Childrens Ibuprofen by H2-Pharma, LLC Children's Ibuprofen
Childrens Ibuprofen by
Drug Labeling and Warnings
Childrens Ibuprofen by is a Otc medication manufactured, distributed, or labeled by H2-Pharma, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CHILDRENS IBUPROFEN- ibuprofen suspension
H2-Pharma, LLC
----------
Children's Ibuprofen
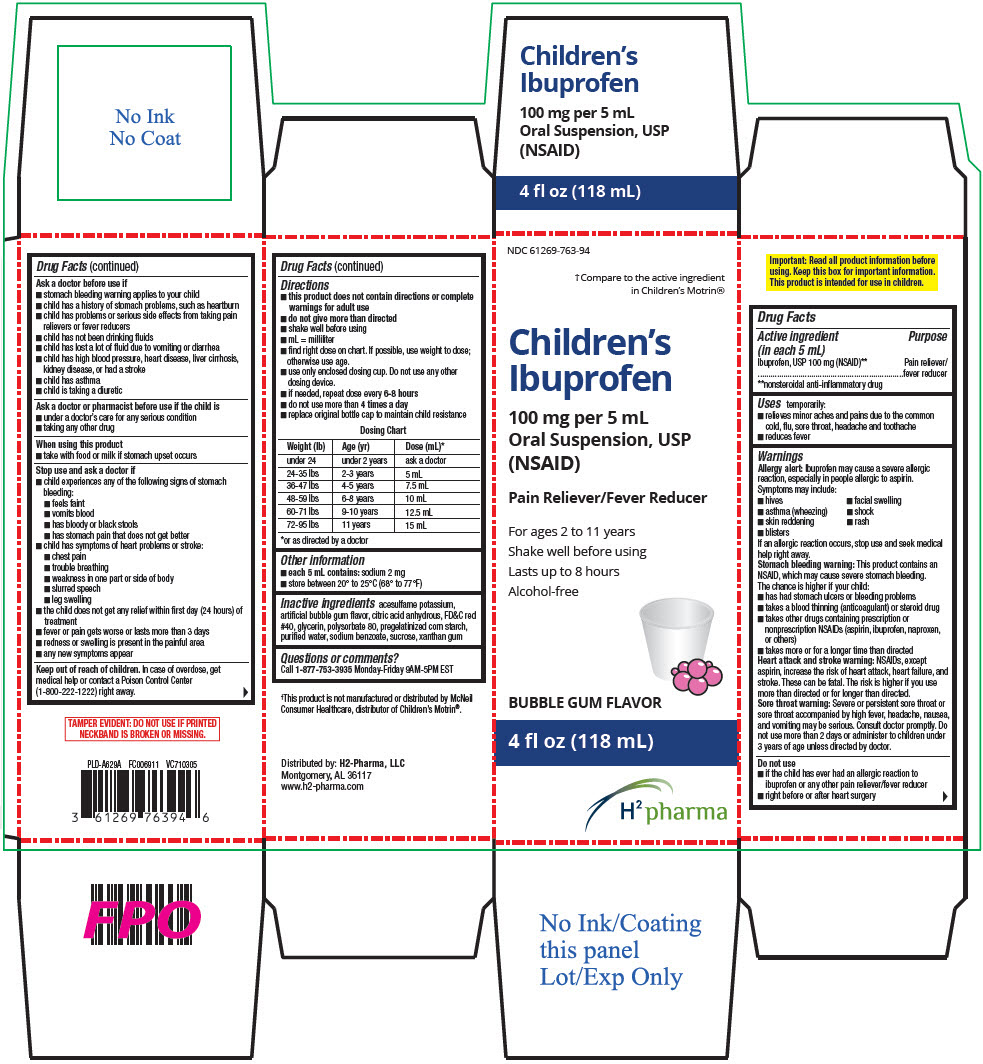
Active ingredient (in each 5 mL)
Ibuprofen, USP 100 mg (NSAID)1
Uses
temporarily:
- relieves minor aches and pains due to the common cold, flu, sore throat, headache and toothache
- reduces fever
Warnings
Allergy alert
Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
- skin reddening
- rash
- blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if your child:
- has had stomach ulcers or bleeding problems
- takes a blood thinning (anticoagulant) or steroid drug
- takes other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- takes more or for a longer time than directed
Heart attack and stroke warning
NSAIDs, except aspirin, increase the risk of heart attack, heart failure, and stroke. These can be fatal. The risk is higher if you use more than directed or for longer than directed.
Sore throat warning
Severe or persistent sore throat or sore throat accompanied by high fever, headache, nausea, and vomiting may be serious. Consult doctor promptly. Do not use more than 2 days or administer to children under 3 years of age unless directed by doctor.
Do not use
- if the child has ever had an allergic reaction to ibuprofen or any other pain reliever/fever reducer
- right before or after heart surgery
Ask a doctor before use if
- stomach bleeding warning applies to your child
- child has a history of stomach problems, such as heartburn
- child has problems or serious side effects from taking pain relievers or fever reducers
- child has not been drinking fluids
- child has lost a lot of fluid due to vomiting or diarrhea
- child has high blood pressure, heart disease, liver cirrhosis, kidney disease, or had a stroke
- child has asthma
- child is taking a diuretic
Ask a doctor or pharmacist before use if the child is
- under a doctor's care for any serious condition
- taking any other drug
Stop use and ask a doctor if
- child experiences any of the following signs of stomach bleeding:
- feels faint
- vomits blood
- has bloody or black stools
- has stomach pain that does not get better
- child has symptoms of heart problems or stroke:
- chest pain
- trouble breathing
- weakness in one part or side of body
- slurred speech
- leg swelling
- the child does not get any relief within first day (24 hours) of treatment
- fever or pain gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appear
Directions
- this product does not contain directions or complete warnings for adult use
- do not give more than directed
- shake well before using
- mL = milliliter
- find right dose on chart. If possible, use weight to dose; otherwise use age.
- use only enclosed dosing cup. Do not use any other dosing device.
- if needed, repeat dose every 6-8 hours
- do not use more than 4 times a day
- replace original bottle cap to maintain child resistance
| Weight (lb) | Age (yr) | Dose (mL)* |
|---|---|---|
|
|
||
| under 24 | under 2 years | ask a doctor |
| 24-35 lbs | 2-3 years | 5 mL |
| 36-47 lbs | 4-5 years | 7.5 mL |
| 48-59 lbs | 6-8 years | 10 mL |
| 60-71 lbs | 9-10 years | 12.5 mL |
| 72-95 lbs | 11 years | 15 mL |
Inactive ingredients
acesulfame potassium, artificial bubble gum flavor, citric acid anhydrous, FD&C red #40, glycerin, polysorbate 80, pregelatinized corn starch, purified water, sodium benzoate, sucrose, xanthan gum
PRINCIPAL DISPLAY PANEL - 118 mL Bottle Carton
NDC: 61269-763-94
†Compare to the active ingredient
in Children's Motrin®
Children's
Ibuprofen
100 mg per 5 mL
Oral Suspension, USP
(NSAID)
Pain Reliever/Fever Reducer
For ages 2 to 11 years
Shake well before using
Lasts up to 8 hours
Alcohol-free
BUBBLE GUM FLAVOR
4 fl oz (118 mL)
H2 pharma
| CHILDRENS IBUPROFEN
ibuprofen suspension |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - H2-Pharma, LLC (028473634) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.