EMUAID® Pain Relief Cream
EMUAID Pain Relief by
Drug Labeling and Warnings
EMUAID Pain Relief by is a Otc medication manufactured, distributed, or labeled by Speer Laboratories LLC, Ariel Laboratories LP. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
EMUAID PAIN RELIEF- menthol cream
Speer Laboratories LLC
----------
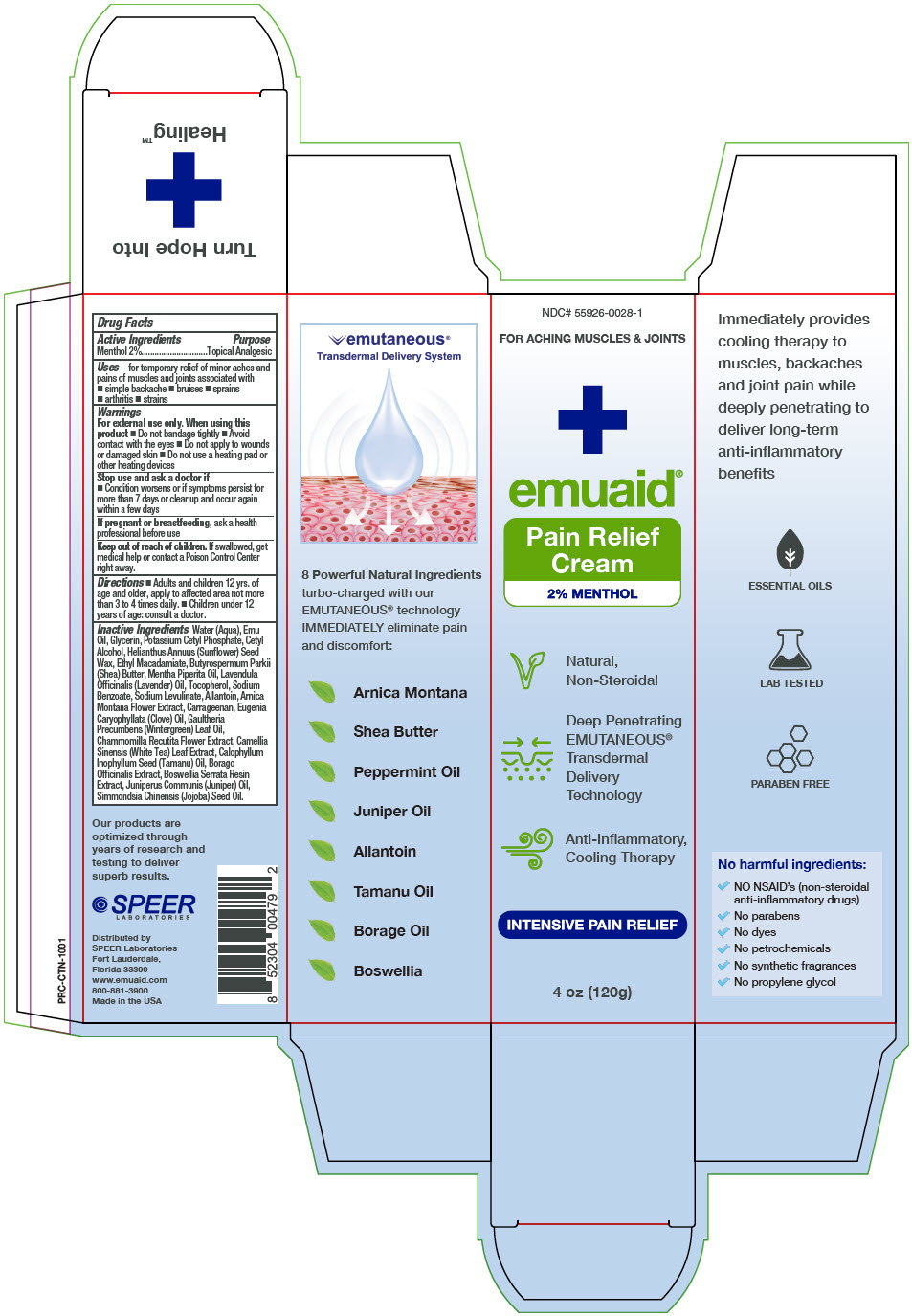
EMUAID® Pain Relief Cream
Uses
for temporary relief of minor aches and pains of muscles and joints associated with
- simple backache
- bruises
- sprains
- arthritis
- strains
Warnings
For external use only. When using this product
- Do not bandage tightly
- Avoid contact with the eyes
- Do not apply to wounds or damaged skin
- Do not use a heating pad or other heating devices
Directions
- Adults and children 12 yrs. of age and older, apply to affected area not more than 3 to 4 times daily.
- Children under 12 years of age: consult a doctor.
Inactive Ingredients
Water (Aqua), Emu Oil, Glycerin, Potassium Cetyl Phosphate, Cetyl Alcohol, Helianthus Annuus (Sunflower) Seed Wax, Ethyl Macadamiate, Butyrospermum Parkii (Shea) Butter, Mentha Piperita Oil, Lavendula Officinalis (Lavender) Oil, Tocopherol, Sodium Benzoate, Sodium Levulinate, Allantoin, Arnica Montana Flower Extract, Carrageenan, Eugenia Caryophyllata (Clove) Oil, Gaultheria Precumbens (Wintergreen) Leaf Oil, Chammomilla Recutita Flower Extract, Camellia Sinensis (White Tea) Leaf Extract, Calophyllum Inophyllum Seed (Tamanu) Oil, Borago Officinalis Extract, Boswellia Serrata Resin Extract, Juniperus Communis (Juniper) Oil, Simmondsia Chinensis (Jojoba) Seed Oil.
| EMUAID PAIN RELIEF
menthol cream |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Speer Laboratories LLC (064900125) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ariel Laboratories LP | 087625133 | MANUFACTURE(55926-0028) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.