DEXLANSOPRAZOLE DELAYED RELEASE- dexlansoprazole capsule, delayed release
Dexlansoprazole by
Drug Labeling and Warnings
Dexlansoprazole by is a Prescription medication manufactured, distributed, or labeled by Bryant Ranch Prepack. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DEXLANSOPRAZOLE DELAYED-RELEASE CAPSULES safely and effectively. See full prescribing information for DEXLANSOPRAZOLE DELAYED-RELEASE CAPSULES.
DEXLANSOPRAZOLE delayed-release capsules, for oral use
Initial U.S. Approval: 1995 (lansoprazole)INDICATIONS AND USAGE
Dexlansoprazole delayed-release capsules are proton pump inhibitor (PPI) indicated in patients 12 years of age and older for:
DOSAGE AND ADMINISTRATION
Recommended dosage in patients 12 years of age and older:
- See full prescribing information for complete dosing information for dexlansoprazole delayed-release capsules by indication and age group and dosage adjustment in patients with hepatic impairment. (2.1, 2.2)
Administration Instructions (2.3):
- Take without regard to food.
- Swallow whole; do not chew.
- See full prescribing information for alternative administration options.
DOSAGE FORMS AND STRENGTHS
Delayed-release capsules: 30 mg and 60 mg. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Gastric Malignancy: In adults, symptomatic response with dexlansoprazole delayed-release capsules does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. (5.1)
- Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. (5.2)
- Clostridium difficile-Associated Diarrhea: PPI therapy may be associated with increased risk. (5.3)
- Bone Fracture: Long-term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist or spine. (5.4)
- Severe Cutaneous Adverse Reactions: Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. (5.5)
- Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue dexlansoprazole delayed-release capsules and refer to specialist for evaluation. (5.6)
- Cyanocobalamin (Vitamin B12) Deficiency: Daily long-term use (e.g., longer than 3 years) may lead to malabsorption or a deficiency of cyanocobalamin. (5.7)
- Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. (5.8)
- Interactions with Investigations for Neuroendocrine Tumors: Increases in intragastric pH may result in hypergastrinemia and enterochromaffin-like cell hyperplasia and increased chromogranin A levels which may interfere with diagnostic investigations for neuroendocrine tumors. (5.9, 7)
- Interaction with Methotrexate: Concomitant use with PPIs may elevate and/or prolong serum concentrations of methotrexate and/or its metabolite, possibly leading to toxicity. With high-dose methotrexate administration, consider a temporary withdrawal of dexlansoprazole delayed-release capsules. (5.10, 7)
- Fundic Gland Polyps: Risk increases with long-term use, especially beyond 1 year. Use the shortest duration of therapy. (5.11)
- Risk of Heart Valve Thickening in Pediatric Patients Less than Two Years of Age: Dexlansoprazole delayed-release capsules are not recommended in pediatric patients less than 2 years of age. (5.12, 8.4)
ADVERSE REACTIONS
The most common adverse reactions are:
- Adults (≥2%): diarrhea, abdominal pain, nausea, upper respiratory tract infection, vomiting, and flatulence. (6.1)
- Patients 12 to 17 years of age (≥5%): headache, abdominal pain, diarrhea, nasopharyngitis, and oropharyngeal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact TWi Pharmaceuticals, Inc. at 1-844-518-2989 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See full prescribing information for a list of clinically important drug interactions. (7)
USE IN SPECIFIC POPULATIONS
- Pregnancy: Based on animal data, may cause adverse effects on fetal bone growth and development. (8.1)
- Pediatrics: Based on data with lansoprazole, dexlansoprazole delayed-release capsules are not effective in patients with symptomatic GERD 1 month to less than 1 year of age and nonclinical studies have demonstrated adverse effects in juvenile rats. (8.4)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Healing of Erosive Esophagitis
1.2 Maintenance of Healed Erosive Esophagitis and Relief of Heartburn
1.3 Treatment of Symptomatic Non-Erosive Gastroesophageal Reflux Disease
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Patients 12 Years of Age and Older
2.2 Dosage Adjustment in Patients with Hepatic Impairment for the Healing of Erosive Esophagitis
2.3 Important Administration Information
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Presence of Gastric Malignancy
5.2 Acute Tubulointerstitial Nephritis
5.3 Clostridium difficile-Associated Diarrhea
5.4 Bone Fracture
5.5 Severe Cutaneous Adverse Reactions
5.6 Cutaneous and Systemic Lupus Erythematosus
5.7 Cyanocobalamin (Vitamin B12) Deficiency
5.8 Hypomagnesemia and Mineral Metabolism
5.9 Interactions with Investigations for Neuroendocrine Tumors
5.10 Interaction with Methotrexate
5.11 Fundic Gland Polyps
5.12 Risk of Heart Valve Thickening in Pediatric Patients Less Than Two Years of Age
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.5 Pharmacogenomics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Healing of Erosive Esophagitis in Adults
14.2 Maintenance of Healed Erosive Esophagitis and Relief of Heartburn in Adults
14.3 Treatment of Symptomatic Non-Erosive GERD in Adults
14.4 Pediatric GERD
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Healing of Erosive Esophagitis
Dexlansoprazole delayed-release capsules are indicated in patients 12 years of age and older for healing of all grades of erosive esophagitis (EE) for up to eight weeks.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Patients 12 Years of Age and Older
Table 1. Recommended Dexlansoprazole Delayed-Release Capsules Dosage Regimen by Indication in Patients 12 Years of Age and Older Indication Dosage of Dexlansoprazole Delayed-Release Capsules Duration Healing of EE One 60 mg capsule once daily. Up to 8 weeks. Maintenance of Healed EE and Relief of Heartburn One 30 mg capsule once daily. Controlled studies did not extend beyond 6 months in adults and 16 weeks in patients 12 to 17 years of age. Symptomatic Non-Erosive GERD One 30 mg capsule once daily. 4 weeks. 2.2 Dosage Adjustment in Patients with Hepatic Impairment for the Healing of Erosive Esophagitis
For patients with moderate hepatic impairment (Child-Pugh Class B), the recommended dosage is 30 mg dexlansoprazole delayed-release capsules once daily for up to eight weeks. Dexlansoprazole delayed-release capsules are not recommended in patients with severe hepatic impairment (Child-Pugh Class C) [see Use in Specific Populations (8.6)].
2.3 Important Administration Information
- Take without regard to food.
- Missed doses: If a dose is missed, administer as soon as possible. However, if the next scheduled dose is due, do not take the missed dose, and take the next dose on time. Do not take two doses at one time to make up for a missed dose.
- Swallow whole; do not chew.
- For patients who have trouble swallowing capsules, dexlansoprazole delayed-release capsules can be opened and administered with applesauce as follows:
- Place one tablespoonful of applesauce into a clean container.
- Open capsule.
- Sprinkle intact granules on applesauce.
- Swallow applesauce and granules immediately. Do not chew granules. Do not save the applesauce and granules for later use.
- Alternatively, the capsule can be administered with water via oral syringe or nasogastric (NG) tube.
Administration with Water in an Oral Syringe- Open the capsule and empty the granules into a clean container with 20 mL of water.
- Withdraw the entire mixture into a syringe.
- Gently swirl the syringe in order to keep granules from settling.
- Administer the mixture immediately into the mouth. Do not save the water and granule mixture for later use.
- Refill the syringe with 10 mL of water, swirl gently, and administer.
- Refill the syringe again with 10 mL of water, swirl gently, and administer.
- Open the capsule and empty the granules into a clean container with 20 mL of water.
- Withdraw the entire mixture into a catheter-tip syringe.
- Swirl the catheter-tip syringe gently in order to keep the granules from settling, and immediately inject the mixture through the NG tube into the stomach. Do not save the water and granule mixture for later use.
- Refill the catheter-tip syringe with 10 mL of water, swirl gently, and flush the tube.
- Refill the catheter-tip syringe again with 10 mL of water, swirl gently, and administer.
-
3 DOSAGE FORMS AND STRENGTHS
Dexlansoprazole delayed-release capsules
- 30 mg: strength is an opaque, blue cap with light grey body, imprinted with "T001" in black ink, the capsule contains white to off white film-coated pellets.
- 60 mg: strength is an opaque, blue cap with blue body, imprinted with "T002" in black ink, the capsule contains white to off white film-coated pellets.
-
4 CONTRAINDICATIONS
- Dexlansoprazole delayed-release capsules are contraindicated in patients with known hypersensitivity to any component of the formulation [see Description (11)]. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis and urticaria [see Warnings and Precautions (5.2), Adverse Reactions (6)].
- PPIs, including dexlansoprazole delayed-release capsules, are contraindicated with rilpivirine-containing products [see Drug Interactions (7)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Presence of Gastric Malignancy
In adults, symptomatic response to therapy with dexlansoprazole delayed-release capsules does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a PPI. In older patients, also consider an endoscopy.
5.2 Acute Tubulointerstitial Nephritis
Acute tubulointerstitial nephritis (TIN) has been observed in patients taking PPIs and may occur at any point during PPI therapy. Patients may present with varying signs and symptoms from symptomatic hypersensitivity reactions to non-specific symptoms of decreased renal function (e.g., malaise, nausea, anorexia). In reported case series, some patients were diagnosed on biopsy and in the absence of extra-renal manifestations (e.g., fever, rash or arthralgia).
Discontinue dexlansoprazole delayed-release capsules and evaluate patients with suspected acute TIN [see Contraindications (4)].
5.3 Clostridium difficile-Associated Diarrhea
Published observational studies suggest that PPI therapy like dexlansoprazole delayed-release capsules may be associated with an increased risk of Clostridium difficile-associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve [see Adverse Reactions (6.2)].
Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
5.4 Bone Fracture
Several published observational studies suggest that PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the conditions being treated. Patients at risk for osteoporosis-related fractures should be managed according to established treatment guidelines [see Dosage and Administration (2), Adverse Reactions (6.2)].
5.5 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs [see Adverse Reactions (6.2)]. Discontinue dexlansoprazole delayed-release capsules at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
5.6 Cutaneous and Systemic Lupus Erythematosus
Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) have been reported in patients taking PPIs. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematosus cases were CLE.
The most common form of CLE reported in patients treated with PPIs was subacute CLE (SCLE) and occurred within weeks to years after continuous drug therapy in patients ranging from infants to the elderly. Generally, histological findings were observed without organ involvement.
Systemic lupus erythematosus (SLE) is less commonly reported than CLE in patients receiving PPIs. PPI-associated SLE is usually milder than nondrug induced SLE. Onset of SLE typically occurred within days to years after initiating treatment primarily in patients ranging from young adults to the elderly. The majority of patients presented with rash; however, arthralgia and cytopenia were also reported.
Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients receiving dexlansoprazole delayed-release capsules, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in four to 12 weeks.
Serological testing (e.g., ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations.
5.7 Cyanocobalamin (Vitamin B12) Deficiency
Daily treatment with any acid-suppressing medications over a long period of time (e.g., longer than three years) may lead to malabsorption of cyanocobalamin (Vitamin B12) caused by hypo- or achlorhydria. Rare reports of cyanocobalamin deficiency occurring with acid-suppressing therapy have been reported in the literature. This diagnosis should be considered if clinical symptoms consistent with cyanocobalamin deficiency are observed in patients treated with dexlansoprazole delayed-release capsules.
5.8 Hypomagnesemia and Mineral Metabolism
Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. Hypomagnesemia may lead to hypocalcemia and/or hypokalemia and may exacerbate underlying hypocalcemia in at-risk patients. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesemia (e.g., diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and periodically [see Adverse Reactions (6.2)].
Consider monitoring magnesium and calcium levels prior to initiation of dexlansoprazole delayed-release capsules and periodically while on treatment in patients with a preexisting risk of hypocalcemia (e.g., hypoparathyroidism). Supplement with magnesium and/or calcium as necessary. If hypocalcemia is refractory to treatment, consider discontinuing the PPI.
5.9 Interactions with Investigations for Neuroendocrine Tumors
Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Healthcare providers should temporarily stop dexlansoprazole treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary [see Drug Interactions (7), Clinical Pharmacology (12.2)].
5.10 Interaction with Methotrexate
Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high- dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients [see Drug Interactions (7)].
5.11 Fundic Gland Polyps
PPI use is associated with an increased risk of fundic gland polyps that increases with long-term use, especially beyond one year. Most PPI users who developed fundic gland polyps were asymptomatic and fundic gland polyps were identified incidentally on endoscopy. Use the shortest duration of PPI therapy appropriate to the condition being treated.
5.12 Risk of Heart Valve Thickening in Pediatric Patients Less Than Two Years of Age
Dexlansoprazole delayed-release capsules are not recommended in pediatric patients less than two years of age. Nonclinical studies in juvenile rats with lansoprazole have demonstrated an adverse effect of heart valve thickening. Dexlansoprazole is the R-enantiomer of lansoprazole [see Use in Specific Populations (8.4)].
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in labeling:
- Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.2)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.3)]
- Bone Fracture [see Warnings and Precautions (5.4)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.5)]
- Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.6)]
- Cyanocobalamin (Vitamin B12) Deficiency [see Warnings and Precautions (5.7)]
- Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.8)]
- Fundic Gland Polyps [see Warnings and Precautions (5.11)]
- Risk of Heart Valve Thickening in Pediatric Patients Less than Two Years of Age [see Warnings and Precautions (5.12)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults
The safety of dexlansoprazole delayed-release capsules was evaluated in 4548 adult patients in controlled and single-arm clinical trials, including 863 patients treated for at least six months and 203 patients treated for one year. Patients ranged in age from 18 to 90 years (median age 48 years), with 54% female, 85% Caucasian, 8% Black, 4% Asian, and 3% Other races. Six randomized controlled clinical trials were conducted for the treatment of EE, maintenance of healed EE, and symptomatic GERD, which included 896 patients on placebo, 455 patients on dexlansoprazole delayed-release capsules 30 mg, 2218 patients on dexlansoprazole delayed-release capsules 60 mg, and 1363 patients on lansoprazole 30 mg once daily.
Common Adverse Reactions
The most common adverse reactions (≥2%) that occurred at a higher incidence for dexlansoprazole delayed-release capsules than placebo in the controlled studies are presented in Table 2.
Table 2. Common Adverse Reactions in Controlled Studies in Adults Adverse Reaction Placebo
(N=896)
%Dexlansoprazole Delayed-Release Capsules
30 mg
(N=455)
%Dexlansoprazole Delayed-Release Capsules
60 mg
(N=2218)
%Dexlansoprazole Delayed-Release Capsules
Total
(N=2621)
%Lansoprazole
30 mg
(N=1363)
%Diarrhea 2.9 5.1 4.7 4.8 3.2 Abdominal Pain 3.5 3.5 4.0 4.0 2.6 Nausea 2.6 3.3 2.8 2.9 1.8 Upper Respiratory Tract Infection 0.8 2.9 1.7 1.9 0.8 Vomiting 0.8 2.2 1.4 1.6 1.1 Flatulence 0.6 2.6 1.4 1.6 1.2 Adverse Reactions Resulting in Discontinuation
In controlled clinical studies, the most common adverse reaction leading to discontinuation from dexlansoprazole delayed-release capsules was diarrhea (0.7%).
Less Common Adverse Reactions
Other adverse reactions that were reported in controlled studies at an incidence of less than 2% are listed below by body system:
Blood and Lymphatic System Disorders: anemia, lymphadenopathy
Cardiac Disorders: angina, arrhythmia, bradycardia, chest pain, edema, myocardial infarction, palpitation, tachycardia
Ear and Labyrinth Disorders: ear pain, tinnitus, vertigo
Endocrine Disorders: goiter
Eye Disorders: eye irritation, eye swelling
Gastrointestinal Disorders: abdominal discomfort, abdominal tenderness, abnormal feces, anal discomfort, Barrett's esophagus, bezoar, bowel sounds abnormal, breath odor, colitis microscopic, colonic polyp, constipation, dry mouth, duodenitis, dyspepsia, dysphagia, enteritis, eructation, esophagitis, gastric polyp, gastritis, gastroenteritis, gastrointestinal disorders, gastrointestinal hypermotility disorders, GERD, GI ulcers and perforation, hematemesis, hematochezia, hemorrhoids, impaired gastric emptying, irritable bowel syndrome, mucus stools, oral mucosal blistering, painful defecation, proctitis, paresthesia oral, rectal hemorrhage, retching
General Disorders and Administration Site Conditions: adverse drug reaction, asthenia, chest pain, chills, feeling abnormal, inflammation, mucosal inflammation, nodule, pain, pyrexia
Hepatobiliary Disorders: biliary colic, cholelithiasis, hepatomegaly
Immune System Disorders: hypersensitivity
Infections and Infestations: candida infections, influenza, nasopharyngitis, oral herpes, pharyngitis, sinusitis, viral infection, vulvo-vaginal infection
Injury, Poisoning and Procedural Complications: falls, fractures, joint sprains, overdose, procedural pain, sunburn
Laboratory Investigations: ALP increased, ALT increased, AST increased, bilirubin decreased/increased, blood creatinine increased, blood gastrin increased, blood glucose increased, blood potassium increased, liver function test abnormal, platelet count decreased, total protein increased, weight increase
Metabolism and Nutrition Disorders: appetite changes, hypercalcemia, hypokalemia
Musculoskeletal and Connective Tissue Disorders: arthralgia, arthritis, muscle cramps, musculoskeletal pain, myalgia
Nervous System Disorders: altered taste, convulsion, dizziness, headaches, migraine, memory impairment, paresthesia, psychomotor hyperactivity, tremor, trigeminal neuralgia
Psychiatric Disorders: abnormal dreams, anxiety, depression, insomnia, libido changes
Renal and Urinary Disorders: dysuria, micturition urgency
Reproductive System and Breast Disorders: dysmenorrhea, dyspareunia, menorrhagia, menstrual disorder
Respiratory, Thoracic and Mediastinal Disorders: aspiration, asthma, bronchitis, cough, dyspnea, hiccups, hyperventilation, respiratory tract congestion, sore throat
Skin and Subcutaneous Tissue Disorders: acne, dermatitis, erythema, pruritus, rash, skin lesion, urticaria
Vascular Disorders: deep vein thrombosis, hot flush, hypertension
Additional adverse reactions that were reported in a long-term single-arm trial and were considered related to dexlansoprazole delayed-release capsules by the treating physician included: anaphylaxis, auditory hallucination, B-cell lymphoma, bursitis, central obesity, cholecystitis acute, dehydration, diabetes mellitus, dysphonia, epistaxis, folliculitis, gout, herpes zoster, hyperlipidemia, hypothyroidism, increased neutrophils, MCHC decrease, neutropenia, rectal tenesmus, restless legs syndrome, somnolence, tonsillitis.
Pediatrics
The safety of dexlansoprazole delayed-release capsules was evaluated in controlled and single-arm clinical trials including 166 pediatric patients, 12 to 17 years of age for the treatment of symptomatic non-erosive GERD, healing of EE, maintenance of healed EE and relief of heartburn [see Clinical Studies (14.4)].
The adverse reaction profile was similar to that of adults. The most common adverse reactions that occurred in ≥5% of patients were headache, abdominal pain, diarrhea, nasopharyngitis and oropharyngeal pain.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of dexlansoprazole delayed-release capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: autoimmune hemolytic anemia, idiopathic thrombocytopenic purpura
Ear and Labyrinth Disorders: deafness
Eye Disorders: blurred vision
Gastrointestinal Disorders: oral edema, pancreatitis, fundic gland polyps
General Disorders and Administration Site Conditions: facial edema
Hepatobiliary Disorders: drug-induced hepatitis
Immune System Disorders: anaphylactic shock (requiring emergency intervention), exfoliative dermatitis, SJS/TEN (some fatal), DRESS, AGEP, erythema multiforme
Infections and Infestations: Clostridium difficile-associated diarrhea Metabolism and Nutrition Disorders: hypomagnesemia, hypocalcemia, hypokalemia, hyponatremia
Musculoskeletal System Disorders: bone fracture
Nervous System Disorders: cerebrovascular accident, transient ischemic attack
Renal and Genitourinary Disorders: acute renal failure, erectile dysfunction
Respiratory, Thoracic and Mediastinal Disorders: pharyngeal edema, throat tightness
Skin and Subcutaneous Tissue Disorders: generalized rash, leukocytoclastic vasculitis
-
7 DRUG INTERACTIONS
Tables 3 and 4 include drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with dexlansoprazole delayed-release capsules and instructions for preventing or managing them.
Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs.
Table 3. Clinically Relevant Interactions Affecting Drugs Co-Administered with Dexlansoprazole Delayed-Release Capsules and Interactions with Diagnostics Antiretrovirals Clinical Impact: The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known. - Decreased exposure of some antiretroviral drugs (e.g., rilpivirine, atazanavir, and nelfinavir) when used concomitantly with dexlansoprazole may reduce antiviral effect and promote the development of drug resistance.
- Increased exposure of other antiretroviral drugs (e.g., saquinavir) when used concomitantly with dexlansoprazole may increase toxicity of the antiretroviral drugs.
- There are other antiretroviral drugs which do not result in clinically relevant interactions with dexlansoprazole.
Intervention: Rilpivirine-containing products: Concomitant use with dexlansoprazole delayed-release capsules is contraindicated [see Contraindications (4)]. See prescribing information.
Atazanavir: See prescribing information for atazanavir for dosing information.
Nelfinavir: Avoid concomitant use with dexlansoprazole delayed-release capsules. See prescribing information for nelfinavir.
Saquinavir: See the prescribing information for saquinavir and monitor for potential saquinavir toxicities.
Other antiretrovirals: See prescribing information.Warfarin Clinical Impact: Increased INR and prothrombin time in patients receiving PPIs and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Intervention: Monitor INR and prothrombin time. Dose adjustment of warfarin may be needed to maintain target INR range. See prescribing information for warfarin. Methotrexate Clinical Impact: Concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.10)]. Intervention: A temporary withdrawal of dexlansoprazole delayed-release capsules may be considered in some patients receiving high-dose methotrexate. Digoxin Clinical Impact: Potential for increased exposure of digoxin. Intervention: Monitor digoxin concentrations. Dose adjustment of digoxin may be needed to maintain therapeutic drug concentrations. See prescribing information for digoxin. Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) Clinical Impact: Dexlansoprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. Intervention: Mycophenolate mofetil (MMF): Coadministration of PPIs in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving dexlansoprazole delayed-release capsules and MMF. Use dexlansoprazole delayed-release capsules with caution in transplant patients receiving MMF.
See the prescribing information for other drugs dependent on gastric pH for absorption.Tacrolimus Clinical Impact: Potentially increased exposure of tacrolimus, especially in transplant patients who are intermediate or poor metabolizers of CYP2C19. Intervention: Monitor tacrolimus whole blood trough concentrations. Dose adjustment of tacrolimus may be needed to maintain therapeutic drug concentrations. See prescribing information for tacrolimus. Interactions with Investigations of Neuroendocrine Tumors Clinical Impact: CgA levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.9), Clinical Pharmacology (12.2)]. Intervention: Temporarily stop dexlansoprazole delayed-release capsules treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. Interaction with Secretin Stimulation Test Clinical Impact: Hyper-response in gastrin secretion in response to secretin stimulation test, falsely suggesting gastrinoma. Intervention: Temporarily stop dexlansoprazole delayed-release capsules treatment at least 30 days before assessing to allow gastrin levels to return to baseline [see Clinical Pharmacology (12.2)]. False Positive Urine Tests for THC Clinical Impact: There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs. Intervention: An alternative confirmatory method should be considered to verify positive results. Table 4. Clinically Relevant Interactions Affecting Dexlansoprazole Delayed-Release Capsules When Co-Administered with Other Drugs and Substances CYP2C19 or CYP3A4 Inducers Clinical Impact: Decreased exposure of dexlansoprazole when used concomitantly with strong inducers [see Clinical Pharmacology (12.3)]. Intervention: St. John's Wort, rifampin: Avoid concomitant use with dexlansoprazole delayed-release capsules. Ritonavir-containing products: See prescribing information. CYP2C19 or CYP3A4 Inhibitors Clinical Impact: Increased exposure of dexlansoprazole is expected when used concomitantly with strong inhibitors [see Clinical Pharmacology (12.3)]. Intervention: Voriconazole: See prescribing information. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no studies with dexlansoprazole use in pregnant women to inform a drug-associated risk. Dexlansoprazole is the R-enantiomer of lansoprazole, and published observational studies of lansoprazole use during pregnancy did not demonstrate an association of adverse pregnancy-related outcomes with lansoprazole (see Data).
In animal reproduction studies, oral administration of lansoprazole to rats during organogenesis through lactation at 1.8 times the maximum recommended human dexlansoprazole dose produced reductions in the offspring in femur weight, femur length, crown-rump length and growth plate thickness (males only) on postnatal Day 21 (see Data). These effects were associated with reduction in body weight gain. Advise pregnant women of the potential risk to the fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Dexlansoprazole is the R-enantiomer of lansoprazole. Available data from published observational studies failed to demonstrate an association of adverse pregnancy-related outcomes and lansoprazole use. Methodological limitations of these observational studies cannot definitely establish or exclude any drug- associated risk during pregnancy. In a prospective study by the European Network of Teratology Information Services, outcomes from a group of 62 pregnant women administered median daily doses of 30 mg of lansoprazole were compared to a control group of 868 pregnant women who did not take any PPIs. There was no difference in the rate of major malformations between women exposed to PPIs and the control group, corresponding to a Relative Risk (RR)=1.04, [95% Confidence Interval (CI) 0.25-4.21]. In a population-based retrospective cohort study covering all live births in Denmark from 1996 to 2008, there was no significant increase in major birth defects during analysis of first trimester exposure to lansoprazole in 794 live births. A meta-analysis that compared 1,530 pregnant women exposed to PPIs in at least the first trimester with 133,410 unexposed pregnant women showed no significant increases in risk for congenital malformations or spontaneous abortion with exposure to PPIs (for major malformations Odds Ratio (OR)=1.12, [95% CI 0.86- 1.45] and for spontaneous abortions OR=1.29, [95% CI 0.84-1.97]).
Animal Data
An embryo-fetal development study conducted in rabbits at oral dexlansoprazole doses up to 30 mg/kg/day (approximately nine times the maximum recommended human dexlansoprazole dose [60 mg/day] based on body surface area) during organogenesis showed no effects on fetuses due to dexlansoprazole. In addition, embryo-fetal development studies performed in rats with oral lansoprazole at doses up to 150 mg/kg/day (40 times the recommended human lansoprazole dose based on body surface area) during organogenesis and in rabbits with oral lansoprazole at doses up to 30 mg/kg/day (16 times the recommended human lansoprazole dose based on body surface area) during organogenesis revealed no effects on fetuses due to lansoprazole.
A pre- and postnatal developmental toxicity study in rats with additional endpoints to evaluate bone development was performed with lansoprazole at oral doses of 10 to 100 mg/kg/day (0.2 to 1.8 times the maximum recommended human dexlansoprazole dose of 60 mg based on dexlansoprazole AUC [area under the plasma concentration-time curve]) administered during organogenesis through lactation. Maternal effects observed at 100 mg/kg/day (1.8 times the maximum recommended human dexlansoprazole dose of 60 mg based on dexlansoprazole AUC) included increased gestation period, decreased body weight gain during gestation, and decreased food consumption. The number of stillbirths was increased at this dose, which may have been secondary to maternal toxicity. Body weight of pups was reduced at 100 mg/kg/day starting on postnatal Day 11. Femur weight, femur length, and crown-rump length were reduced at 100 mg/kg/day on postnatal Day 21. Femur weight was still decreased in the 100 mg/kg/day group at age 17 to 18 weeks. Growth plate thickness was decreased in the 100 mg/kg/day males on postnatal Day 21, and was increased in the 30 and 100 mg/kg/day males at age 17 to 18 weeks. The effects on bone parameters were associated with reduction in body weight gain.
8.2 Lactation
Risk Summary
There is no information regarding the presence of dexlansoprazole in human milk, the effects on the breastfed infant, or the effects on milk production. However, lansoprazole and its metabolites are present in rat milk. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for dexlansoprazole delayed-release capsules and any potential adverse effects on the breastfed child from dexlansoprazole delayed-release capsules or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of dexlansoprazole delayed-release capsules have been established in pediatric patients 12 years to 17 years of age for the healing of all grades of EE, the maintenance of healed EE and relief of heartburn, and treatment of heartburn associated with symptomatic non-erosive GERD. Use of dexlansoprazole delayed-release capsules in this age group is supported by evidence from adequate and well-controlled studies of dexlansoprazole delayed-release capsules in adults with additional safety, efficacy and pharmacokinetic data in pediatric patients 12 to 17 years of age. The adverse reaction profile in patients 12 to 17 years of age was similar to adults [see Dosage and Administration (2.1), Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14)].
The safety and effectiveness of dexlansoprazole delayed-release capsules has not been established in pediatric patients less than 12 years of age.
Dexlansoprazole delayed-release capsules are not recommended in pediatric patients less than two years of age [see Warnings and Precautions (5.12)]. Nonclinical studies in juvenile rats treated with lansoprazole (the racemic mixture) have demonstrated adverse effects of heart valve thickening and bone changes at dexlansoprazole exposures which are expected to be similar to or higher than the dexlansoprazole exposure in pediatric patients one year to two years of age, as described below in Juvenile Animal Toxicity Data.
The use of dexlansoprazole delayed-release capsules is not recommended for the treatment of symptomatic GERD in pediatric patients one month to less than one year of age because lansoprazole was not shown to be effective in a multicenter, double-blind controlled trial.
Juvenile Animal Toxicity Data
Heart Valve Thickening
In two oral toxicity studies, thickening of the mitral heart valve occurred in juvenile rats treated with lansoprazole. Heart valve thickening was observed primarily with oral dosing initiated on postnatal Day 7 (age equivalent to neonatal humans) and postnatal Day 14 (human age equivalent of approximately one year) at doses of 250 mg/kg/day and higher (at postnatal Day 7 and postnatal Day 14 respectively, 2.5 and 1.8 times the expected dexlansoprazole exposure based on AUC in pediatric patients one year to two years of age). The treatment durations associated with heart valve thickening ranged from 5 days to 8 weeks. The findings reversed or trended towards reversibility after a 4-week drug-free recovery period. The incidence of heart valve thickening after initiation of dosing on postnatal Day 21 (human age equivalent of approximately two years) was limited to a single rat (1/24) in groups given 500 mg/kg/day for 4 or 8 weeks (2.1 times the expected dexlansoprazole exposure based on AUC in pediatric patients one year to two years of age). Based on the low incidence of heart valve thickening in 21-day old rats and the equivalent human age, the risk of heart valve injury does not appear to be relevant to patients two years of age and older.
Bone Changes
In an eight-week oral toxicity study of lansoprazole in juvenile rats with dosing initiated on postnatal Day 7, doses equal to or greater than 100 mg/kg/day (dexlansoprazole exposure based on AUC approximately equal to that in pediatric patients one year to two years of age) produced delayed growth, with impairment of weight gain observed as early as postnatal Day 10 (age equivalent to neonatal humans). At the end of treatment, the signs of impaired growth at 100 mg/kg/day and higher included reductions in body weight (14 to 44% compared to controls), absolute weight of multiple organs, femur weight, femur length and crown-rump length. Femoral growth plate thickness was reduced only in males and only at the 500 mg/kg/day dose. The effects related to delayed growth persisted through the end of the four-week recovery period. Longer term data were not collected.
8.5 Geriatric Use
Of the total number of patients (n=4548) in clinical studies of dexlansoprazole delayed-release capsules, 11% of patients were aged 65 years and over, while 2% were 75 years and over. No overall differences in safety or effectiveness were observed between these patients and younger patients and other reported clinical experience has not identified significant differences in responses between geriatric and younger patients, but greater sensitivity of some older individuals cannot be ruled out [see Clinical Pharmacology (12.3)].
8.6 Hepatic Impairment
No dosage adjustment for dexlansoprazole delayed-release capsules is necessary for patients with mild hepatic impairment (Child-Pugh Class A).
In a study of adult patients with moderate hepatic impairment (Child-Pugh Class B) who received a single dose of 60 mg dexlansoprazole delayed-release capsules, there was a significant increase in systemic exposure of dexlansoprazole compared to healthy subjects with normal hepatic function [see Clinical Pharmacology (12.3)]. Therefore, for patients with moderate hepatic impairment (Child-Pugh Class B), dosage reduction is recommended for the healing of EE [see Dosage and Administration (2.2)].
No studies have been conducted in patients with severe hepatic impairment (Child-Pugh Class C); the use of dexlansoprazole delayed-release capsules is not recommended for these patients [see Dosage and Administration (2.2)].
-
10 OVERDOSAGE
There have been no reports of significant overdose with dexlansoprazole delayed-release capsules. Multiple doses of dexlansoprazole delayed-release capsules 120 mg and a single dose of dexlansoprazole delayed-release capsules 300 mg did not result in death or other severe adverse events. However, serious adverse events of hypertension have been reported in association with twice daily doses of dexlansoprazole delayed-release capsules 60 mg. Nonserious adverse reactions observed with twice daily doses of dexlansoprazole delayed-release capsules 60 mg include hot flashes, contusion, oropharyngeal pain, and weight loss. Dexlansoprazole is not expected to be removed from the circulation by hemodialysis.
In the event of over-exposure, treatment should be symptomatic and supportive.
If over-exposure occurs, call your poison control center at 1-800-222-1222 for current information on the management of poisoning or over-exposure.
-
11 DESCRIPTION
The active ingredient in dexlansoprazole delayed-release capsules, a proton pump inhibitor, is (+)-2-[(R)-{[3-methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl] methyl} sulfinyl]-1H-benzimidazole, a compound that inhibits gastric acid secretion. Dexlansoprazole is the R-enantiomer of lansoprazole (a racemic mixture of the R- and S-enantiomers). Its empirical formula is: C16H14F3N3O2S, with a molecular weight of 369.36.
Dexlansoprazole has the following chemical structure:
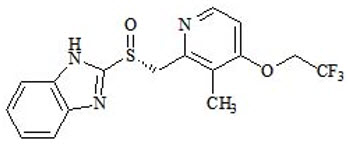
Dexlansoprazole is a white to nearly white crystalline powder which melts with decomposition at 140°C. Dexlansoprazole is freely soluble in dimethylformamide, methanol, dichloromethane, ethanol, and ethyl acetate; and soluble in acetonitrile; slightly soluble in ether; and very slightly soluble in water; and practically insoluble in hexane.
Dexlansoprazole is stable when exposed to light. Dexlansoprazole is more stable in neutral and alkaline conditions than acidic conditions.
Dexlansoprazole is supplied for oral administration as a dual delayed-release formulation in capsules. The capsules contain dexlansoprazole in a mixture of two types of enteric-coated granules with different pH-dependent dissolution profiles [see Clinical Pharmacology (12.3)].
Dexlansoprazole delayed-release capsules are available in two dosage strengths: 30 and 60 mg, per capsule. Each capsule contains enteric-coated granules consisting of dexlansoprazole (active ingredient) and the following inactive ingredients: sugar spheres, hypromellose, sucrose, sodium hydroxide, magnesium carbonate, titanium dioxide, methacrylic acid and ethyl acrylate copolymer dispersion, triethyl citrate, polyethylene glycol, polysorbate 80 and talc. The components of the capsule shell include the following inactive ingredients: FDA/E172 Black iron oxide, titanium dioxide, hypromellose, and FD&C Blue #1. The black imprinting ink contains: shellac, black iron oxide, FD&C Blue #2, FD&C Red #40, FD&C Blue #1, and D&C Yellow #10.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Dexlansoprazole belongs to a class of antisecretory compounds, the substituted benzimidazoles, that suppress gastric acid secretion by specific inhibition of the (H+, K+)-ATPase at the secretory surface of the gastric parietal cell. Because this enzyme is regarded as the acid (proton) pump within the parietal cell, dexlansoprazole has been characterized as a gastric proton-pump inhibitor, in that it blocks the final step of acid production.
12.2 Pharmacodynamics
Antisecretory Activity
The effects of dexlansoprazole delayed-release capsules 60 mg (n=20) or lansoprazole 30 mg (n=23) once daily for five days on 24 hour intragastric pH were assessed in healthy subjects in a multiple-dose crossover study. The results are summarized in Table 5.
Table 5. Effect on 24 Hour Intragastric pH on Day 5 After Administration of Dexlansoprazole Delayed-Release Capsules or Lansoprazole Dexlansoprazole Delayed-Release Capsules
60 mgLansoprazole
30 mgMean Intragastric pH 4.55 4.13 % Time Intragastric pH >4 (hours) 71
(17 hours)60
(14 hours)Serum Gastrin Effects
The effect of dexlansoprazole on serum gastrin concentrations was evaluated in approximately 3460 patients in clinical trials up to eight weeks and in 1023 patients for up to six to 12 months. The mean fasting gastrin concentrations increased from baseline during treatment with 30 and 60 mg dexlansoprazole delayed-release capsules. In patients treated for more than six months, mean serum gastrin levels increased during approximately the first three months of treatment and were stable for the remainder of treatment. Mean serum gastrin levels returned to pretreatment levels within one month of discontinuation of treatment.
Increased gastrin causes enterochromaffin-like cell hyperplasia and increased serum CgA levels. The increased CgA levels may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.9)].
Enterochromaffin-Like Cell (ECL) Effects
There were no reports of ECL cell hyperplasia in gastric biopsy specimens obtained from 653 patients treated with dexlansoprazole delayed-release capsules 30, 60, or 90 mg for up to 12 months.
During lifetime exposure of rats dosed daily with up to 150 mg/kg/day of lansoprazole, marked hypergastrinemia was observed followed by ECL cell proliferation and formation of carcinoid tumors, especially in female rats [see Nonclinical Toxicology (13.1)].
12.3 Pharmacokinetics
The dual delayed-release formulation of dexlansoprazole capsules results in a dexlansoprazole plasma concentration-time profile with two distinct peaks; the first peak occurs one to two hours after administration, followed by a second peak within four to five hours (see Figure 1). Dexlansoprazole is eliminated with a half-life of approximately one to two hours in healthy subjects and in patients with symptomatic GERD. No accumulation of dexlansoprazole occurs after multiple, once daily doses of dexlansoprazole delayed-release capsules 30 or 60 mg although mean AUCt and Cmax values of dexlansoprazole were slightly higher (less than 10%) on Day 5 than on Day 1.
Figure 1: Mean Plasma Dexlansoprazole Concentration – Time Profile Following Oral Administration of 30 or 60 mg Dexlansoprazole Delayed-Release Capsules Once Daily for 5 Days in Healthy Adult Subjects 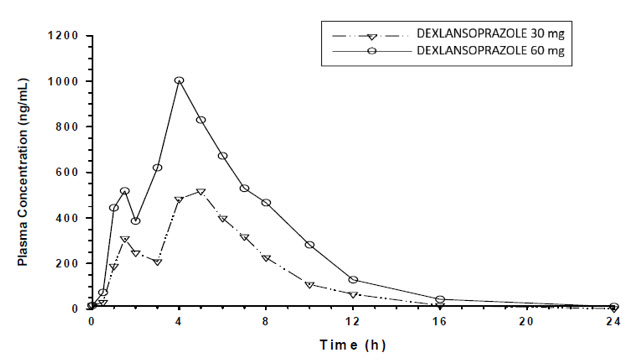
The pharmacokinetics of dexlansoprazole are highly variable, with percent coefficient of variation (%CV) values for Cmax, AUC, and CL/F of greater than 30% (see Table 6).
Table 6. Mean (%CV) Pharmacokinetic Parameters for Adult Subjects on Day 5 After Administration of Dexlansoprazole Delayed-Release Capsules Dose
(mg)Cmax
(ng/mL)AUC24
(ng∙h/mL)CL/F
(L/h)30 658 (40%)
(N=44)3275 (47%)
(N=43)11.4 (48%)
(N=43)60 1397 (51%)
(N=79)6529 (60%)
(N=73)11.6 (46%)
(N=41)Absorption
After oral administration of dexlansoprazole delayed-release capsules 30 or 60 mg to healthy subjects and symptomatic GERD patients, mean Cmax and AUC values of dexlansoprazole increased approximately dose proportionally (see Figure 1).
When granules of dexlansoprazole delayed-release capsules 60 mg are mixed with water and dosed via NG tube or orally via syringe, the bioavailability (Cmax and AUC) of dexlansoprazole was similar to that when dexlansoprazole delayed-release capsules 60 mg was administered as an intact capsule [see Dosage and Administration (2.3)].
Effect on Food
In food-effect studies in healthy subjects receiving dexlansoprazole delayed-release capsules under various fed conditions compared to fasting, increases in Cmax ranged from 12 to 55%, increases in AUC ranged from 9 to 37%, and Tmax varied (ranging from a decrease of 0.7 hours to an increase of three hours) [see Dosage and Administration (2.3)].
Distribution
Plasma protein binding of dexlansoprazole ranged from 96 to 99% in healthy subjects and was independent of concentration from 0.01 to 20 mcg/mL. The apparent volume of distribution (Vz/F) after multiple doses in symptomatic GERD patients was 40 L.
Elimination
Metabolism
Dexlansoprazole is extensively metabolized in the liver by oxidation, reduction, and subsequent formation of sulfate, glucuronide and glutathione conjugates to inactive metabolites. Oxidative metabolites are formed by the cytochrome P450 (CYP) enzyme system including hydroxylation mainly by CYP2C19, and oxidation to the sulfone by CYP3A4.
CYP2C19 is a polymorphic liver enzyme which exhibits three phenotypes in the metabolism of CYP2C19 substrates: extensive metabolizers (*1/*1), intermediate metabolizers (*1/mutant) and poor metabolizers (mutant/mutant). Dexlansoprazole is the major circulating component in plasma regardless of CYP2C19 metabolizer status. In CYP2C19 intermediate and extensive metabolizers, the major plasma metabolites are 5-hydroxy dexlansoprazole and its glucuronide conjugate, while in CYP2C19 poor metabolizers dexlansoprazole sulfone is the major plasma metabolite.
Excretion
Following the administration of dexlansoprazole delayed-release capsules, no unchanged dexlansoprazole is excreted in urine. Following the administration of [14C] dexlansoprazole to six healthy male subjects, approximately 50.7% (standard deviation (SD): 9.0%) of the administered radioactivity was excreted in urine and 47.6% (SD: 7.3%) in the feces.
Apparent clearance (CL/F) in healthy subjects was 11.4 to 11.6 L/hour, respectively, after five days of 30 or 60 mg once daily administration.
Specific Populations
Age: Pediatric Population
The pharmacokinetics of dexlansoprazole in patients under the age of 12 years have not been studied.
Patients 12 to 17 Years of Age
The pharmacokinetics of dexlansoprazole were studied in 36 patients 12 to 17 years of age with symptomatic GERD in a multicenter trial. Patients were randomized to receive dexlansoprazole delayed-release capsules 30 or 60 mg once daily for seven days. The dexlansoprazole mean Cmax and AUC in patients 12 to 17 years of age were 105 and 88%, respectively, compared to those observed in adults at the 30 mg dose, and were 81 and 78%, respectively, at the 60 mg dose (see Tables 6 and 7).
Table 7. Mean (%CV) Pharmacokinetic Parameters in Patients 12 to 17 Years of Age with Symptomatic GERD on Day 7 After Administration of Dexlansoprazole Delayed-Release Capsules Once Daily for 7 Days Dose Cmax
(ng/mL)AUCtau
(ng∙h/mL)CL/F
(L/h)30 mg
(N=17)691
(53)2886
(47)12.8
(48)60 mg
(N=18)1136
(51)5120
(58)15.3
(49)Age: Geriatric Population The terminal elimination half-life of dexlansoprazole is significantly increased in geriatric subjects compared to younger subjects (2.2 and 1.5 hours, respectively). Dexlansoprazole exhibited higher systemic exposure (AUC) in geriatric subjects (34% higher) than younger subjects [see Use in Specific Populations (8.5)].
Sex
In a study of 12 male and 12 female healthy subjects who received a single dose of dexlansoprazole delayed-release capsules 60 mg, females had higher systemic exposure (AUC) (43% higher) than males. This difference in exposure between males and females does not represent a significant safety concern.
Renal Impairment
Dexlansoprazole is extensively metabolized in the liver to inactive metabolites, and no parent drug is recovered in the urine following an oral dose of dexlansoprazole. Therefore, the pharmacokinetics of dexlansoprazole are not expected to be altered in patients with renal impairment, and no studies were conducted in patients with renal impairment. In addition, the pharmacokinetics of lansoprazole were not clinically different in patients with mild, moderate or severe renal impairment compared to healthy subjects with normal renal function.
Hepatic Impairment
In a study of 12 patients with moderate hepatic impairment (Child-Pugh Class B) who received a single dose of 60 mg dexlansoprazole delayed-release capsules, the systemic exposure (AUC) of bound and unbound dexlansoprazole was approximately two times greater compared to subjects with normal hepatic function. This difference in exposure was not due to a difference in protein binding. No studies have been conducted in patients with severe hepatic impairment (Child-Pugh Class C) [see Dosage and Administration (2.2), Use in Specific Populations (8.6)].
Drug-Drug Interactions
Effect of Dexlansoprazole on Other Drugs
Cytochrome P 450 Interactions
Dexlansoprazole is metabolized, in part, by CYP2C19 and CYP3A4 [see Clinical Pharmacology (12.3)].
In vitro studies have shown that dexlansoprazole is not likely to inhibit CYP isoforms 1A1, 1A2, 2A6, 2B6, 2C8, 2C9, 2D6, 2E1 or 3A4. As such, no clinically relevant interactions with drugs metabolized by these CYP enzymes would be expected. Furthermore, in vivo studies showed that dexlansoprazole delayed-release capsules did not have an impact on the pharmacokinetics of co-administered phenytoin (CYP2C9 substrate) or theophylline (CYP1A2 substrate).
The subjects' CYP1A2 genotypes in the drug-drug interaction study with theophylline were not determined. Although in vitro studies indicated that dexlansoprazole delayed-release capsules has the potential to inhibit CYP2C19 in vivo, an in vivo drug- drug interaction study in mainly CYP2C19 extensive and intermediate metabolizers has shown that dexlansoprazole delayed-release capsules does not affect the pharmacokinetics of diazepam (CYP2C19 substrate).
Clopidogrel
Clopidogrel is metabolized to its active metabolite in part by CYP2C19. A study of healthy subjects who were CYP2C19 extensive metabolizers, receiving once daily administration of clopidogrel 75 mg alone or concomitantly with dexlansoprazole delayed-release capsules 60 mg (n=40), for nine days was conducted. The mean AUC of the active metabolite of clopidogrel was reduced by approximately 9% (mean AUC ratio was 91%, with 90% CI of 86 to 97%) when dexlansoprazole delayed-release capsules were co-administered compared to administration of clopidogrel alone.
Pharmacodynamic parameters were also measured and demonstrated that the change in inhibition of platelet aggregation (induced by 5 mcM ADP) was related to the change in the exposure to clopidogrel active metabolite. The effect on exposure to the active metabolite of clopidogrel and on clopidogrel-induced platelet inhibition is not considered clinically important.
12.5 Pharmacogenomics
Effect of CYP2C19 Polymorphism on Systemic Exposure of Dexlansoprazole
Systemic exposure of dexlansoprazole is generally higher in intermediate and poor metabolizers. In male Japanese subjects who received a single dose of dexlansoprazole delayed-release capsules 30 or 60 mg (N=2 to 6 subjects/group), mean dexlansoprazole Cmax and AUC values were up to two times higher in intermediate compared to extensive metabolizers; in poor metabolizers, mean Cmax was up to four times higher and mean AUC was up to 12 times higher compared to extensive metabolizers. Though such study was not conducted in Caucasians and African Americans, it is expected dexlansoprazole exposure in these races will be affected by CYP2C19 phenotypes as well.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of dexlansoprazole was assessed using lansoprazole studies. In two, 24 month carcinogenicity studies, Sprague-Dawley rats were treated orally with lansoprazole at doses of 5 to 150 mg/kg/day, about one to 40 times the exposure on a body surface (mg/m2) basis of a 50 kg person of average height [1.46 m2 body surface area (BSA)] given the recommended human dose of lansoprazole 30 mg/day.
Lansoprazole produced dose-related gastric ECL cell hyperplasia and ECL cell carcinoids in both male and female rats [see Clinical Pharmacology (12.2)].
In rats, lansoprazole also increased the incidence of intestinal metaplasia of the gastric epithelium in both sexes. In male rats, lansoprazole produced a dose-related increase of testicular interstitial cell adenomas. The incidence of these adenomas in rats receiving doses of 15 to 150 mg/kg/day (four to 40 times the recommended human lansoprazole dose based on BSA) exceeded the low background incidence (range = 1.4 to 10%) for this strain of rat.
In a 24 month carcinogenicity study, CD-1 mice were treated orally with lansoprazole doses of 15 to 600 mg/kg/day, two to 80 times the recommended human lansoprazole dose based on BSA. Lansoprazole produced a dose-related increased incidence of gastric ECL cell hyperplasia. It also produced an increased incidence of liver tumors (hepatocellular adenoma plus carcinoma). The tumor incidences in male mice treated with 300 and 600 mg lansoprazole/kg/day (40 to 80 times the recommended human lansoprazole dose based on BSA) and female mice treated with 150 to 600 mg lansoprazole/kg/day (20 to 80 times the recommended human lansoprazole dose based on BSA) exceeded the ranges of background incidences in historical controls for this strain of mice. Lansoprazole treatment produced adenoma of rete testis in male mice receiving 75 to 600 mg/kg/day (10 to 80 times the recommended human lansoprazole dose based on BSA).
A 26 week p53 (+/-) transgenic mouse carcinogenicity study of lansoprazole was not positive.
Lansoprazole was positive in the Ames test and the in vitro human lymphocyte chromosomal aberration assay. Lansoprazole was not genotoxic in the ex vivo rat hepatocyte unscheduled DNA synthesis (UDS) test, the in vivo mouse micronucleus test or the rat bone marrow cell chromosomal aberration test.
Dexlansoprazole was positive in the Ames test and in the in vitro chromosome aberration test using Chinese hamster lung cells. Dexlansoprazole was negative in the in vivo mouse micronucleus test.
The potential effects of dexlansoprazole on fertility and reproductive performance were assessed using lansoprazole studies. Lansoprazole at oral doses up to 150 mg/kg/day (40 times the recommended human lansoprazole dose based on BSA) was found to have no effect on fertility and reproductive performance of male and female rats.
-
14 CLINICAL STUDIES
14.1 Healing of Erosive Esophagitis in Adults
Two multicenter, double-blind, active-controlled, randomized, eight week studies were conducted in patients with endoscopically confirmed EE. Severity of the disease was classified based on the Los Angeles Classification Grading System (Grades A-D). Patients were randomized to one of the following three treatment groups: dexlansoprazole delayed-release capsules 60 mg once daily, dexlansoprazole delayed-release capsules 90 mg once daily or lansoprazole 30 mg once daily. Patients who were H. pylori positive or who had Barrett's Esophagus and/or definite dysplastic changes at baseline were excluded from these studies. A total of 4092 patients were enrolled and ranged in age from 18 to 90 years (median age 48 years) with 54% male. Race was distributed as follows: 87% Caucasian, 5% Black and 8% Other. Based on the Los Angeles Classification, 71% of patients had mild EE (Grades A and B) and 29% of patients had moderate to severe EE (Grades C and D) before treatment.
The studies were designed to test noninferiority. If noninferiority was demonstrated then superiority would be tested. Although noninferiority was demonstrated in both studies, the finding of superiority in one study was not replicated in the other.
The proportion of patients with healed EE at Week 4 or 8 is presented below in Table 8.
Table 8. EE Healing Rates* in Adults: All Grades Study Number of Patients
(N)†Treatment Group
(daily)Week 4
% HealedWeek 8‡
% Healed(95% CI) for the Treatment Difference
(Dexlansoprazole Delayed-Release Capsules–Lansoprazole) by Week 8CI = Confidence interval 1 657 Dexlansoprazole Delayed-Release Capsules
60 mg70 87 (-1.5, 6.1) 648 Lansoprazole 30 mg 65 85 2 639 Dexlansoprazole Delayed-Release Capsules
60 mg66 85 (2.2, 10.5)§ 656 Lansoprazole 30 mg 65 79 *Based on crude rate estimates, patients who did not have endoscopically documented healed EE and prematurely discontinued were considered not healed.
† Patients with at least one postbaseline endoscopy.
‡ Primary efficacy endpoint.
§Demonstrated noninferiority to lansoprazole.Dexlansoprazole delayed-release capsules 90 mg once daily was studied and did not provide additional clinical benefit over dexlansoprazole delayed-release capsules 60 mg once daily.
14.2 Maintenance of Healed Erosive Esophagitis and Relief of Heartburn in Adults
A multicenter, double-blind, placebo-controlled, randomized study was conducted in patients who successfully completed an EE study and showed endoscopically confirmed healed EE. Maintenance of healing and symptom resolution over a six month period was evaluated with dexlansoprazole delayed-release capsules 30 or 60 mg once daily compared to placebo. A total of 445 patients were enrolled and ranged in age from 18 to 85 years (median age 49 years), with 52% female. Race was distributed as follows: 90% Caucasian, 5% Black and 5% Other.
Sixty-six percent of patients treated with 30 mg of dexlansoprazole delayed-release capsules remained healed over the six month time period as confirmed by endoscopy (see Table 9).
Table 9. Maintenance Rates* of Healed EE at Month 6 in Adults Number of Patients
(N)†Treatment Group
(daily)Maintenance Rate
(%)125 Dexlansoprazole Delayed-Release Capsules 30 mg 66.4‡ 119 Placebo 14.3 * Based on crude rate estimates, patients who did not have endoscopically documented relapse and prematurely
discontinued were considered to have relapsed.
† Patients with at least one postbaseline endoscopy
‡ Statistically significant vs placeboDexlansoprazole delayed-release capsules 60 mg once daily was studied and did not provide additional clinical benefit over dexlansoprazole delayed-release capsules 30 mg once daily.
The effect of dexlansoprazole delayed-release capsules 30 mg on maintenance of relief of heartburn was also evaluated. Upon entry into the maintenance study, a majority of patients' baseline heartburn severity was rated as none. Dexlansoprazole delayed-release capsules 30 mg demonstrated a statistically significantly higher percent of 24 hour heartburn-free periods compared to placebo over the six month treatment period (see Table 10). The majority of patients treated with placebo discontinued due to relapse of EE between Month 2 and Month 6.
Table 10. Median Percentage of 24 Hour Heartburn-Free Periods of the Maintenance of Healed EE Study in Adults Overall Treatment* Month 1 Month 6 Treatment Group
(daily)N Heartburn-Free 24 hour Periods
(%)N Heartburn-Free 24 hour Periods
(%)N Heartburn-Free 24 hour Periods
(%)Dexlansoprazole Delayed-Release Capsules
30 mg132 96.1† 126 96.7 80 98.3 Placebo 141 28.6 117 28.6 23 73.3 * Secondary efficacy endpoint
† Statistically significant vs placebo14.3 Treatment of Symptomatic Non-Erosive GERD in Adults
A multicenter, double-blind, placebo-controlled, randomized, four week study was conducted in patients with a diagnosis of symptomatic non-erosive GERD made primarily by presentation of symptoms. These patients who identified heartburn as their primary symptom, had a history of heartburn for six months or longer, had heartburn on at least four of seven days immediately prior to randomization and had no esophageal erosions as confirmed by endoscopy. However, patients with symptoms which were not acid-related may not have been excluded using these inclusion criteria. Patients were randomized to one of the following treatment groups: dexlansoprazole delayed-release capsules 30 mg daily, 60 mg daily, or placebo. A total of 947 patients were enrolled and ranged in age from 18 to 86 years (median age 48 years) with 71% female. Race was distributed as follows: 82% Caucasian, 14% Black and 4% Other.
Dexlansoprazole delayed-release capsules 30 mg provided statistically significantly greater percent of days with heartburn-free 24 hour periods over placebo as assessed by daily diary over four weeks (see Table 11). Dexlansoprazole delayed-release capsules 60 mg once daily was studied and provided no additional clinical benefit over dexlansoprazole delayed-release capsules 30 mg once daily.
Table 11. Median Percentages of 24 Hour Heartburn-Free Periods During the 4 Week Treatment Period of the Symptomatic Non-Erosive GERD Study in Adults N Treatment Group
(daily)Heartburn-Free 24 hour Periods
(%)312 Dexlansoprazole Delayed-Release Capsules 30 mg 54.9* 310 Placebo 18.5 * Statistically significant vs placebo
A higher percentage of patients on dexlansoprazole delayed-release capsules 30 mg had heartburn-free 24 hour periods compared to placebo as early as the first three days of treatment and this was sustained throughout the treatment period (percentage of patients on Day 3: dexlansoprazole delayed-release capsules 38% vs placebo 15%; on Day 28: dexlansoprazole delayed-release capsules 63% vs placebo 40%).
14.4 Pediatric GERD
Use of dexlansoprazole delayed-release capsules in patients 12 to 17 years of age is supported by evidence from adequate and well-controlled studies of dexlansoprazole delayed-release capsules in adults, with additional safety, efficacy, and pharmacokinetic data from studies performed in pediatric patients.
Healing of EE, Maintenance of Healed EE and Relief of Heartburn
In a multicenter, 36 week trial, 62 patients 12 to 17 years of age with a documented history of GERD for at least three months and endoscopically-proven erosive esophagitis (EE) were enrolled to evaluate the healing of EE, maintenance of healed EE and relief of heartburn, followed by an additional 12 weeks without treatment. The median age was 15 years, with males accounting for 61% of the patients. Based on the Los Angeles Classification Grading Scale, 97% of patients had mild EE (Grades A and B), and 3% of patients had moderate to severe EE (Grades C and D) before treatment.
In the first eight weeks, 62 patients were treated with dexlansoprazole delayed-release capsules 60 mg once daily to evaluate the healing of EE. Of the 62 patients, 58 patients completed the eight week trial, and 51 (88%) patients achieved healing of EE, as confirmed by endoscopy, over eight weeks of treatment (see Table 12).
Table 12. Healing of EE at Week 8 in Pediatric Patients 12 to 17 Years of Age Dexlansoprazole Delayed-Release Capsules 60 mg Proportion of randomized patients healed n (%) 51/62 (82%) 95% CI (70, 91)† Proportion of evaluable patients healed* n (%) 51/58 (88%) 95% CI (77, 95)† * Includes only patients who underwent postbaseline endoscopy.
† Reported are the exact confidence limits.After the initial eight weeks of treatment, all 51 patients with healed EE were randomized to receive treatment with dexlansoprazole delayed-release capsules 30 mg or placebo, once daily for an additional 16 weeks to evaluate maintenance of healing and symptom resolution. Maintenance of healing was assessed by endoscopy at Week 24. Of the 51 patients randomized, 13 patients discontinued early. Of these, five patients did not undergo postbaseline endoscopy. Eighteen of 22 (82%) evaluable patients treated with dexlansoprazole delayed-release capsules 30 mg remained healed over the 16 week treatment period as confirmed by endoscopy, compared with 14 of 24 (58%) in placebo (see Table 13).
Table 13. Maintenance of Healed EE at Week 24* in Pediatric Patients 12 to 17 Years of Age Dexlansoprazole Delayed-Release Capsules 30 mg Placebo Proportion of randomized patients who maintained healing of EE 18/25 (72%) 14/26 (54%) n (%) 95% Cl (51, 88)‡ (33, 73)‡ Proportion of evaluable patients who maintained healing of EE† 18/22 (82%) 14/24 (58%) n (%) 95% Cl (60, 95)‡ (37, 78)‡ * Following eight weeks of initial therapy and 16 weeks of maintenance therapy.
† Includes patients with at least one postbaseline endoscopy.
‡ Reported are the exact confidence limits.Relief of heartburn was assessed in randomized patients during the 16 week maintenance period. The median percentage of 24 hour heartburn-free periods was 87% for those receiving dexlansoprazole delayed-release capsules 30 mg compared to 68% for those receiving placebo.
Out of the 32 patients who maintained healing of EE at the end of the 16 week maintenance period, 27 patients (16 treated with dexlansoprazole delayed-release capsules and 11 treated with placebo during the double-blind phase) were followed for an additional 12 weeks without therapy. Twenty-four of the 27 patients completed the 12 week follow-up period. One patient required treatment with acid suppression therapy.
Treatment of Symptomatic Non-Erosive GERD
In a single-arm, open-label, multicenter trial, 104 pediatric patients 12 to 17 years of age with symptomatic non-erosive GERD were treated with dexlansoprazole delayed-release capsules 30 mg once daily, for four weeks to evaluate safety and effectiveness. Patients had a documented history of GERD symptoms for at least three months prior to screening, reported heartburn on at least three out of seven days during screening, and had no esophageal erosions as confirmed by endoscopy. The median age was 15 years, with females accounting for 70% of the patients. During the four week treatment period, the median percentage of 24 hour heartburn free periods was 47%.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Dexlansoprazole delayed-release capsules, 60 mg, are opaque, blue cap with blue body, imprinted with "T002" in black ink, the capsule contains white to off white film-coated pellets. And supplied as:
- NDC: 72162-2239-3: 30 Capsules in a BOTTLE
- NDC: 72162-2239-9: 90 Capsules in a BOTTLE
Store at 20 to 25°C (68 to 77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature].
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504 -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Adverse Reactions
Advise patients to report to their healthcare provider if they experience any signs or symptoms consistent with:
- Hypersensitivity Reactions [see Contraindications (4)]
- Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.2)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.3)]
- Bone Fracture [see Warnings and Precautions (5.4)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.5)]
- Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.6)]
- Cyanocobalamin (Vitamin B12) Deficiency [see Warnings and Precautions (5.7)]
- Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.8)]
Drug Interactions
Advise patients to report to their healthcare provider if they are taking rilpivirine-containing products [see Contraindications (4)] or high-dose methotrexate [see Warnings and Precautions (5.10)].
Pregnancy
Advise a pregnant woman of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider of a known or suspected pregnancy [see Use in Specific Populations (8.1)].
Administration
- Take without regard to food.
- Missed doses: If a dose is missed, administer as soon as possible. However, if the next scheduled dose is due, do not take the missed dose, and take the next dose on time. Do not take two doses at one time to make up for a missed dose.
- Swallow whole; do not chew.
- Can be opened and sprinkled on applesauce for patients who have trouble swallowing the capsule.
- Alternatively, the capsule can be administered with water via oral syringe or NG tube, as described in the Instructions for Use.
-
MEDICATION GUIDE
MEDICATION GUIDE
Dexlansoprazole
(DEX lan SOE pra zol)
delayed-release capsules, for oral useThis Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 07/2023 Read this Medication Guide before you start taking dexlansoprazole delayed-release capsules and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment. What is the most important information that I should know about dexlansoprazole delayed-release capsules?
Dexlansoprazole delayed-release capsules may help your acid-related symptoms, but you could still have serious stomach problems. Talk with your doctor.
Dexlansoprazole delayed-release capsules can cause serious side effects, including:- A type of kidney problem (acute tubulointerstitial nephritis). Some people who take proton pump inhibitor (PPI) medicines, including dexlansoprazole delayed-release capsules, may develop a kidney problem called acute tubulointerstitial nephritis, that can happen at any time during treatment with PPI medicines. Call your doctor right away if you have a decrease in the amount that you urinate or if you have blood in your urine.
-
Diarrhea. Dexlansoprazole delayed-release capsules may increase your risk of getting severe diarrhea. This diarrhea may be caused by an infection (Clostridium difficile) in your intestines.
Call your doctor right away if you have watery stool, stomach pain, and fever that does not go away. - Bone fractures. People who take multiple daily doses of PPI medicines for a long period of time (a year or longer) may have an increased risk of fractures of the hip, wrist or spine. You should take dexlansoprazole delayed-release capsules exactly as prescribed, at the lowest dose possible for your treatment and for the shortest time needed. Talk to your doctor about your risk of bone fracture if you take dexlansoprazole delayed-release capsules.
- Certain types of lupus erythematosus. Lupus erythematosus is an autoimmune disorder (the body's immune cells attack other cells or organs in the body). Some people who take PPI medicines may develop certain types of lupus erythematosus or have worsening of the lupus they already have. Call your doctor right away if you have new or worsening joint pain or a rash on your cheeks or arms that gets worse in the sun.
What are dexlansoprazole delayed-release capsules?
Dexlansoprazole delayed-release capsules are prescription medicine called a proton pump inhibitor (PPI). Dexlansoprazole delayed-release capsules reduce the amount of acid in your stomach.
Dexlansoprazole delayed-release capsules are used in people 12 years of age and older:- for up to 8 weeks to heal acid-related damage to the lining of the esophagus (called erosive esophagitis or EE)
- for up to 6 months in adults and up to 16 weeks in children 12 to 17 years of age to continue healing of erosive esophagitis and relief of heartburn
- for 4 weeks to treat heartburn related to gastroesophageal reflux disease (GERD)
It is not known if dexlansoprazole delayed-release capsules are safe and effective in children under 12 years of age. Dexlansoprazole delayed-release capsules are not recommended in children under 2 years of age and may harm them.
Dexlansoprazole delayed-release capsules are not effective for symptoms of GERD in children under 1 year of age.Who should not take dexlansoprazole delayed-release capsules? Do not take dexlansoprazole delayed-release capsules if you: - are allergic to dexlansoprazole or any of the other ingredients in dexlansoprazole delayed-release capsules. See the end of this Medication Guide for a complete list of ingredients in dexlansoprazole delayed-release capsules.
- are taking a medicine that contains rilpivirine (EDURANT, COMPLERA, JULUCA) used to treat HIV-1 (Human Immunodeficiency Virus)
What should I tell my doctor before taking dexlansoprazole delayed-release capsules?
Before you take dexlansoprazole delayed-release capsules, tell your doctor about all of your medical conditions, including if you:- have been told that you have low magnesium, calcium, potassium or sodium levels in your blood or you are taking a diuretic.
- have liver problems.
- are pregnant, think you may be pregnant or plan to become pregnant. Dexlansoprazole delayed-release capsules may harm your unborn baby. Talk to your doctor about the possible risks to an unborn baby if dexlansoprazole delayed-release capsules are taken during pregnancy.
- are breastfeeding or plan to breastfeed. It is not known if dexlansoprazole passes into your breast milk or if it will affect your baby or your breast milk. Talk to your doctor about the best way to feed your baby if you take dexlansoprazole delayed-release capsules.
Dexlansoprazole delayed-release capsules may affect how other medicines work, and other medicines may affect how dexlansoprazole delayed-release capsules work. Especially tell your doctor if you take methotrexate (Otrexup, Rasuvo, Trexall, Reditrex, Xatmep) or digoxin.
Know the medicines that you take.How should I take dexlansoprazole delayed-release capsules? - Take dexlansoprazole delayed-release capsules exactly as prescribed by your doctor.
- Do not change your dose or stop taking dexlansoprazole delayed-release capsules without talking to your doctor first.
- Take dexlansoprazole delayed-release capsules with or without food.
- Swallow dexlansoprazole delayed-release capsules whole. Do not chew the capsules or the granules that are in the capsules.
- If you have trouble swallowing a whole capsule, you can open the capsule and take the contents in applesauce. See the "Instructions for Use" at the end of this Medication Guide for instructions on how to take dexlansoprazole delayed-release capsules with applesauce.
- See the "Instructions for Use" at the end of this Medication Guide for instructions on how to mix and give dexlansoprazole delayed-release capsules with water using an oral syringe or through a nasogastric tube.
- If you miss a dose of dexlansoprazole delayed-release capsules, take it as soon as you remember. If it is almost time for your next dose, do not take the missed dose. Take your next dose at your regular time. Do not take 2 doses at the same time to make up for the missed dose.
- If you take too much dexlansoprazole delayed-release capsules, call your doctor or your poison control center at 1-800-222-1222 right away or go to the nearest hospital emergency room.
What are the possible side effects of dexlansoprazole delayed-release capsules?
Dexlansoprazole may cause serious side effects, including:- See "What is the most important information I should know about dexlansoprazole delayed-release capsules?".
- Vitamin B12 deficiency. Dexlansoprazole delayed-release capsules reduce the amount of acid in your stomach. Stomach acid is needed to absorb Vitamin B12 properly. Talk with your doctor about the possibility of Vitamin B12 deficiency if you have been on dexlansoprazole delayed-release capsules for a long time (more than 3 years).
- Low magnesium levels in your body can happen in people who have taken dexlansoprazole delayed release capsules for at least 3 months. Tell your doctor right away if you have symptoms of low magnesium levels, including seizures, dizziness, irregular heartbeat, jitteriness, muscle aches or weakness, and spasms of hands, feet or voice.
- Stomach growths (fundic gland polyps). People who take PPI medicines for a long time have an increased risk of developing a certain type of stomach growth called fundic gland polyps, especially after taking PPI medicines for more than 1 year.
-
Severe skin reactions.Dexlansoprazole delayed-release capsules can cause rare but severe skin reactions that may affect any part of your body. These serious skin reactions may need to be treated in a hospital and may be life threatening:
- Skin rash which may have blistering, peeling or bleeding on any part of your skin (including your lips, eyes, mouth, nose, genitals, hands or feet).
- You may also have fever, chills, body aches, shortness of breath, or enlarged lymph nodes.
The most common side effects of dexlansoprazole delayed-release capsules in adults include: - diarrhea
- stomach pain
- nausea
- common cold
- vomiting
- gas
- headache
- stomach pain
- diarrhea
- pain or swelling (inflammation) in your mouth, nose or throat
Serious allergic reactions.Tell your doctor if you get any of the following symptoms with dexlansoprazole delayed-release capsules:- rash
- face swelling
- throat tightness
- difficulty breathing
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of dexlansoprazole delayed-release capsules. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store dexlansoprazole delayed-release capsules? - Store dexlansoprazole delayed-release capsules at room temperature between 68°F to 77°F (20°C to 25°C).
General information about the safe and effective use of dexlansoprazole delayed-release capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use dexlansoprazole delayed-release capsules for a condition for which it was not prescribed. Do not give dexlansoprazole delayed-release capsules to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about dexlansoprazole delayed-release capsules. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about dexlansoprazole delayed-release capsules that is written for health professionals.
For more information, go to pvsafetyamericas@takeda.com or call 1-877-825-3327.What are the ingredients in dexlansoprazole delayed-release capsules?
Active ingredient: dexlansoprazole.
Inactive ingredients: sugar spheres, hypromellose, sucrose, sodium hydroxide, magnesium carbonate, titanium dioxide, methacrylic acid and ethyl acrylate copolymer dispersion, triethyl citrate, polyethylene glycol, polysorbate 80 and talc. The components of the capsule shell include the following inactive ingredients: FDA/E172 Black iron oxide, titanium dioxide, hypromellose, and colorants FD&C Blue #1. The black imprinting ink contains: shellac, black iron oxide, FD&C Blue #2, FD&C Red #40, FD&C Blue #1, and D&C Yellow #10. -
INSTRUCTIONS FOR USEDexlansoprazole (DEX lan SOE pra zol)delayed-release capsules, for oral use
Taking dexlansoprazole delayed-release capsules with applesauce:
- Place 1 tablespoon of applesauce into a clean container.
- Carefully open the capsule and sprinkle the granules onto the applesauce.
- Swallow the applesauce and granules right away. Do not chew the granules. Do not save the applesauce and granules for later use.
Giving dexlansoprazole delayed-release capsules with water using an oral syringe:
- Place 20 mL of water into a clean container.
- Carefully open the capsule and empty the granules into the container of water.
- Use an oral syringe to draw up the water and granule mixture.
- Gently swirl the oral syringe to keep the granules from settling.
- Place the tip of the oral syringe in your mouth. Give the medicine right away. Do not save the water and granule mixture for later use.
- Refill the syringe with 10 mL of water and swirl gently. Place the tip of the oral syringe in your mouth and give the medicine that is left in the syringe.
- Repeat step 6.
Giving dexlansoprazole delayed-release capsules with water through a nasogastric tube (NG tube):
For people who have an NG tube that is size 16 French or larger, dexlansoprazole delayed-release capsules may be given as follows:
- Place 20 mL of water into a clean container.
- Carefully open the capsule and empty the granules into the container of water.
- Use a 60 mL catheter-tip syringe to draw up the water and granule mixture.
- Gently swirl the catheter-tip syringe to keep the granules from settling.
- Connect the catheter-tip syringe to the NG tube.
- Give the mixture right away through the NG tube that goes into the stomach. Do not save the water and granule mixture for later use.
- Refill the catheter-tip syringe with 10 mL of water and swirl gently. Flush the NG tube with the water.
- Repeat step 7.
How should I store dexlansoprazole delayed-release capsules?
- Store dexlansoprazole delayed-release capsules at room temperature between 68°F to 77°F (20°C to 25°C).
Keep dexlansoprazole delayed-release capsules and all medicines out of the reach of children.
Manufactured for:
TWi Pharmaceuticals USA, Inc.
Paramus, NJ 07652This Instructions for Use have been approved by the U.S. Food and Drug Administration.
Manufactured by:
TWi Pharmaceuticals, Inc.
Taoyuan City, 320023, Taiwan
Revised: 07/2023
All trademark names are the property of their respective owners.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DEXLANSOPRAZOLE DELAYED RELEASE
dexlansoprazole capsule, delayed releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 72162-2239(NDC:24979-002) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXLANSOPRAZOLE (UNII: UYE4T5I70X) (DEXLANSOPRAZOLE - UNII:UYE4T5I70X) DEXLANSOPRAZOLE 60 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) SUCROSE (UNII: C151H8M554) MAGNESIUM CARBONATE (UNII: 0E53J927NA) SODIUM HYDROXIDE (UNII: 55X04QC32I) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) POLYSORBATE 80 (UNII: 6OZP39ZG8H) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:2) (UNII: 5KY68S2577) METHACRYLIC ACID - ETHYL ACRYLATE COPOLYMER (1:1) TYPE A (UNII: NX76LV5T8J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SHELLAC (UNII: 46N107B71O) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) Product Characteristics Color BLUE (opaque) , blue (opaque) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code T002 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 72162-2239-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2022 2 NDC: 72162-2239-9 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202666 12/01/2022 Labeler - Bryant Ranch Prepack (171714327) Registrant - Bryant Ranch Prepack (171714327) Establishment Name Address ID/FEI Business Operations Bryant Ranch Prepack 171714327 REPACK(72162-2239) , RELABEL(72162-2239)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
