Active Ingredient
Ethyl Alcohol 75% (v/v)
Uses
- To help reduce bacteria on the skin.
Warnings
- For external use only.
- Do not dispose of wipes in flush toilets.
- Keep away from fire or flame.
Flammable
When using this product
- avoid contact with eyes.
- if contact occurs, rinse thoroughly with water.
Stop using and ask a doctor if
- irritation or redness develops and lasts.
Keep out of reach of children
- In case of accidental ingestion, get medical help or contact a poison control center immediately.
Directions
- Apply to hands or surfaces.
- No rinsing required.
- Discard after use.
Other Information
- Store below 110°F (43°C).
- May discolor certain fabrics or surfaces.
Inactive ingredients
Aqua (Water), Glycerin. Polyquaternium-37, Melaleuca Alternifolia (Tea Tree) Leaf Oil.
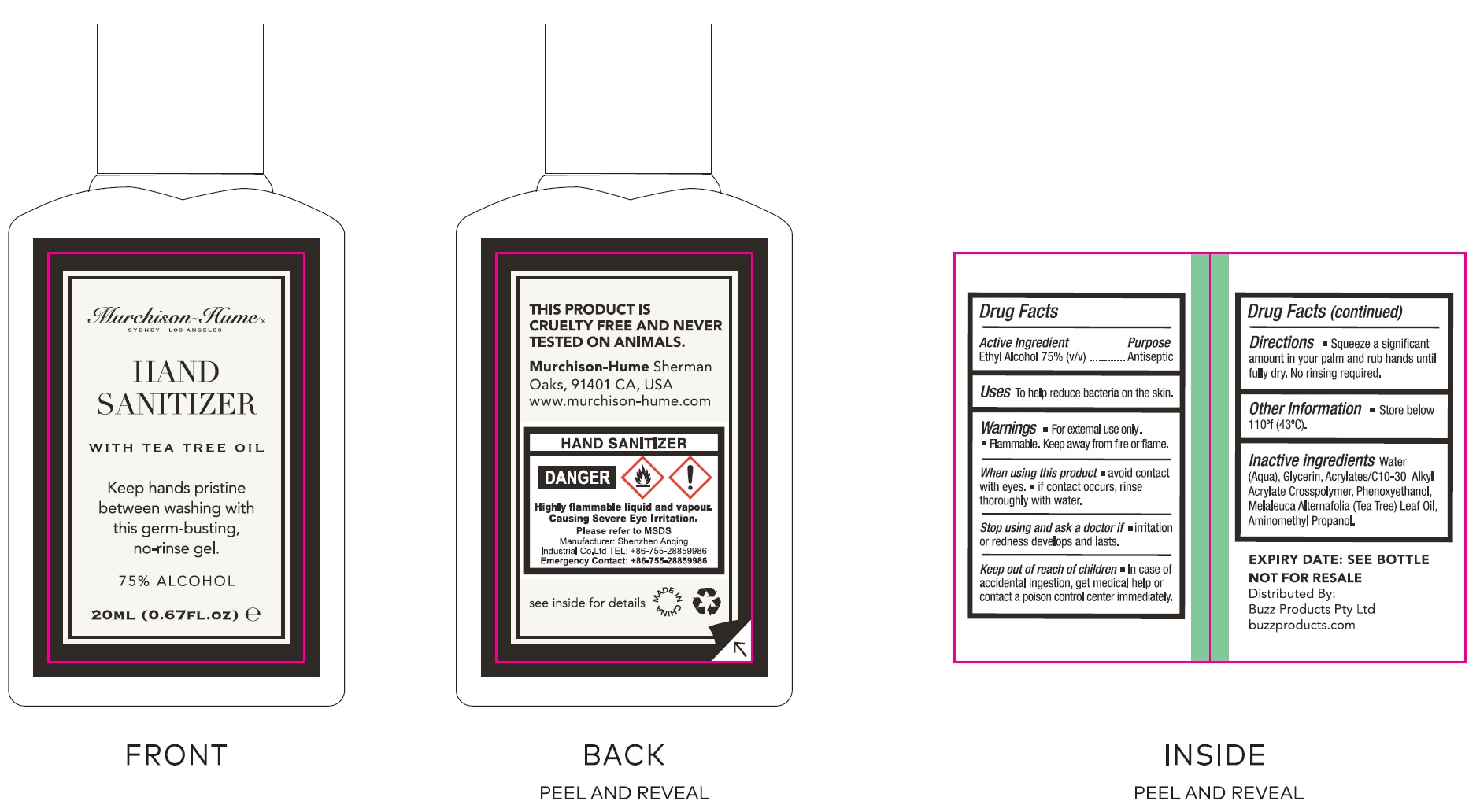
Active Ingredient
Ethyl Alcohol 75% (v/v)
Uses
To help reduce bacteria on the skin.
Warnings
- For external use only.
- Flammable. Keep away from fire or flame.
When using this product
- avoid contact with eyes.
- if contact occurs, rinse thoroughly with water.
Stop using and ask a doctor if
irritation or redness develops and lasts.
Keep out of reach of children
In case of accidental ingestion, get medical help or contact a poison control center immediately.
Directions
- Squeeze a significant amount in your palm and rub hands until full dry. No rinsing required.
Other Information
- Store below 110°f (43°C).
Inactive ingredients
Water (Aqua), Glycerin, Acrylates/C-10-30 Alkyl Acrylate Crosspolymer, Phenoxyethanol, Melaleuca Alternafolia (Tea Tree) Leaf Oil, Aminomethyl Propanol.
Kit Label:

Package Labeling:69491-303-00

Package Labeling:69491-301-00