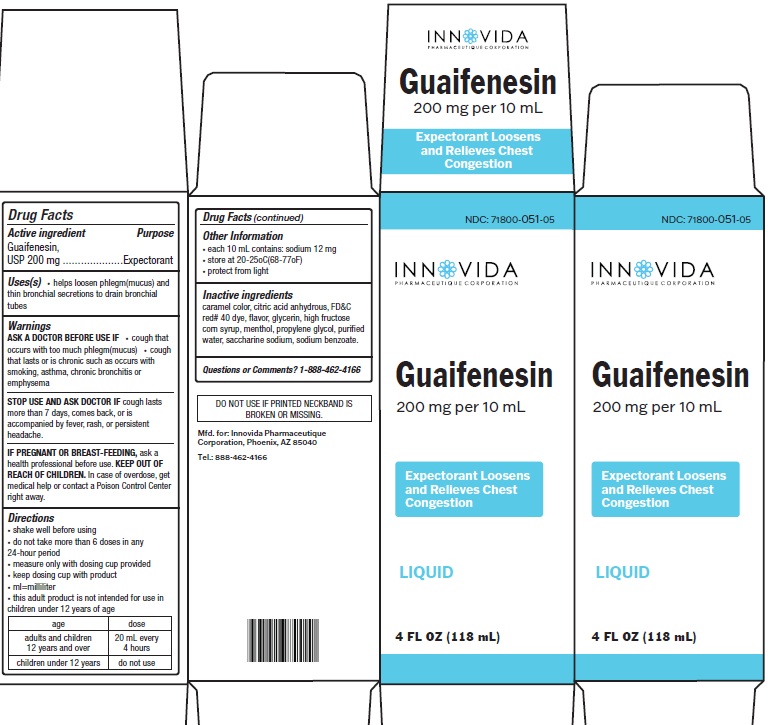
Guaifenesin by Innovida Pharmaeutique Corporation
Guaifenesin by
Drug Labeling and Warnings
Guaifenesin by is a Otc medication manufactured, distributed, or labeled by Innovida Pharmaeutique Corporation. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
GUAIFENESIN- guaifenesin liquid
Innovida Pharmaeutique Corporation
----------
Warnings
ASK A DOCTOR BEFORE USE IF
- cough that occurs with too much phlegm(mucus)
- cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis or emphysema
STOP USE AND ASK DOCTOR IFcough lasts more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache.
IF PREGNANT OR BREAST-FEEDING, ask a health professional before use. KEEP OUT OF REACH OF CHILDREN. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- shake well before using
- do not take more than 6 doses in any 24-hour period
- measure only with dosing cup provided
- keep dosing cup with product
- ml=milliliter
- this adult product is not intended for use in children under 12 years of age
| age | dose |
|
adults and children 12 years and over children under 12 years |
20 mL every 4 hours do not use |
| GUAIFENESIN
guaifenesin liquid |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Innovida Pharmaeutique Corporation (080892908) |
Revised: 12/2025
Document Id: 46e11bc1-bb6a-9eca-e063-6394a90a4288
Set id: d3c0289f-9b8a-4d8f-93bc-e8711ecdf0ce
Version: 2
Effective Time: 20251226