AUSTEDO- deutetrabenazine tablet, coated AUSTEDO- deutetrabenazine kit
AUSTEDO XR by
Drug Labeling and Warnings
AUSTEDO XR by is a Prescription medication manufactured, distributed, or labeled by Teva Neuroscience, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use AUSTEDO safely and effectively. See full prescribing information for AUSTEDO.
AUSTEDO® (deutetrabenazine) tablets, for oral use
Initial U.S. Approval: 2017WARNING: DEPRESSION AND SUICIDALITY IN PATIENTS WITH HUNTINGTON’S DISEASE
See full prescribing information for complete boxed warning.
- Increases the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease (5.1)
- Balance risks of depression and suicidality with the clinical need for treatment of chorea when considering the use of AUSTEDO (5.1)
- Monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior (5.1)
- Inform patients, caregivers, and families of the risk of depression and suicidality and instruct to report behaviors of concern promptly to the treating physician (5.1)
- Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation (5.1)
- AUSTEDO is contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression (4, 5.1)
RECENT MAJOR CHANGES
Warnings and Precautions (5.6)
7/2019
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
Initial Dose
Recommended Dose
Maximum Dose
Chorea associated with Huntington’s disease
6 mg/day
6 mg– 48 mg/day
48 mg/day
Tardive dyskinesia
12 mg/day
12 mg– 48 mg/day
48 mg/day
- Titrate at weekly intervals by 6 mg per day based on reduction of chorea or tardive dyskinesia, and tolerability, up to a maximum recommended daily dosage of 48 mg (24 mg twice daily) (2.1)
- Administer total daily dosages of 12 mg or above in two divided doses (2.1)
- For patients at risk for QT prolongation, assess the QT interval before and after increasing the total dosage above 24 mg per day (2.1)
- Administer with food (2.1)
- Swallow tablets whole; do not chew, crush, or break (2.1)
- If switching patients from tetrabenazine, discontinue tetrabenazine and initiate AUSTEDO the following day. See full prescribing information for recommended conversion table (2.2)
- Maximum recommended dosage of AUSTEDO in poor CYP2D6 metabolizers is 36 mg per day (i.e., 18 mg twice daily) (2.4, 8.7)
DOSAGE FORMS AND STRENGTHS
Tablets: 6 mg, 9 mg, and 12 mg (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- QT Prolongation: May cause an increase in QT interval. Avoid use in patients with congenital long QT syndrome or with arrhythmias associated with a prolonged QT interval (5.3)
- Neuroleptic Malignant Syndrome (NMS): Discontinue if this occurs (5.4)
- Akathisia, agitation, restlessness, and parkinsonism: Reduce dose or discontinue if this occurs (5.5, 5.6)
- Sedation/somnolence: May impair the patient’s ability to drive or operate complex machinery (5.7)
ADVERSE REACTIONS
Most common adverse reactions (>8% of AUSTEDO-treated patients with Huntington’s disease and greater than placebo): somnolence, diarrhea, dry mouth, and fatigue (6.1)
Most common adverse reactions (that occurred in 4% of AUSTEDO-treated patients with tardive dyskinesia and greater than placebo): nasopharyngitis and insomnia (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Teva Pharmaceuticals at 1-888-483-8279 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
Pregnancy: Based on animal data, may cause fetal harm (8.1)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 10/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
RECENT MAJOR CHANGES
WARNING: DEPRESSION AND SUICIDALITY IN PATIENTS WITH HUNTINGTON’S DISEASE
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
2.2 Switching Patients from Tetrabenazine (XENAZINE®) to AUSTEDO
2.3 Dosage Adjustment with Strong CYP2D6 Inhibitors
2.4 Dosage Adjustment in Poor CYP2D6 Metabolizers
2.5 Discontinuation and Interruption of Treatment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Depression and Suicidality in Patients with Huntington’s Disease
5.2 Clinical Worsening and Adverse Events in Patients with Huntington’s Disease
5.3 QTc Prolongation
5.4 Neuroleptic Malignant Syndrome (NMS)
5.5 Akathisia, Agitation, and Restlessness
5.6 Parkinsonism
5.7 Sedation and Somnolence
5.8 Hyperprolactinemia
5.9 Binding to Melanin-Containing Tissues
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Strong CYP2D6 Inhibitors
7.2 Drugs that Cause QTc Prolongation
7.3 Reserpine
7.4 Monoamine Oxidase Inhibitors (MAOIs)
7.5 Neuroleptic Drugs
7.6 Alcohol or Other Sedating Drugs
7.7 Concomitant Tetrabenazine or Valbenazine
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Poor CYP2D6 Metabolizers
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Chorea Associated with Huntington’s Disease
14.2 Tardive Dyskinesia
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: DEPRESSION AND SUICIDALITY IN PATIENTS WITH HUNTINGTON’S DISEASE
AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Anyone considering the use of AUSTEDO must balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Patients, their caregivers, and families should be informed of the risk of depression and suicidality and should be instructed to report behaviors of concern promptly to the treating physician.
Particular caution should be exercised in treating patients with a history of depression or prior suicide attempts or ideation, which are increased in frequency in Huntington’s disease. AUSTEDO is contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression [see Contraindications (4) and Warnings and Precautions (5.1)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
The dose of AUSTEDO is determined individually for each patient based on reduction of chorea or tardive dyskinesia and tolerability. When first prescribed to patients who are not being switched from tetrabenazine (a related VMAT2 inhibitor), the recommended starting dose of AUSTEDO is 6 mg administered orally once daily for patients with Huntington’s disease and 12 mg per day (6 mg twice daily) for patients with tardive dyskinesia.
- The dose of AUSTEDO may be increased at weekly intervals in increments of 6 mg per day to a maximum recommended daily dosage of 48 mg.
- Administer total daily dosages of 12 mg or above in two divided doses.
- Administer AUSTEDO with food [see Clinical Pharmacology (12.3)].
- Swallow AUSTEDO whole. Do not chew, crush, or break tablets.
- For patients at risk for QT prolongation, assess the QT interval before and after increasing total AUSTEDO dosage above 24 mg per day [see Warnings and Precautions (5.3) and Drug Interactions (7.2)].
2.2 Switching Patients from Tetrabenazine (XENAZINE®) to AUSTEDO
Discontinue tetrabenazine (XENAZINE®) and initiate AUSTEDO the following day. The recommended initial dosing regimen of AUSTEDO in patients switching from tetrabenazine (XENAZINE®) to AUSTEDO is shown in Table 1.
Table 1: Recommended Initial Dosing Regimen when Switching from Tetrabenazine (XENAZINE®) to AUSTEDO Current tetrabenazine
daily dosageInitial regimen of
AUSTEDO12.5 mg
6 mg once daily
25 mg
6 mg twice daily
37.5 mg
9 mg twice daily
50 mg
12 mg twice daily
62.5 mg
15 mg twice daily
75 mg
18 mg twice daily
87.5 mg
21 mg twice daily
100 mg
24 mg twice daily
After patients are switched to AUSTEDO, the dose may be adjusted at weekly intervals [see Dosage and Administration (2.1)].
2.3 Dosage Adjustment with Strong CYP2D6 Inhibitors
In patients receiving strong CYP2D6 inhibitors (e.g., quinidine, antidepressants such as paroxetine, fluoxetine, and bupropion), the total daily dosage of AUSTEDO should not exceed 36 mg (maximum single dose of 18 mg) [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
2.4 Dosage Adjustment in Poor CYP2D6 Metabolizers
In patients who are poor CYP2D6 metabolizers, the total daily dosage of AUSTEDO should not exceed 36 mg (maximum single dose of 18 mg) [see Use in Specific Populations (8.7)].
2.5 Discontinuation and Interruption of Treatment
Treatment with AUSTEDO can be discontinued without tapering. Following treatment interruption of greater than one week, AUSTEDO therapy should be re-titrated when resumed. For treatment interruption of less than one week, treatment can be resumed at the previous maintenance dose without titration.
-
3 DOSAGE FORMS AND STRENGTHS
AUSTEDO tablets are available in the following strengths:
- The 6 mg tablets are round, purple-coated tablets, with “SD” over “6” printed in black ink on one side.
- The 9 mg tablets are round, blue-coated tablets, with “SD” over “9” printed in black ink on one side.
- The 12 mg tablets are round, beige-coated tablets, with “SD” over “12” printed in black ink on one side.
-
4 CONTRAINDICATIONS
AUSTEDO is contraindicated in patients:
- With Huntington’s disease who are suicidal, or have untreated or inadequately treated depression [see Warnings and Precautions (5.1)].
- With hepatic impairment [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
- Taking reserpine. At least 20 days should elapse after stopping reserpine before starting AUSTEDO [see Drug Interactions (7.3)].
- Taking monoamine oxidase inhibitors (MAOIs). AUSTEDO should not be used in combination with an MAOI, or within 14 days of discontinuing therapy with an MAOI [see Drug Interactions (7.4)].
- Taking tetrabenazine (XENAZINE®) or valbenazine [see Drug Interactions (7.7)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Depression and Suicidality in Patients with Huntington’s Disease
Patients with Huntington’s disease are at increased risk for depression, and suicidal ideation or behaviors (suicidality). AUSTEDO may increase the risk for suicidality in patients with Huntington’s disease.
In a 12-week, double-blind, placebo-controlled trial, suicidal ideation was reported by 2% of patients treated with AUSTEDO, compared to no patients on placebo; no suicide attempts and no completed suicides were reported. Depression was reported by 4% of patients treated with AUSTEDO.
When considering the use of AUSTEDO, the risk of suicidality should be balanced against the need for treatment of chorea. All patients treated with AUSTEDO should be observed for new or worsening depression or suicidality. If depression or suicidality does not resolve, consider discontinuing treatment with AUSTEDO.
Patients, their caregivers, and families should be informed of the risks of depression, worsening depression, and suicidality associated with AUSTEDO, and should be instructed to report behaviors of concern promptly to the treating physician. Patients with Huntington’s disease who express suicidal ideation should be evaluated immediately.
5.2 Clinical Worsening and Adverse Events in Patients with Huntington’s Disease
Huntington’s disease is a progressive disorder characterized by changes in mood, cognition, chorea, rigidity, and functional capacity over time. VMAT2 inhibitors, including AUSTEDO, may cause a worsening in mood, cognition, rigidity, and functional capacity.
Prescribers should periodically re-evaluate the need for AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects, including sedation/somnolence, depression and suicidality, parkinsonism, akathisia, restlessness, and cognitive decline. It may be difficult to distinguish between adverse reactions and progression of the underlying disease; decreasing the dose or stopping the drug may help the clinician to distinguish between the two possibilities. In some patients, the underlying chorea itself may improve over time, decreasing the need for AUSTEDO.
5.3 QTc Prolongation
Tetrabenazine, a closely related VMAT2 inhibitor, causes an increase (about 8 msec) in the corrected QT (QTc) interval.
A clinically relevant QT prolongation may occur in some patients treated with AUSTEDO who are CYP2D6 poor metabolizers or are co-administered a strong CYP2D6 inhibitor [see Clinical Pharmacology (12.2, 12.3)].
For patients who are CYP2D6 poor metabolizers or are taking a strong CYP2D6 inhibitor, dose reduction may be necessary [see Dosage and Administration (2.3, 2.4)]. The use of AUSTEDO in combination with other drugs that are known to prolong QTc may result in clinically significant QT prolongations [see Drug Interactions (7.2)].
For patients requiring AUSTEDO doses greater than 24 mg per day who are using AUSTEDO with other drugs known to prolong QTc, assess the QTc interval before and after increasing the dose of AUSTEDO or other medications that are known to prolong QTc.
AUSTEDO should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias. Certain circumstances may increase the risk of the occurrence of torsade de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval, including (1) bradycardia; (2) hypokalemia or hypomagnesemia; (3) concomitant use of other drugs that prolong the QTc interval; and (4) presence of congenital prolongation of the QT interval.
5.4 Neuroleptic Malignant Syndrome (NMS)
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with drugs that reduce dopaminergic transmission. While NMS has not been observed in patients receiving AUSTEDO, it has been observed in patients receiving tetrabenazine (a closely related VMAT2 inhibitor). Clinicians should be alerted to the signs and symptoms associated with NMS. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatinine phosphokinase, myoglobinuria, rhabdomyolysis, and acute renal failure. The diagnosis of NMS can be complicated; other serious medical illness (e.g., pneumonia, systemic infection) and untreated or inadequately treated extrapyramidal disorders can present with similar signs and symptoms. Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology.
The management of NMS should include (1) immediate discontinuation of AUSTEDO; (2) intensive symptomatic treatment and medical monitoring; and (3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for NMS.
Recurrence of NMS has been reported with resumption of drug therapy. If treatment with AUSTEDO is needed after recovery from NMS, patients should be monitored for signs of recurrence.
5.5 Akathisia, Agitation, and Restlessness
AUSTEDO may increase the risk of akathisia, agitation, and restlessness in patients with Huntington’s disease and tardive dyskinesia.
In a 12-week, double-blind, placebo-controlled trial in Huntington’s disease patients, akathisia, agitation, or restlessness was reported by 4% of patients treated with AUSTEDO, compared to 2% of patients on placebo; in patients with tardive dyskinesia, 2% of patients treated with AUSTEDO and 1% of patients on placebo experienced these events.
Patients receiving AUSTEDO should be monitored for signs and symptoms of restlessness and agitation, as these may be indicators of developing akathisia. If a patient develops akathisia during treatment with AUSTEDO, the AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
5.6 Parkinsonism
AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors.
Because rigidity can develop as part of the underlying disease process in Huntington’s disease, it may be difficult to distinguish between potential drug-induced parkinsonism and progression of underlying Huntington’s disease. Drug-induced parkinsonism has the potential to cause more functional disability than untreated chorea for some patients with Huntington’s disease.
Postmarketing cases of parkinsonism in patients treated with AUSTEDO for tardive dyskinesia have been reported. Signs and symptoms in reported cases have included bradykinesia, gait disturbances, which led to falls in some cases, and the emergence or worsening of tremor. In most cases, the development of parkinsonism occurred within the first two weeks after starting or increasing the dose of AUSTEDO. In cases in which follow-up clinical information was available, parkinsonism was reported to resolve following discontinuation of AUSTEDO therapy.If a patient develops parkinsonism during treatment with AUSTEDO, the AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
5.7 Sedation and Somnolence
Sedation is a common dose-limiting adverse reaction of AUSTEDO. In a 12-week, double-blind, placebo-controlled trial examining patients with Huntington’s disease, 11% of AUSTEDO-treated patients reported somnolence compared with 4% of patients on placebo and 9% of AUSTEDO-treated patients reported fatigue compared with 4% of placebo-treated patients.
Patients should not perform activities requiring mental alertness to maintain the safety of themselves or others, such as operating a motor vehicle or operating hazardous machinery, until they are on a maintenance dose of AUSTEDO and know how the drug affects them.
5.8 Hyperprolactinemia
Serum prolactin levels were not evaluated in the AUSTEDO development program. Tetrabenazine, a closely related VMAT2 inhibitor, elevates serum prolactin concentrations in humans. Following administration of 25 mg of tetrabenazine to healthy volunteers, peak plasma prolactin levels increased 4- to 5-fold.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if AUSTEDO is being considered for a patient with previously detected breast cancer. Although amenorrhea, galactorrhea, gynecomastia, and impotence can be caused by elevated serum prolactin concentrations, the clinical significance of elevated serum prolactin concentrations for most patients is unknown.
Chronic increase in serum prolactin levels (although not evaluated in the AUSTEDO or tetrabenazine development programs) has been associated with low levels of estrogen and increased risk of osteoporosis. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO.
5.9 Binding to Melanin-Containing Tissues
Since deutetrabenazine or its metabolites bind to melanin-containing tissues, it could accumulate in these tissues over time. This raises the possibility that AUSTEDO may cause toxicity in these tissues after extended use. Neither ophthalmologic nor microscopic examination of the eye has been conducted in the chronic toxicity studies in a pigmented species such as dogs. Ophthalmologic monitoring in humans was inadequate to exclude the possibility of injury occurring after long-term exposure.
The clinical relevance of deutetrabenazine’s binding to melanin-containing tissues is unknown. Although there are no specific recommendations for periodic ophthalmologic monitoring, prescribers should be aware of the possibility of long-term ophthalmologic effects [see Clinical Pharmacology (12.2)].
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Depression and Suicidality in Patients with Huntington’s disease [see Warnings and Precautions (5.1)]
- QTc Prolongation [see Warnings and Precautions (5.3)]
- Neuroleptic Malignant Syndrome (NMS) [see Warnings and Precautions (5.4)]
- Akathisia, Agitation, and Restlessness [see Warnings and Precautions (5.5)]
- Parkinsonism [see Warnings and Precautions (5.6)]
- Sedation and Somnolence [see Warnings and Precautions (5.7)]
- Hyperprolactinemia [see Warnings and Precautions (5.8)]
- Binding to Melanin-Containing Tissues [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Patients with Huntington’s Disease
Study 1 was a randomized, 12-week, placebo-controlled study in patients with chorea associated with Huntington’s disease. A total of 45 patients received AUSTEDO, and 45 patients received placebo. Patients ranged in age between 23 and 74 years (mean 54 years); 56% were male, and 92% were Caucasian. The most common adverse reactions occurring in greater than 8% of AUSTEDO-treated patients were somnolence, diarrhea, dry mouth, and fatigue. Adverse reactions occurring in 4% or more of patients treated with AUSTEDO, and with a greater incidence than in patients on placebo, are summarized in Table 2.
Table 2: Adverse Reactions in Patients with Huntington’s Disease (Study 1) Experienced by at Least 4% of Patients on AUSTEDO and with a Greater Incidence than on Placebo Adverse Reaction AUSTEDO
(N = 45)
%Placebo
(N = 45)
%Somnolence
11
4
Diarrhea
9
0
Dry mouth
9
7
Fatigue
9
4
Urinary tract infection
7
2
Insomnia
7
4
Anxiety
4
2
Constipation
4
2
Contusion
4
2
One or more adverse reactions resulted in a reduction of the dose of study medication in 7% of patients in Study 1. The most common adverse reaction resulting in dose reduction in patients receiving AUSTEDO was dizziness (4%).
Agitation led to discontinuation in 2% of patients treated with AUSTEDO in Study 1.
Patients with Tardive Dyskinesia
The data described below reflect 410 tardive dyskinesia patients participating in clinical trials. AUSTEDO was studied primarily in two 12-week, placebo-controlled trials (fixed dose, dose escalation). The population was 18 to 80 years of age, and had tardive dyskinesia and had concurrent diagnoses of mood disorder (33%) or schizophrenia/schizoaffective disorder (63%). In these studies, AUSTEDO was administered in doses ranging from 12-48 mg per day. All patients continued on previous stable regimens of antipsychotics; 71% and 14% respective atypical and typical antipsychotic medications at study entry.
The most common adverse reactions occurring in greater than 3% of AUSTEDO-treated patients and greater than placebo were nasopharyngitis and insomnia. The adverse reactions occurring in >2% or more patients treated with AUSTEDO (12-48 mg per day) and greater than in placebo patients in two double-blind, placebo-controlled studies in patients with tardive dyskinesia (Study 1 and Study 2) are summarized in Table 3.
Table 3: Adverse Reactions in 2 Placebo-Controlled Tardive Dyskinesia Studies (Study 1 and Study 2) of 12-week Treatment on AUSTEDO Reported in at Least 2% of Patients and Greater than Placebo Preferred Term
AUSTEDO
(N=279)
(%)Placebo
(N=131)
(%)Nasopharyngitis
4
2
Insomnia
4
1
Depression/ Dysthymic disorder
2
1
Akathisia/Agitation/Restlessness
2
1
One or more adverse reactions resulted in a reduction of the dose of study medication in 4% of AUSTEDO-treated patients and in 2% of placebo-treated patients.
-
7 DRUG INTERACTIONS
7.1 Strong CYP2D6 Inhibitors
A reduction in AUSTEDO dose may be necessary when adding a strong CYP2D6 inhibitor in patients maintained on a stable dose of AUSTEDO. Concomitant use of strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine, quinidine, bupropion) has been shown to increase the systemic exposure to the active dihydro-metabolites of deutetrabenazine by approximately 3-fold. The daily dose of AUSTEDO should not exceed 36 mg per day, and the maximum single dose of AUSTEDO should not exceed 18 mg in patients taking strong CYP2D6 inhibitors [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
7.2 Drugs that Cause QTc Prolongation
Tetrabenazine, a closely related VMAT2 inhibitor, may cause an increase in the corrected QT (QTc) interval. Clinically relevant QT prolongation may also occur with AUSTEDO [see Warnings and Precautions (5.3), Clinical Pharmacology (12.2)].
For patients requiring AUSTEDO doses above 24 mg per day, who are using AUSTEDO in combination with other drugs known to prolong QTc, assess the QTc interval before and after increasing the dose of AUSTEDO or other medications that are known to prolong QTc. Drugs known to prolong QTc include antipsychotic medications (e.g., chlorpromazine, haloperidol, thioridazine, ziprasidone), antibiotics (e.g., moxifloxacin), Class 1A (e.g., quinidine, procainamide), and Class III (e.g., amiodarone, sotalol) antiarrhythmic medications.
7.3 Reserpine
Reserpine binds irreversibly to VMAT2 and the duration of its effect is several days. Prescribers should wait for chorea or dyskinesia to reemerge before administering AUSTEDO to help reduce the risk of overdosage and major depletion of serotonin and norepinephrine in the central nervous system. At least 20 days should elapse after stopping reserpine before starting AUSTEDO. AUSTEDO and reserpine should not be used concomitantly [see Contraindications (4)].
7.4 Monoamine Oxidase Inhibitors (MAOIs)
AUSTEDO is contraindicated in patients taking MAOIs. AUSTEDO should not be used in combination with an MAOI, or within 14 days of discontinuing therapy with an MAOI [see Contraindications (4)].
7.5 Neuroleptic Drugs
The risk of parkinsonism, NMS, and akathisia may be increased by concomitant use of AUSTEDO and dopamine antagonists or antipsychotics.
7.6 Alcohol or Other Sedating Drugs
Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence [see Warnings and Precautions (5.7)].
7.7 Concomitant Tetrabenazine or Valbenazine
AUSTEDO is contraindicated in patients currently taking tetrabenazine or valbenazine. AUSTEDO may be initiated the day following discontinuation of tetrabenazine [see Dosage and Administration (2.2)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate data on the developmental risk associated with the use of AUSTEDO in pregnant women. Administration of deutetrabenazine to rats during organogenesis produced no clear adverse effect on embryofetal development. However, administration of tetrabenazine to rats throughout pregnancy and lactation resulted in an increase in stillbirths and postnatal offspring mortality [see Data].
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
Data
Animal Data
Oral administration of deutetrabenazine (5, 10, or 30 mg/kg/day) or tetrabenazine (30 mg/kg/day) to pregnant rats during organogenesis had no clear effect on embryofetal development. The highest dose tested was 6 times the maximum recommended human dose of 48 mg/day, on a body surface area (mg/m2) basis.
The effects of deutetrabenazine when administered during organogenesis to rabbits or during pregnancy and lactation to rats have not been assessed.
Tetrabenazine had no effects on embryofetal development when administered to pregnant rabbits during the period of organogenesis at oral doses up to 60 mg/kg/day. When tetrabenazine was administered to female rats (doses of 5, 15, and 30 mg/kg/day) from the beginning of organogenesis through the lactation period, an increase in stillbirths and offspring postnatal mortality was observed at 15 and 30 mg/kg/day, and delayed pup maturation was observed at all doses.
8.2 Lactation
Risk Summary
There are no data on the presence of deutetrabenazine or its metabolites in human milk, the effects on the breastfed infant, or the effects of the drug on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for AUSTEDO and any potential adverse effects on the breastfed infant from AUSTEDO or from the underlying maternal condition.
8.5 Geriatric Use
Clinical studies of AUSTEDO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of hepatic, renal, and cardiac dysfunction, and of concomitant disease or other drug therapy.
8.6 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of deutetrabenazine and its primary metabolites has not been studied; however, in a clinical study conducted with tetrabenazine, a closely related VMAT2 inhibitor, there was a large increase in exposure to tetrabenazine and its active metabolites in patients with hepatic impairment. The clinical significance of this increased exposure has not been assessed, but because of concerns for a greater risk for serious adverse reactions, the use of AUSTEDO in patients with hepatic impairment is contraindicated [see Contraindications (4), Clinical Pharmacology (12.3)].
8.7 Poor CYP2D6 Metabolizers
Although the pharmacokinetics of deutetrabenazine and its metabolites have not been systematically evaluated in patients who do not express the drug metabolizing enzyme, it is likely that the exposure to α-HTBZ and β-HTBZ would be increased similarly to taking a strong CYP2D6 inhibitor (approximately 3-fold). In patients who are CYP2D6 poor metabolizers, the daily dose of AUSTEDO should not exceed 36 mg (maximum single dose of 18 mg) [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Overdoses ranging from 100 mg to 1 g have been reported in the literature with tetrabenazine, a closely related VMAT2 inhibitor. The following adverse reactions occurred with overdosing: acute dystonia, oculogyric crisis, nausea and vomiting, sweating, sedation, hypotension, confusion, diarrhea, hallucinations, rubor, and tremor.
Treatment should consist of those general measures employed in the management of overdosage with any central nervous system-active drug. General supportive and symptomatic measures are recommended. Cardiac rhythm and vital signs should be monitored. In managing overdosage, the possibility of multiple drug involvement should always be considered. The physician should consider contacting a poison control center on the treatment of any overdose. Telephone numbers for certified poison control centers are listed on the American Association of Poison Control Centers website www.aapcc.org.
-
11 DESCRIPTION
AUSTEDO (deutetrabenazine) is a vesicular monoamine transporter 2 (VMAT2) inhibitor for oral administration. The molecular weight of deutetrabenazine is 323.46; the pKa is 6.31. Deutetrabenazine is a hexahydro-dimethoxybenzoquinolizine derivative and has the following chemical name: (RR, SS)-1, 3, 4, 6, 7, 11b-hexahydro-9, 10-di(methoxy-d3)-3-(2-methylpropyl)-2H-benzo[a]quinolizin-2-one.
The molecular formula for deutetrabenazine is C19H21D6NO3. Deutetrabenazine is a racemic mixture containing the following structures:
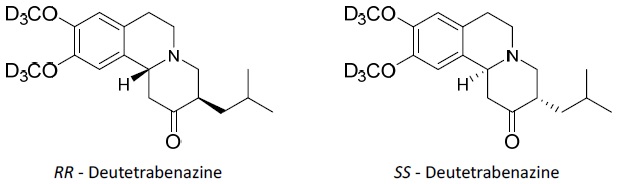
Deutetrabenazine is a white to slightly yellow crystalline powder that is sparingly soluble in water and soluble in ethanol.
AUSTEDO tablets contain 6 mg, 9 mg, or 12 mg deutetrabenazine, and the following inactive ingredients: ammonium hydroxide, black iron oxide, n-butyl alcohol, butylated hydroxyanisole, butylated hydroxytoluene, magnesium stearate, mannitol, microcrystalline cellulose, polyethylene glycol, polyethylene oxide, polysorbate 80, polyvinyl alcohol, povidone, propylene glycol, shellac, talc, titanium dioxide, and FD&C blue #2 lake. The 6 mg tablets also contain FD&C red #40 lake. The 12 mg tablets also contain FD&C yellow #6 lake.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The precise mechanism by which deutetrabenazine exerts its effects in the treatment of tardive dyskinesia and chorea in patients with Huntington’s disease is unknown but is believed to be related to its effect as a reversible depletor of monoamines (such as dopamine, serotonin, norepinephrine, and histamine) from nerve terminals. The major circulating metabolites (α-dihydrotetrabenazine [HTBZ] and β-HTBZ) of deutetrabenazine, are reversible inhibitors of VMAT2, resulting in decreased uptake of monoamines into synaptic vesicles and depletion of monoamine stores.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of a single 12-mg or 24-mg dose of AUSTEDO on the QT interval was studied in a randomized, double-blind, placebo-controlled crossover study in healthy male and female subjects with moxifloxacin as a positive control. At 24 mg, AUSTEDO caused an approximately 4.5 msec mean increase in QTc (90% CI: 2.4, 6.5 msec). Effects at higher exposures to AUSTEDO or its metabolites have not been evaluated.
Melanin Binding
Deutetrabenazine or its metabolites bind to melanin-containing tissues (i.e., eye, skin, fur) in pigmented rats. After a single oral dose of radiolabeled deutetrabenazine, radioactivity was still detected in eye and fur at 35 days following dosing [see Warnings and Precautions (5.9)].
12.3 Pharmacokinetics
After oral dosing up to 25 mg, plasma concentrations of deutetrabenazine are generally below the limit of detection because of the extensive hepatic metabolism of deutetrabenazine to the active deuterated dihydro metabolites (HTBZ), α-HTBZ and β-HTBZ. Linear dose dependence of Cmax and AUC was observed for the active metabolites following single or multiple doses of deutetrabenazine (6 mg to 24 mg and 7.5 mg twice daily to 22.5 mg twice daily).
Absorption
Following oral administration of deutetrabenazine, the extent of absorption is at least 80%.
Plasma concentrations of deutetrabenazine are generally below the limit of detection after oral dosing. Peak plasma concentrations (Cmax) of deuterated α-HTBZ and β-HTBZ are reached within 3 to 4 hours after dosing.
Effect of Food
The effects of food on the bioavailability of AUSTEDO were studied in subjects administered a single dose with and without food. Food had no effect on the area under the plasma concentration-time curve (AUC) of α-HTBZ or β-HTBZ, although Cmax was increased by approximately 50% in the presence of food [see Dosage and Administration (2.1)].
Distribution
The median volume of distribution (Vc/F) of the α-HTBZ, and the β-HTBZ metabolites of AUSTEDO are approximately 500 L and 730 L, respectively.
Results of PET-scan studies in humans show that following intravenous injection of 11C-labeled tetrabenazine or α-HTBZ, radioactivity is rapidly distributed to the brain, with the highest binding in the striatum and lowest binding in the cortex.
The in vitro protein binding of tetrabenazine, α-HTBZ, and β-HTBZ was examined in human plasma for concentrations ranging from 50 to 200 ng/mL. Tetrabenazine binding ranged from 82% to 85%, α-HTBZ binding ranged from 60% to 68%, and β-HTBZ binding ranged from 59% to 63%.
Elimination
AUSTEDO is primarily renally eliminated in the form of metabolites.
The half-life of total (α+β)-HTBZ from deutetrabenazine is approximately 9 to 10 hours.
The median clearance values (CL/F) of the α-HTBZ, and the β-HTBZ metabolites of AUSTEDO are approximately 47 L/hour and 70 L/hour, respectively, in the Huntington’s disease patient population.
Metabolism
In vitro experiments in human liver microsomes demonstrate that deutetrabenazine is extensively biotransformed, mainly by carbonyl reductase, to its major active metabolites, α-HTBZ and β-HTBZ, which are subsequently metabolized primarily by CYP2D6, with minor contributions of CYP1A2 and CYP3A4/5, to form several minor metabolites.
Excretion
In a mass balance study in 6 healthy subjects, 75% to 86% of the deutetrabenazine dose was excreted in the urine, and fecal recovery accounted for 8% to 11% of the dose. Urinary excretion of the α-HTBZ and β-HTBZ metabolites from deutetrabenazine each accounted for less than 10% of the administered dose. Sulfate and glucuronide conjugates of the α-HTBZ and β-HTBZ metabolites of deutetrabenazine, as well as products of oxidative metabolism, accounted for the majority of metabolites in the urine.
Specific Populations
Male and Female Patients
There is no apparent effect of gender on the pharmacokinetics of α-HTBZ and β‑HTBZ of deutetrabenazine.
Patients with Renal Impairment
No clinical studies have been conducted to assess the effect of renal impairment on the PK of AUSTEDO.
Patients with Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of deutetrabenazine and its primary metabolites has not been studied. However, in a clinical study conducted to assess the effect of hepatic impairment on the pharmacokinetics of tetrabenazine, a closely related VMAT2 inhibitor, the exposure to α-HTBZ and β-HTBZ was up to 40% greater in patients with hepatic impairment, and the mean tetrabenazine Cmax in patients with hepatic impairment was up to 190-fold higher than in healthy subjects [see Contraindications (4), Use in Specific Populations (8.6)].
Poor CYP2D6 Metabolizers
Although the pharmacokinetics of deutetrabenazine and its metabolites have not been systematically evaluated in patients who do not express the drug metabolizing enzyme CYP2D6, it is likely that the exposure to α-HTBZ and β-HTBZ would be increased similarly to taking strong CYP2D6 inhibitors (approximately 3-fold) [see Dosage and Administration (2.4), Drug Interactions (7.1)].
Drug Interaction StudiesDeutetrabenazine, α-HTBZ, and β-HTBZ have not been evaluated in in vitro studies for induction or inhibition of CYP enzymes or interaction with P-glycoprotein. The results of in vitro studies of tetrabenazine do not suggest that tetrabenazine or its α-HTBZ or β-HTBZ metabolites are likely to result in clinically significant inhibition of CYP2D6, CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2E1, or CYP3A. In vitro studies suggest that neither tetrabenazine nor its α-HTBZ or β-HTBZ metabolites are likely to result in clinically significant induction of CYP1A2, CYP3A4, CYP2B6, CYP2C8, CYP2C9, or CYP2C19. Neither tetrabenazine nor its α-HTBZ or β-HTBZ metabolites are likely to be a substrate or inhibitor of P-glycoprotein at clinically relevant concentrations in vivo.
The deutetrabenazine metabolites, 2-methylpropanoic acid of β-HTBZ (M1) and monohydroxy tetrabenazine (M4), have been evaluated in a panel of in vitro drug-drug interaction studies; the results indicate that M1/M4 are not expected to cause clinically relevant drug interactions.
CYP2D6 Inhibitors
In vitro studies indicate that the α-HTBZ and β-HTBZ metabolites of deutetrabenazine are substrates for CYP2D6. The effect of CYP2D6 inhibition on the pharmacokinetics of deutetrabenazine and its metabolites was studied in 24 healthy subjects following a single 22.5 mg dose of deutetrabenazine given after 8 days of administration of the strong CYP2D6 inhibitor paroxetine 20 mg daily. In the presence of paroxetine, systemic exposure (AUCinf) of α-HTBZ was 1.9-fold higher and β-HTBZ was 6.5-fold higher, resulting in approximately 3-fold increase in AUCinf for total (α+β)-HTBZ. Paroxetine decreased the clearance of α-HTBZ and β-HTBZ metabolites of AUSTEDO with corresponding increases in mean half-life of approximately 1.5-fold and 2.7-fold, respectively. In the presence of paroxetine, Cmax of α-HTBZ and β-HTBZ were 1.2-fold and 2.2-fold higher, respectively.
The effect of moderate or weak CYP2D6 inhibitors such as duloxetine, terbinafine, amiodarone, or sertraline on the exposure of deutetrabenazine and its metabolites has not been evaluated.
Digoxin
AUSTEDO was not evaluated for interaction with digoxin. Digoxin is a substrate for P-glycoprotein. A study in healthy subjects showed that tetrabenazine (25 mg twice daily for 3 days) did not affect the bioavailability of digoxin, suggesting that at this dose, tetrabenazine does not affect P‑glycoprotein in the intestinal tract. In vitro studies also do not suggest that tetrabenazine or its metabolites are P-glycoprotein inhibitors.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No carcinogenicity studies were performed with deutetrabenazine.
No increase in tumors was observed in p53+/– transgenic mice treated orally with tetrabenazine at doses of 0, 5, 15, and 30 mg/kg/day for 26 weeks.
Mutagenesis
Deutetrabenazine and its deuterated α-HTBZ and β-HTBZ metabolites were negative in in vitro (bacterial reverse mutation and chromosome aberration in human peripheral blood lymphocytes) assays in the presence or absence of metabolic activation and in the in vivo micronucleus assay in mice.
Impairment of Fertility
The effects of deutetrabenazine on fertility have not been evaluated. Oral administration of deutetrabenazine (doses of 5, 10, or 30 mg/kg/day) to female rats for 3 months resulted in estrous cycle disruption at all doses; the lowest dose tested was similar to the maximum recommended human dose (48 mg/day) on a body surface area (mg/m2) basis.
Oral administration of tetrabenazine (doses of 5, 15, or 30 mg/kg/day) to female rats prior to and throughout mating, and continuing through day 7 of gestation, resulted in disrupted estrous cyclicity at doses greater than 5 mg/kg/day. No effects on mating and fertility indices or sperm parameters (motility, count, density) were observed when males were treated orally with tetrabenazine at doses of 5, 15 or 30 mg/kg/day prior to and throughout mating with untreated females.
-
14 CLINICAL STUDIES
14.1 Chorea Associated with Huntington’s Disease
Double-Blind, Placebo-Controlled Study
The efficacy of AUSTEDO as a treatment for chorea associated with Huntington's disease was established primarily in Study 1, a randomized, double-blind, placebo-controlled, multi-center trial conducted in 90 ambulatory patients with manifest chorea associated with Huntington’s disease. The diagnosis of Huntington’s disease was based on family history, neurological exam, and genetic testing. Treatment duration was 12 weeks, including an 8-week dose titration period and a 4-week maintenance period, followed by a 1-week washout. Patients were not blinded to discontinuation. AUSTEDO was started at 6 mg per day and titrated upward, at weekly intervals, in 6 mg increments until satisfactory treatment of chorea was achieved, intolerable side effects occurred, or until a maximal dose of 48 mg per day was reached. The primary efficacy endpoint was the Total Maximal Chorea Score, an item of the Unified Huntington's Disease Rating Scale (UHDRS). On this scale, chorea is rated from 0 to 4 (with 0 representing no chorea) for 7 different parts of the body. The total score ranges from 0 to 28.
Of the 90 patients enrolled, 87 patients completed the study. The mean age was 54 (range 23 to 74). Patients were 56% male and 92% Caucasian. The mean dose after titration was 40 mg per day. Table 4 and Figure 1 summarize the effects of AUSTEDO on chorea based on the Total Maximal Chorea Score. Total Maximal Chorea Scores for patients receiving AUSTEDO improved by approximately 4.4 units from baseline to the maintenance period (average of Week 9 and Week 12), compared to approximately 1.9 units in the placebo group. The treatment effect of -2.5 units was statistically significant (p<0.0001). The Maintenance Endpoint is the mean of the Total Maximal Chorea Scores for the Week 9 and Week 12 visits. At the Week 13 follow-up visit (1 week after discontinuation of the study medication), the Total Maximal Chorea Scores of patients who had received AUSTEDO returned to baseline (Figure 1).
Table 4: Change from Baseline to Maintenance Therapy in Total Maximal Chorea (TMC)* Score in Patients with Huntington’s Disease Treated with AUSTEDO in Study 1 Motor Endpoint AUSTEDO
N = 45Placebo
N = 45p value Change in Total Chorea Score* from Baseline to Maintenance Therapy†
-4.4
-1.9
<0.0001
*TMC is a subscale of the Unified Huntington's Disease Rating Scale (UHDRS)
†Primary efficacy endpointFigure 1: Total Maximal Chorea Score Over Time in Study 1
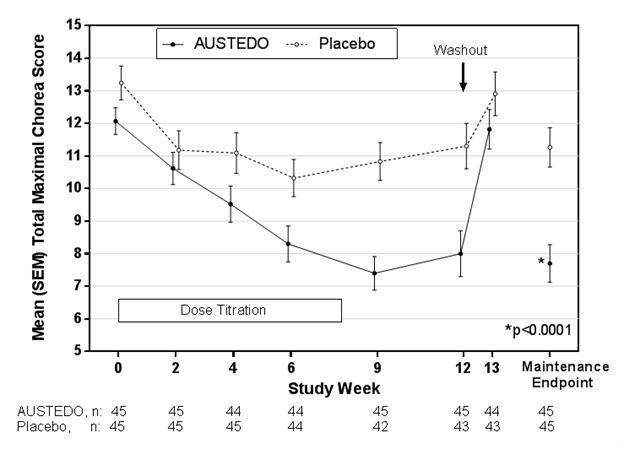
Figure 2: Distribution of the Change in Total Maximal Chorea Scores in Study 1
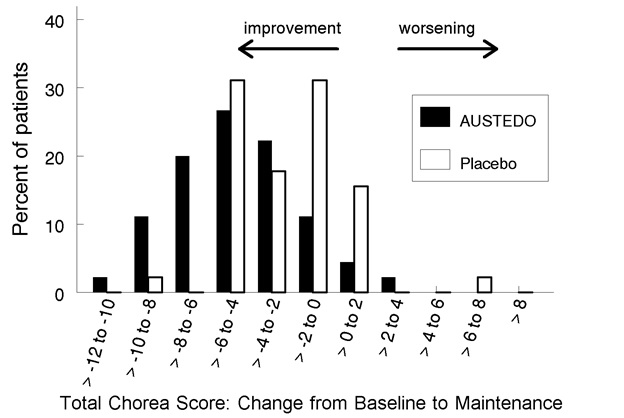
Figure 2 shows the distribution of values for the change in Total Maximal Chorea Score in Study 1. Negative values indicate a reduction in chorea and positive numbers indicate an increase in chorea.
A patient-rated global impression of change assessed how patients rated their overall Huntington’s disease symptoms. Fifty-one percent of patients treated with AUSTEDO rated their symptoms as “Much Improved” or “Very Much Improved” at the end of treatment, compared to 20% of placebo-treated patients.
In a physician-rated clinical global impression of change, 42% percent of patients treated with AUSTEDO rated their symptoms as “Much Improved” or “Very Much Improved” at the end of treatment compared to 13% of placebo-treated patients.
14.2 Tardive Dyskinesia
The efficacy of AUSTEDO in the treatment for tardive dyskinesia was established in two 12‑week, randomized, double-blind, placebo-controlled, multi-center trials conducted in 335 adult ambulatory patients with tardive dyskinesia caused by use of dopamine receptor antagonists. Patients had a history of using a dopamine receptor antagonist (antipsychotics, metoclopramide) for at least 3 months (or 1 month in patients 60 years of age and older). Concurrent diagnoses included schizophrenia/schizoaffective disorder (62%) and mood disorder (33%). With respect to concurrent antipsychotic use, 64% of patients were receiving atypical antipsychotics, 12% were receiving typical or combination antipsychotics, and 24% were not receiving antipsychotics.
The Abnormal Involuntary Movement Scale (AIMS) was the primary efficacy measure for the assessment of tardive dyskinesia severity. The AIMS is a 12-item scale; items 1 to 7 assess the severity of involuntary movements across body regions and these items were used in this study. Each of the 7 items was scored on a 0 to 4 scale, rated as: 0=not present; 1=minimal, may be extreme normal (abnormal movements occur infrequently and/or are difficult to detect); 2=mild (abnormal movements occur infrequently and are easy to detect); 3=moderate (abnormal movements occur frequently and are easy to detect) or 4 =severe (abnormal movements occur almost continuously and/or of extreme intensity). The AIMS total score (sum of items 1 to 7) could thus range from 0 to 28, with a decrease in score indicating improvement.
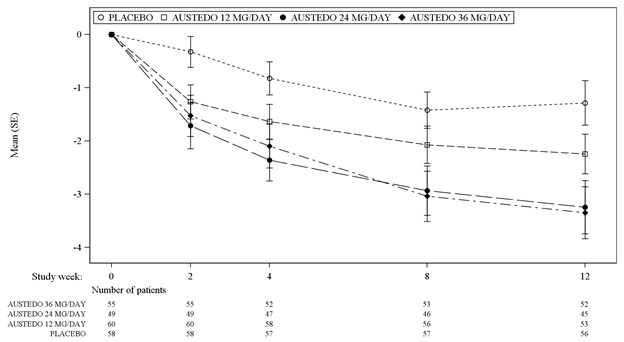
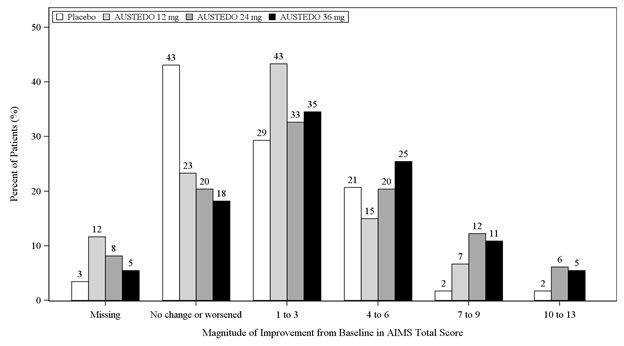
In Study 1, a 12-week, placebo-controlled, fixed-dose trial, adults with tardive dyskinesia were randomized 1:1:1:1 to 12 mg AUSTEDO, 24 mg AUSTEDO, 36 mg AUSTEDO, or placebo. Treatment duration included a 4-week dose escalation period and an 8-week maintenance period followed by a 1-week washout. The dose of AUSTEDO was started at 12 mg per day and increased at weekly intervals in 6 mg/day increments to a dose target of 12 mg, 24 mg or 36 mg per day. The population (n= 222) was 21 to 81 years old (mean 57 years), 48% male, and 79% Caucasian. In Study 1, the AIMS total score for patients receiving AUSTEDO demonstrated statistically significant improvement, from baseline to Week 12, of 3.3 and 3.2 units for the 36 mg and 24 mg arms, respectively, compared with 1.4 units in placebo (Study 1 in Table 5). The improvements on the AIMS total score over the course of the study are displayed in Figure 3. Data did not suggest substantial differences in efficacy across various demographic groups. The treatment response rate distribution, based on magnitude of AIMS total score from baseline to week 12 is displayed in Figure 4.
The mean changes in the AIMS total score by visit are shown in Figure 3.
In Study 2, a 12-week, placebo-controlled, flexible-dose trial, adults with tardive dyskinesia (n=113) received daily doses of placebo or AUSTEDO, starting at 12 mg per day with increases allowed in 6-mg increments at 1-week intervals until satisfactory control of dyskinesia was achieved, until intolerable side effects occurred, or until a maximal dose of 48 mg per day was reached. Treatment duration included a 6-week dose titration period and a 6-week maintenance period followed by a 1-week washout. The population was 25 to 75 years old (mean 55 years), 48% male, and 70% Caucasian. Patients were titrated to an optimal dose over 6 weeks. The average dose of AUSTEDO after treatment was 38.3 mg per day. There was no evidence suggesting substantial differences in efficacy across various demographic groups. In Study 2, AIMS total score for patients receiving AUSTEDO demonstrated statistically significant improvement by 3.0 units from baseline to endpoint (Week 12), compared with 1.6 units in the placebo group with a treatment effect of -1.4 units. Table 5 summarizes the effects of AUSTEDO on tardive dyskinesia based on the AIMS.
Table 5: Improvement in AIMS Total Score in Patients Treated with AUSTEDO in Study 1 and Study 2 Study Treatment Group Primary Efficacy Measure: AIMS Total Score Mean Baseline Score (SD) LS Mean Change from Baseline (SE) Treatment Effect (95% CI) Study 1
AUSTEDO 36 mg*
(n= 55)
10.1 (3.21)
-3.3 (0.42)
-1.9 (-3.09, -0.79)
AUSTEDO 24 mg
(n= 49)
9.4 (2.93)
-3.2 (0.45)
-1.8 (-3.00, -0.63)
AUSTEDO 12 mg
(n= 60)
9.6 (2.40)
-2.1 (0.42)
-0.7 (-1.84, 0.42)
Placebo (n= 58)
9.5 (2.71)
-1.4 (0.41)
Study 2
AUSTEDO
(12-48 mg/day)*
(n= 56)
9.7 (4.14)
-3.0 (0.45)
-1.4 (-2.6, -0.2)
Placebo (n= 57)
9.6 (3.78)
-1.6 (0.46)
*Dose that was statistically significantly different from placebo after adjusting for multiplicity.
LS Mean = Least-squares mean; SD = Standard deviation; SE = Standard error; CI = 2-sided 95% confidence interval
Figure 3: Least Square Means of Change in AIMS Total Score from Baseline for AUSTEDO Compared to Placebo (Study 1)
SE = Standard error
Figure 4: Percent of Patients with Specified Magnitude of AIMS Total Score Improvement at the End of Week 12 (Study 1)
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
AUSTEDO tablets are available in the following strengths and packages:
6 mg: round, purple-coated tablets, with “SD” over “6” printed in black ink on one side.
Bottles of 60 tablets: NDC: 68546-170-60.
9 mg: round, blue-coated tablets, with “SD” over “9” printed in black ink on one side.
Bottles of 60 tablets: NDC: 68546-171-60.
12 mg: round, beige-coated tablets, with “SD” over “12” printed in black ink on one side.
Bottles of 60 tablets: NDC: 68546-172-60.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
Administration Instructions
Advise patients to take AUSTEDO with food. AUSTEDO tablets should be swallowed whole and not chewed, crushed, or broken [see Dosage and Administration (2.1)].
Risk of Depression and Suicide in Patients with Huntington’s Disease
Advise patients, their caregivers, and families that AUSTEDO may increase the risk of depression, worsening depression, and suicidality, and to immediately report any symptoms to a healthcare provider [see Contraindications (4), Warnings and Precautions (5.2)].
Prolongation of the QTc Interval
Inform patients to consult their physician immediately if they feel faint, lose consciousness, or have heart palpitations [see Warnings and Precautions (5.3)]. Advise patients to inform physicians that they are taking AUSTEDO before any new drug is taken.
Parkinsonism
Inform patients that AUSTEDO may cause Parkinson-like symptoms, which could be severe. Advise patients to consult their healthcare provider if they experience slight shaking, body stiffness, trouble moving, trouble keeping their balance, or falls [see Warnings and Precautions (5.6)].
Risk of Sedation and Somnolence
Advise patients that AUSTEDO may cause sedation and somnolence and may impair the ability to perform tasks that require complex motor and mental skills. Until they learn how they respond to a stable dose of AUSTEDO, patients should be careful doing activities that require them to be alert, such as driving a car or operating machinery [see Warnings and Precautions (5.7)].
Interaction with Alcohol or Other Sedating Drugs
Advise patients that alcohol or other drugs that cause sleepiness will worsen somnolence [see Drug Interactions (7.6)].
Concomitant Medications
Advise patients to notify their physician of all medications they are taking and to consult with their healthcare provider before starting any new medications because of a potential for interactions [see Contraindications (4) and Drug Interactions (7.1, 7.5)].
Distributed by:
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454AUS-003
U.S. Patent Nos: 8,524,733; 9,233,959; 9,296,739; 9,550,780
XENAZINE® is a trademark of Valeant Pharmaceuticals Luxembourg S.A.R.L.
-
MEDICATION GUIDE
MEDICATION GUIDE
AUSTEDO® (aw-STED-oh)
(deutetrabenazine)
tablets, for oral useWhat is the most important information I should know about AUSTEDO?
- AUSTEDO can cause serious side effects in people with Huntington’s disease, including:
- depression
- suicidal thoughts
- suicidal actions
- Do not start taking AUSTEDO if you have Huntington’s disease and are depressed (have untreated depression or depression that is not well controlled by medicine) or have suicidal thoughts.
- Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is especially important when AUSTEDO is started and when the dose is changed.
Call your healthcare provider right away if you become depressed or have any of the following symptoms, especially if they are new, worse, or worry you:
- feel sad or have crying spells
- lose interest in seeing your friends or doing things you used to enjoy
- sleep a lot more or a lot less than usual
- feel unimportant
- feel guilty
- feel hopeless or helpless
- feel more irritable, angry, or aggressive than usual
- feel more or less hungry than usual or notice a big change in your body weight
- have trouble paying attention
- feel tired or sleepy all the time
- have thoughts about hurting yourself or ending your life
What is AUSTEDO?
AUSTEDO is a prescription medicine that is used to treat:
- the involuntary movements (chorea) of Huntington’s disease. AUSTEDO does not cure the cause of the involuntary movements, and it does not treat other symptoms of Huntington’s disease, such as problems with thinking or emotions.
- movements in the face, tongue, or other body parts that cannot be controlled (tardive dyskinesia).
It is not known if AUSTEDO is safe and effective in children.
Who should not take AUSTEDO?
Do not take AUSTEDO if you:
- have Huntington’s disease and are depressed or have thoughts of suicide. See “What is the most important information I should know about AUSTEDO?”
- have liver problems.
- are taking a monoamine oxidase inhibitor (MAOI) medicine. Do not take an MAOI within 14 days after you stop taking AUSTEDO. Do not start AUSTEDO if you stopped taking an MAOI in the last 14 days. Ask your healthcare provider or pharmacist if you are not sure.
- are taking reserpine. Do not take medicines that contain reserpine (such as Serpalan and Renese-R) with AUSTEDO. If your healthcare provider plans to switch you from taking reserpine to AUSTEDO, you must wait at least 20 days after your last dose of reserpine before you start taking AUSTEDO.
- are taking tetrabenazine (Xenazine). If your healthcare provider plans to switch you from tetrabenazine (Xenazine) to AUSTEDO, take your first dose of AUSTEDO on the day after your last dose of tetrabenazine (Xenazine).
- are taking valbenazine (Ingrezza).
Before taking AUSTEDO, tell your healthcare provider about all of your medical conditions, including if you:
- have emotional or mental problems (for example, depression, nervousness, anxiety, anger, agitation, psychosis, previous suicidal thoughts or suicide attempts).
- have liver disease.
- have an irregular heart rhythm or heartbeat (QT prolongation, cardiac arrhythmia) or a heart problem called congenital long QT syndrome.
- have low levels of potassium or magnesium in your blood (hypokalemia or hypomagnesemia).
- have breast cancer or a history of breast cancer.
- are pregnant or plan to become pregnant. It is not known if AUSTEDO can harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if AUSTEDO passes into breast milk.
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Taking AUSTEDO with certain other medicines may cause side effects. Do not start any new medicines while taking AUSTEDO without talking to your healthcare provider first.
How should I take AUSTEDO?
- Take AUSTEDO exactly as your healthcare provider tells you to take it.
- Take AUSTEDO by mouth and with food.
- Swallow AUSTEDO tablets whole with water. Do not chew, crush, or break AUSTEDO tablets before swallowing. If you cannot swallow AUSTEDO tablets whole, tell your healthcare provider. You may need a different medicine.
- If your dose of AUSTEDO is 12 mg or more each day, take AUSTEDO tablets 2 times a day in equal doses with food.
- Your healthcare provider will increase your dose of AUSTEDO each week for several weeks, until you and your healthcare provider find the right dose for you.
- Tell your healthcare provider if you stop taking AUSTEDO for more than 1 week. Do not take another dose until you talk to your healthcare provider.
What should I avoid while taking AUSTEDO?
Sleepiness (sedation) is a common side effect of AUSTEDO. While taking AUSTEDO, do not drive a car or operate dangerous machinery until you know how AUSTEDO affects you. Drinking alcohol and taking other drugs that may also cause sleepiness while you are taking AUSTEDO may increase any sleepiness caused by AUSTEDO.
What are the possible side effects of AUSTEDO?
AUSTEDO can cause serious side effects, including:
- Depression and suicidal thoughts or actions in people with Huntington’s disease. See “What is the most important information I should know about AUSTEDO?”
- Irregular heartbeat (QT prolongation). AUSTEDO increases your chance of having certain changes in the electrical activity in your heart. These changes can lead to a dangerous abnormal heartbeat. Taking AUSTEDO with certain medicines may increase this chance.
- If you are at risk of QT prolongation, your healthcare provider should check your heart before and after increasing your AUSTEDO dose above 24 mg a day.
-
Neuroleptic Malignant Syndrome (NMS). Call your healthcare provider right away and go to the nearest emergency room if you develop these signs and symptoms that do not have another obvious cause:
- high fever
- problems thinking
- increased sweating
- stiff muscles
- very fast or uneven heartbeat
- high fever
- Restlessness. You may get a condition where you feel a strong urge to move. This is called akathisia.
- Parkinsonism. Symptoms of parkinsonism include: slight shaking, body stiffness, trouble moving, trouble keeping your balance, or falls.
The most common side effects of AUSTEDO in people with Huntington’s disease include:
- sleepiness (sedation)
- diarrhea
- tiredness
- dry mouth
The most common side effects of AUSTEDO in people with tardive dyskinesia include:
- inflammation of the nose and throat (nasopharyngitis)
- problems sleeping (insomnia)
These are not all the possible side effects of AUSTEDO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store AUSTEDO?
- Store AUSTEDO tablets at room temperature, between 68°F to 77°F (20°C to 25°C).
- Keep the bottle tightly closed to protect AUSTEDO from light and moisture.
Keep AUSTEDO tablets and all medications out of reach of children.
General information about the safe and effective use of AUSTEDO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use AUSTEDO for a condition for which it was not prescribed. Do not give AUSTEDO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about AUSTEDO that is written for health professionals.
What are the ingredients in AUSTEDO?
Active ingredient: deutetrabenazine
Inactive ingredients: ammonium hydroxide, black iron oxide, n‑butyl alcohol, butylated hydroxyanisole, butylated hydroxytoluene, magnesium stearate, mannitol, microcrystalline cellulose, polyethylene glycol, polyethylene oxide, polysorbate 80, polyvinyl alcohol, povidone, propylene glycol, shellac, talc, titanium dioxide, and FD&C blue #2 lake. The 6 mg tablets also contain FD&C red #40 lake. The 12 mg tablets also contain FD&C yellow #6 lake.
Distributed by:
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454AUSMG-003
For more information, go to www.AUSTEDO.com or call 1-888-483-8279.
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: July 2019 - AUSTEDO can cause serious side effects in people with Huntington’s disease, including:
- Package/Label Display Panel
- Package/Label Display Panel
- Package/Label Display Panel
-
Package/Label Display Panel
NDC: 68546-176-28 Rx Only
1 PRESS AND HOLD HERE
Tardive Dyskinesia Titration Pack 12 mg Starting Dose
AUSTEDO® (deutetrabenazine) tablets
6 mg and 9 mg
SAMPLE – NOT FOR SALE
1 Press & hold button 2 Pull out card
Medication Guide Required: Each time AUSTEDO is dispensed, give the patient the accompanying Medication Guide.
1 tablet dispenser containing 28 tablets in the following order:
- Week 1: 14 purple tablets containing 6 mg deutetrabenazine
- Week 2: 14 blue tablets containing 9 mg deutetrabenazine
2 PULL OUT HERE
teva

-
Package/Label Display Panel
NDC: 68546-177-21 Rx Only
1 PRESS AND HOLD HERE
Chorea associated with Huntington's disease Titration Pack 6 mg Starting Dose
AUSTEDO® (deutetrabenazine) tablets
6 mg
SAMPLE – NOT FOR SALE
1 Press & hold button 2 Pull out card
Medication Guide Required: Each time AUSTEDO is dispensed, give the patient the accompanying Medication Guide.
1 tablet dispenser containing 21 tablets in the following order:
- Week 1: 7 purple tablets containing 6 mg deutetrabenazine
- Week 2: 14 purple tablets containing 6 mg deutetrabenazine
2 PULL OUT HERE
teva

-
INGREDIENTS AND APPEARANCE
AUSTEDO
deutetrabenazine tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68546-170 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEUTETRABENAZINE (UNII: P341G6W9NB) (DEUTETRABENAZINE - UNII:P341G6W9NB) DEUTETRABENAZINE 6 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) POLYETHYLENE GLYCOL 2000000 (UNII: 5K3991GVWI) Product Characteristics Color PURPLE Score no score Shape ROUND Size 10mm Flavor Imprint Code SD;6 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68546-170-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2017 2 NDC: 68546-170-66 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 04/12/2017 AUSTEDO
deutetrabenazine tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68546-171 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEUTETRABENAZINE (UNII: P341G6W9NB) (DEUTETRABENAZINE - UNII:P341G6W9NB) DEUTETRABENAZINE 9 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) POLYETHYLENE GLYCOL 2000000 (UNII: 5K3991GVWI) Product Characteristics Color BLUE Score no score Shape ROUND Size 10mm Flavor Imprint Code SD;9 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68546-171-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2017 2 NDC: 68546-171-66 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 04/12/2017 AUSTEDO
deutetrabenazine tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68546-172 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEUTETRABENAZINE (UNII: P341G6W9NB) (DEUTETRABENAZINE - UNII:P341G6W9NB) DEUTETRABENAZINE 12 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) POLYETHYLENE GLYCOL 2000000 (UNII: 5K3991GVWI) Product Characteristics Color BROWN (beige) Score no score Shape ROUND Size 10mm Flavor Imprint Code SD;12 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68546-172-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2017 2 NDC: 68546-172-66 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 04/12/2017 AUSTEDO
deutetrabenazine kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68546-177 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68546-177-21 1 in 1 CARTON 02/01/2020 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 21 Part 1 of 1 AUSTEDO
deutetrabenazine tabletProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEUTETRABENAZINE (UNII: P341G6W9NB) (DEUTETRABENAZINE - UNII:P341G6W9NB) DEUTETRABENAZINE 6 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) POLYETHYLENE GLYCOL 2000000 (UNII: 5K3991GVWI) Product Characteristics Color PURPLE Score no score Shape ROUND Size 10mm Flavor Imprint Code SD;6 Contains Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 02/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 02/01/2020 AUSTEDO
deutetrabenazine kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68546-176 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68546-176-28 1 in 1 CARTON 02/01/2020 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 14 Part 2 14 Part 1 of 2 AUSTEDO
deutetrabenazine tabletProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEUTETRABENAZINE (UNII: P341G6W9NB) (DEUTETRABENAZINE - UNII:P341G6W9NB) DEUTETRABENAZINE 6 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) POLYETHYLENE GLYCOL 2000000 (UNII: 5K3991GVWI) Product Characteristics Color PURPLE Score no score Shape ROUND Size 10mm Flavor Imprint Code SD;6 Contains Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 02/01/2020 Part 2 of 2 AUSTEDO
deutetrabenazine tabletProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEUTETRABENAZINE (UNII: P341G6W9NB) (DEUTETRABENAZINE - UNII:P341G6W9NB) DEUTETRABENAZINE 9 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) POLYETHYLENE GLYCOL 2000000 (UNII: 5K3991GVWI) Product Characteristics Color BLUE Score no score Shape ROUND Size 10mm Flavor Imprint Code SD;9 Contains Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 02/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208082 02/01/2020 Labeler - Teva Neuroscience, Inc. (009906397)
Trademark Results [AUSTEDO XR]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 AUSTEDO XR 97261591 not registered Live/Pending |
Auspex Pharmaceuticals, Inc. 2022-02-10 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.


