THERAFLU SINUS AND PAIN- acetaminophen, pheniramine maleate, phenylephrine hcl powder, for solution
THERAFLU by
Drug Labeling and Warnings
THERAFLU by is a Otc medication manufactured, distributed, or labeled by GlaxoSmithKline Consumer Healthcare Holdings (US) LLC, SpecGX LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredients (in each packet)
- Purposes
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting consult a doctor promptly.
Do not use
- in a child under 12 years of age
- if you are allergic to acetaminophen
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or a pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema, or chronic bronchitis
- a sodium-restricted diet
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers
- taking the blood thinning drug warfarin
When using this product
- do not exceed recommended dosage
- avoid alcoholic drinks
- may cause drowsiness
- alcohol, sedatives and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
-
Directions
- do not use more than directed
- take every 4 hours, while symptoms persist. Do not take more than 5 packets in 24 hours unless directed by a doctor
Age
Dose
adults and children 12 years of age and over
one packet
children under 12 years of age
do not use
- dissolve contents of one packet into 8 oz. hot water: sip while hot. Consume entire drink within 10-15 minutes.
- if using a microwave, add contents of one packet to 8 oz. of cool water: stir briskly before and after heating, Do not overheat.
- Other information
- Inactive ingredients
- Questions or Comments?
-
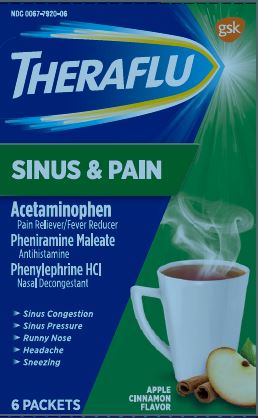
Principal Display
NDC: 0067-7920-06
THERAFLU
Sinus & Pain
ACETAMINOPHEN – PAIN RELIEVER/FEVER REDUCER
PHENIRAMINE MALEATE - ANTIHISTAMINE
PHENYLEPHRINE HCl – NASAL DECONGESTANT
- SINUS CONGESTION
- SINUS PRESSURE
- RUNNY NOSE
- HEADACHE
- SNEEZING
Apple Cinnamon Flavor
6 PACKETS
SINUS CONGESTION & PRESSURE
RUNNY NOSE
HEADACHE
SNEEZING
READ ALL WARNINGS AND DIRECTIONS ON CARTON BEFORE USE.
KEEP CARTON FOR REFERENCE. DO NOT DISCARD.
TAMPER EVIDENT INNER UNIT
DO NOT USE IF SEALED THERAFLU PACKET IS TORN OR BROKEN.
1-855-328-5259
Distributed by:
GSK Consumer Healthcare
Warren, NJ 07059
©2019
GSK group of companies or its licensor.
Trademarks are owned by or licensed to the GSK group of companies.
62000000042281
-
INGREDIENTS AND APPEARANCE
THERAFLU SINUS AND PAIN
acetaminophen, pheniramine maleate, phenylephrine hcl powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0067-7920 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 650 mg PHENIRAMINE MALEATE (UNII: NYW905655B) (PHENIRAMINE - UNII:134FM9ZZ6M) PHENIRAMINE MALEATE 20 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) MALTODEXTRIN (UNII: 7CVR7L4A2D) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SUCROSE (UNII: C151H8M554) TRIACETIN (UNII: XHX3C3X673) TRIBASIC CALCIUM PHOSPHATE (UNII: 91D9GV0Z28) Product Characteristics Color Score Shape Size Flavor APPLE, CINNAMON Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0067-7920-06 6 in 1 CARTON 01/01/2020 1 NDC: 0067-7920-01 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 01/01/2020 Labeler - GlaxoSmithKline Consumer Healthcare Holdings (US) LLC (079944263) Establishment Name Address ID/FEI Business Operations SpecGX LLC 097722284 ANALYSIS(0067-7920) , API MANUFACTURE(0067-7920)
Trademark Results [THERAFLU]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 THERAFLU 88962047 not registered Live/Pending |
GSK Consumer Healthcare S.A. 2020-06-12 |
 THERAFLU 88234661 not registered Live/Pending |
GSK Consumer Healthcare S.A. 2018-12-19 |
 THERAFLU 86467428 4890428 Live/Registered |
GSK CONSUMER HEALTHCARE S.A. 2014-12-01 |
 THERAFLU 86031514 4896050 Live/Registered |
Novartis AG 2013-08-07 |
 THERAFLU 79209949 5362147 Live/Registered |
GSK Consumer Healthcare S.A. 2017-04-28 |
 THERAFLU 74004825 1612634 Dead/Cancelled |
SANDOZ PHARMACEUTICALS CORPORATION 1989-11-24 |
 THERAFLU 74004798 1612633 Dead/Cancelled |
SANDOZ PHARMACEUTICALS CORPORATION 1989-11-24 |
 THERAFLU 73637147 1452879 Live/Registered |
SANDOZ PHARMACEUTICALS CORPORATION 1986-12-24 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.