PAIN RELIEF- acetaminophen tablet, film coated
Pain Relief by
Drug Labeling and Warnings
Pain Relief by is a Otc medication manufactured, distributed, or labeled by Publix Super Markets Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
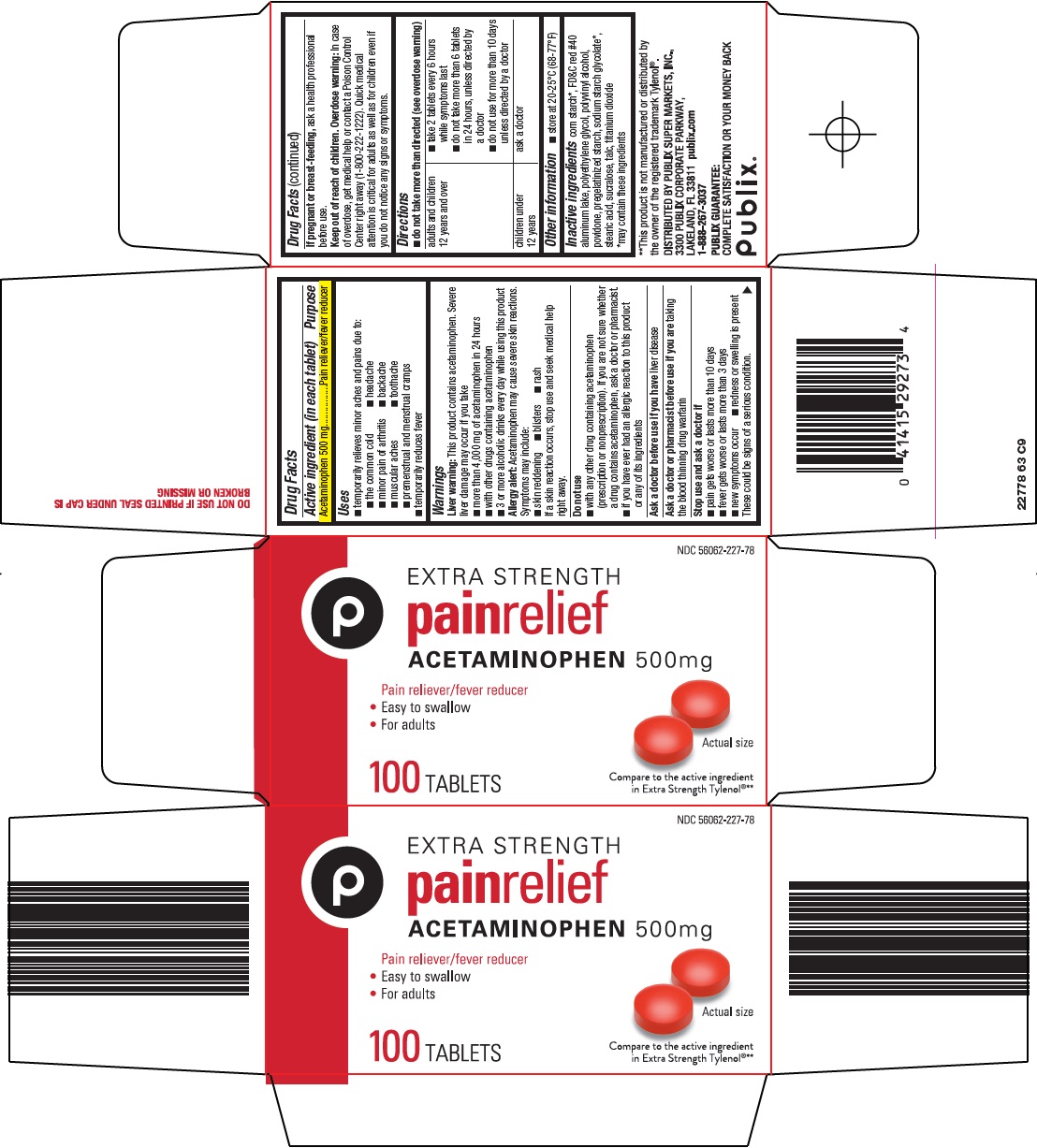
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you have ever had an allergic reaction to this product or any of its ingredients
-
Directions
- do not take more than directed (see overdose warning)
adults and children 12 years and over
- take 2 tablets every 6 hours while symptoms last
- do not take more than 6 tablets in 24 hours, unless directed by a doctor
- do not use for more than 10 days unless directed by a doctor
children under 12 years
ask a doctor
- Other information
- Inactive ingredients
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
PAIN RELIEF
acetaminophen tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 56062-227 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) FD&C RED NO. 40 (UNII: WZB9127XOA) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) STEARIC ACID (UNII: 4ELV7Z65AP) SUCRALOSE (UNII: 96K6UQ3ZD4) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color RED Score no score Shape ROUND (convex) Size 10mm Flavor Imprint Code L227 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 56062-227-71 1 in 1 CARTON 11/26/2007 1 50 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC: 56062-227-78 1 in 1 CARTON 11/26/2007 2 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 11/26/2007 Labeler - Publix Super Markets Inc (006922009)
Trademark Results [Pain Relief]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 PAIN RELIEF 97238381 not registered Live/Pending |
Liu, Caiqing 2022-01-25 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.