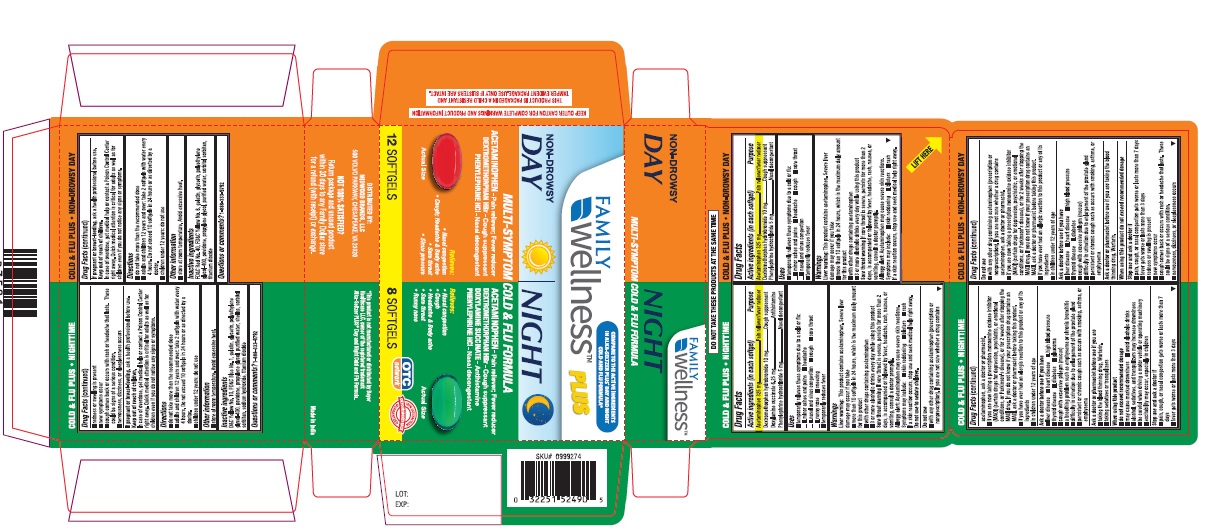
NON DROWSY DAY COLD AND FLU PLUS AND NIGHTTIME COLD AND FLU PLUS- acetaminophen, dextromethorphan hydrobromide, doxylamine succinate, phenylephrine hydrochloride kit
Non Drowsy DAY Cold and Flu Plus and NIGHTTIME Cold and Flu Plus by
Drug Labeling and Warnings
Non Drowsy DAY Cold and Flu Plus and NIGHTTIME Cold and Flu Plus by is a Otc medication manufactured, distributed, or labeled by Spirit Pharmaceuticals LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- COLD & FLU FORMULA DAY NON-DROWSY Active ingredients (in each softgel)
- COLD & FLU FORMULA NIGHT Active ingredients (in each softgel)
- Purposes
- Purposes
- Uses
- Uses
-
DAY COLD & FLU FORMULA
Warnings
Liver warning
These products contain acetaminophen. Severe liver damage may occur if you take
- more than 10 softgels in 24 hours, which is the maximum daily amount for this product
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
- in children under 12 years of age
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- cough with excessive phlegm (mucus)
- difficulty in urination due to enlargement of the prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Stop use and ask a doctor if
- pain, cough, or nasal congestion gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
- nervousness, dizziness, or sleeplessness occurs
-
NIGHTTIME COLD & FLU FORMULA
Warnings
Liver warning
These products contain acetaminophen. Severe liver damage may occur if you take
- more than 10 softgels in 24 hours, which is the maximum daily amount for this product
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use to sedate children.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
- in children under 12 years of age
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- cough with excessive phlegm (mucus)
- a breathing problem such as emphysema or chronic bronchitis
- difficulty in urination due to enlargement of the prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- do not exceed recommended dosage
- may cause marked drowsiness
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Stop use and ask a doctor if
- pain, cough, or nasal congestion gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
- nervousness, dizziness, or sleeplessness occurs
- DAY COLD & FLU FORMULA Directions
- NIGHTTIME COLD & FLU FORMULA Directions
- DAY COLD & FLU FORMULA Other information
- NIGHTTIME COLD & FLU FORMULA Other information
- DAY COLD & FLU FORMULA Inactive ingredients
- NIGHTTIME COLD & FLU FORMULA Inactive ingredients
- DAY COLD & FLU FORMULA
- NIGHTTIME COLD & FLU FORMULA
-
PRINCIPAL DISPLAY PANEL
Compare to the active ingredients in
Alka-Seltzer PLUS® Day and Night Cold and Flu Formula
NON-DROWSY DAY
Cold & Flu Plus
ACETAMINOPHEN- Pain reliever; Fever reducer
DEXTROMETHORPHAN HBr - Cough suppressant
PHENYLEPHRINE HCI - Nasal decongestant
Relieves:
Nasal congestion; Cough;
Headache & body ache;
Sore throat; Sinus pressure12 SOFTGELS
NIGHTTIME
Cold & Flu Plus
ACETAMINOPHEN- Pain reliever; Fever reducer
DEXTROMETHORPHAN HBr - Cough suppressant
DOXYLAMINE SUCCINATE - Antihistamine
PHENYLEPHRINE HCI - Nasal decongestant
Relieves:
Nasal congestion; Cough;
Headache & body ache;
Sore throat; Runny nose
8 SOFTGELS*This product is not manufactured or distributed by Bayer Healthcare LLC, owner of the registered trademark
Alka-Seltzer PLUS® Day and Night Cold and Flu Formula
-
INGREDIENTS AND APPEARANCE
NON DROWSY DAY COLD AND FLU PLUS AND NIGHTTIME COLD AND FLU PLUS
acetaminophen, dextromethorphan hydrobromide, doxylamine succinate, phenylephrine hydrochloride kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68210-1111 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68210-1111-2 1 in 1 CARTON; Type 0: Not a Combination Product 03/11/2020 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BLISTER PACK 12 Part 2 1 BLISTER PACK 8 Part 1 of 2 DAY COLD AND FLU PLUS
acetaminophen, dextromethorphan hydrobromide, phenylephrine hydrochloride capsule, liquid filledProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 5 mg Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange (transparent) Score no score Shape CAPSULE (oblong) Size 21mm Flavor Imprint Code 512 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 CARTON 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 03/11/2020 Part 2 of 2 NIGHTTIME COLD AND FLU PLUS
acetaminophen, dextromethorphan hydrobromide, doxylamine succinate, phenylephrine hydrochloride capsule, liquid filledProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 5 mg DOXYLAMINE SUCCINATE (UNII: V9BI9B5YI2) (DOXYLAMINE - UNII:95QB77JKPL) DOXYLAMINE SUCCINATE 6.25 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SHELLAC (UNII: 46N107B71O) SODIUM HYDROXIDE (UNII: 55X04QC32I) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color green (transparent) Score no score Shape CAPSULE (oblong) Size 21mm Flavor Imprint Code 116 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 CARTON 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 06/05/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 03/11/2020 Labeler - Spirit Pharmaceuticals LLC (179621011) Registrant - Spirit Pharmaceuticals LLC (179621011) Establishment Name Address ID/FEI Business Operations MEDGEL PRIVATE LTD 677385498 manufacture(68210-1111)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.