MUCINEX CHILDRENS COUGH- dextromethorphan hydrobromide and guaifenesin solution
Mucinex Childrens by
Drug Labeling and Warnings
Mucinex Childrens by is a Otc medication manufactured, distributed, or labeled by RB Health (US) LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help your child get to sleep
-
Warnings
Do not use in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
Ask a doctor before use if the child has
- cough that occurs with too much phlegm (mucus)
- persistent or chronic cough such as occurs with asthma
-
Directions
- do not give more than 6 doses in any 24-hour period
- measure only with dosing cup provided
- do not use dosing cup with other products
- dose as follows or as directed by a doctor
Age Dose children 6 years to under 12 years of age 5 mL – 10 mL every 4 hours children 4 years to under 6 years of age 2.5 mL – 5 mL every 4 hours children under 4 years of age do not use - Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
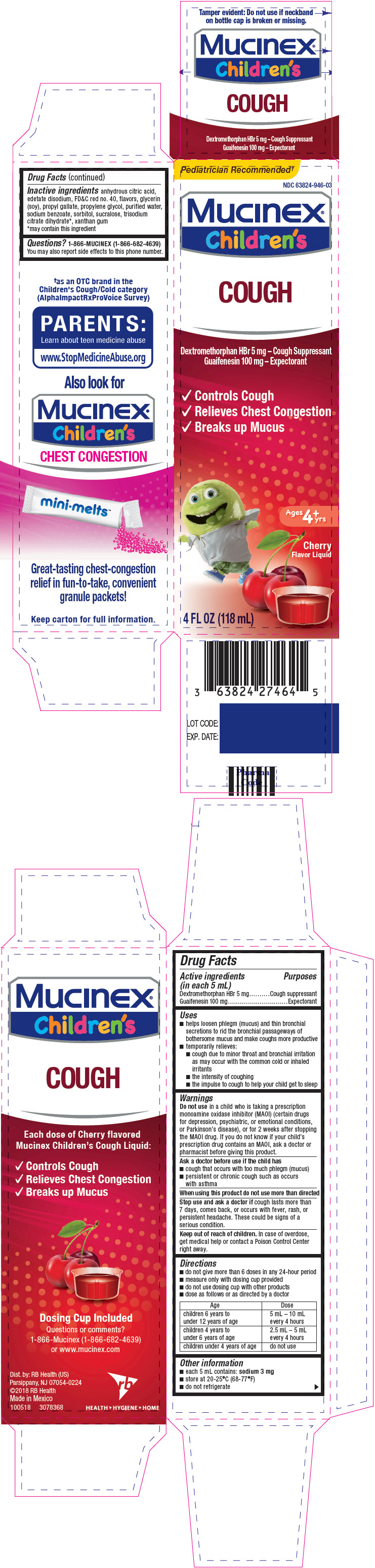
PRINCIPAL DISPLAY PANEL - 118 mL Bottle Carton
Pediatrician Recommended†
NDC: 63824-946-03
Mucinex®
Children'sCOUGH
Dextromethorphan HBr 5 mg – Cough Suppressant
Guaifenesin 100 mg – Expectorant- ✓ Controls Cough
- ✓ Relieves Chest Congestion
- ✓ Breaks up Mucus
Ages
4+
yrsCherry
Flavor Liquid4 FL OZ (118 mL)
-
INGREDIENTS AND APPEARANCE
MUCINEX CHILDRENS COUGH
dextromethorphan hydrobromide and guaifenesin solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 63824-946 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Dextromethorphan Hydrobromide (UNII: 9D2RTI9KYH) (Dextromethorphan - UNII:7355X3ROTS) Dextromethorphan Hydrobromide 5 mg in 5 mL Guaifenesin (UNII: 495W7451VQ) (Guaifenesin - UNII:495W7451VQ) Guaifenesin 100 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) EDETATE DISODIUM (UNII: 7FLD91C86K) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) PROPYL GALLATE (UNII: 8D4SNN7V92) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL (UNII: 506T60A25R) SUCRALOSE (UNII: 96K6UQ3ZD4) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color RED Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 63824-946-03 1 in 1 CARTON 05/01/2018 1 118 mL in 1 BOTTLE; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/01/2018 Labeler - RB Health (US) LLC (081049410)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.