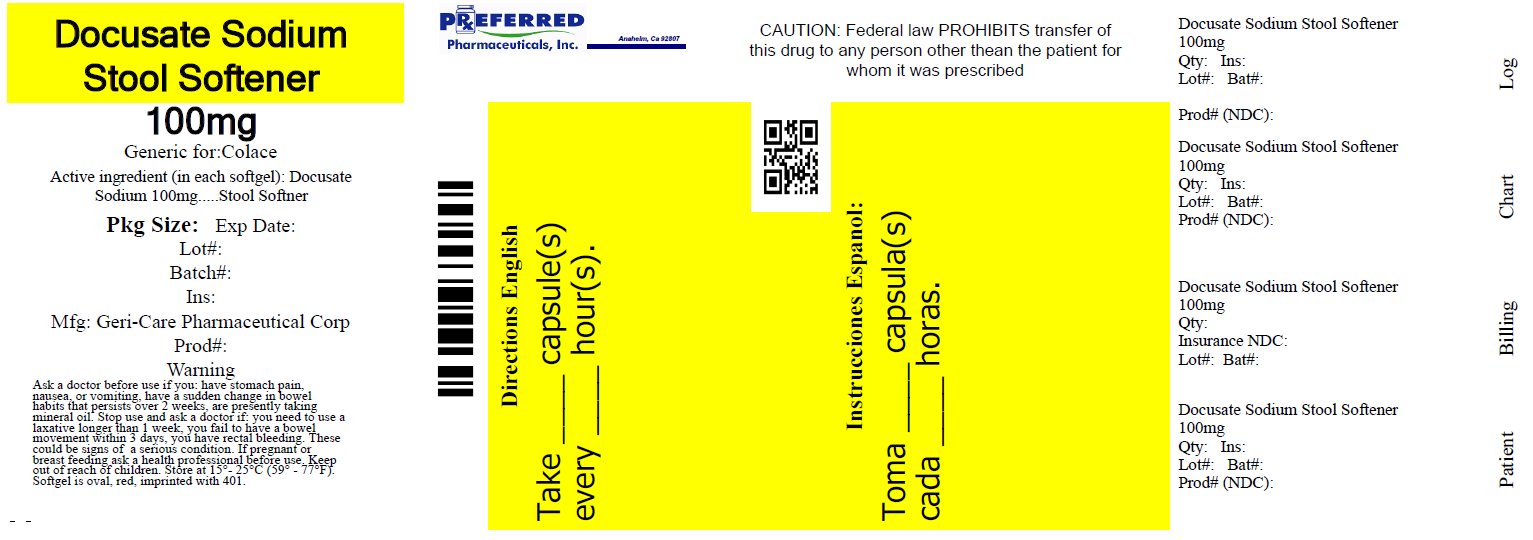
Stool Softener by Preferred Pharmaceuticals, Inc. GC 401
Stool Softener by
Drug Labeling and Warnings
Stool Softener by is a Otc medication manufactured, distributed, or labeled by Preferred Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
STOOL SOFTENER- docusate sodium capsule, liquid filled
Preferred Pharmaceuticals, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
GC 401
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 12 to 72 hours
Warnings
Ask a doctor before use if you
have stomach pain, nausea or vomiting
have a sudden change in bowel habits that persists over a period of 2 weeks
are presently taking mineral oil
Stop use and ask a doctor if
you need to use a laxative longer than 1 week
you have rectal bleeding or fail to have a bowel movement. These
could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Directions
adults and children 12 years and older: take 1-2 softgel daily until first bowel movement; 1 softgel daily thereafter, or as directed by doctor
children under 12: consult a doctor
do not exceed recommended dose
Other information
each softgel contains: sodium 5 mg. very low sodium
store at 15°C-25°C(59° F-77° F)
keep tightly closed
product from USA or Canada
Tamper Evident: Do not use if imprinted seal under cap is missing or broken.
- Bottle of 30 NDC: 68788-7744-03
Inactive ingredients
FD&C red #40, gelatin,
glycerin, edible ink, PEG, propylene glycol,
sorbitol sorbitan solution, water. Also contains
D&C yellow #10 or FD&C yellow #6 (sunset yellow).
NDC: 68788-7744
Repackaged by Preferred Pharmaceuticals, Inc.
| STOOL SOFTENER
docusate sodium capsule, liquid filled |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Preferred Pharmaceuticals, Inc. (791119022) |
| Registrant - Preferred Pharmaceuticals, Inc. (791119022) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Preferred Pharmaceuticals, Inc. | 791119022 | REPACK(68788-7744) | |