STOOL SOFTENER DOCUSATE SODIUM 50 MG- docusate sodium capsule, liquid filled
STOOL SOFTENER DOCUSATE SODIUM 50 MG by
Drug Labeling and Warnings
STOOL SOFTENER DOCUSATE SODIUM 50 MG by is a Otc medication manufactured, distributed, or labeled by PuraCap Pharmaceutical LLC, Humanwell PuraCap Pharmaceutical (Wuhan) Co., Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each softgel)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
- Directions
- Other information
- Inactive ingredients
-
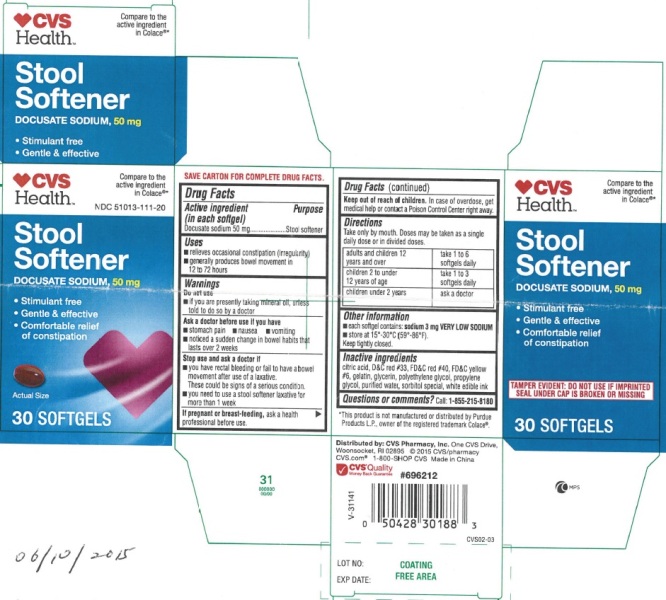
PRINCIPAL DISPLAY PANEL - 30ct
STOOL SOFTENERNDC: 51013-111-20
Compare to the active ingredient in Colace® 50 mg
50 MG DOCUSATE SODIUM PER SOFTGEL 30 Softgels

-
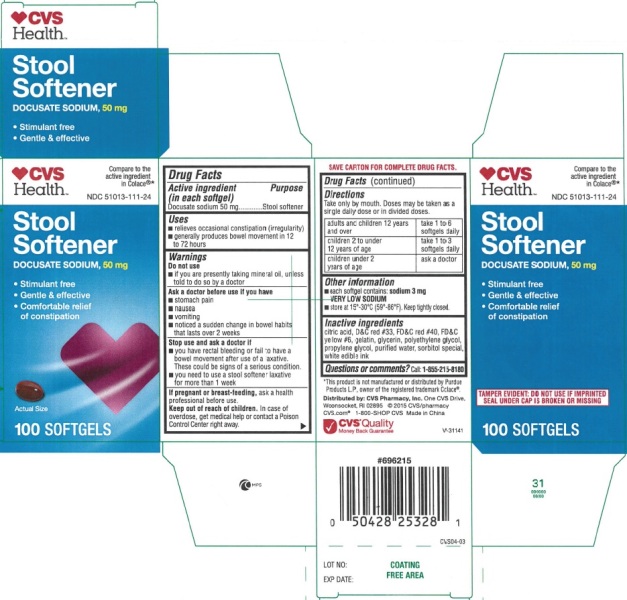
PRINCIPAL DISPLAY PANEL - 100ct
STOOL SOFTENER NDC: 51013-111-24
Compare to the active ingredient in Colace® 50 mg
50 MG DOCUSATE SODIUM PER SOFTGEL 100 Softgels

-
INGREDIENTS AND APPEARANCE
STOOL SOFTENER DOCUSATE SODIUM 50 MG
docusate sodium capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 51013-111 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 50 mg Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Product Characteristics Color red (clear) Score no score Shape CAPSULE (OVAL) Size 13mm Flavor Imprint Code PC20 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51013-111-20 1 in 1 CARTON 08/22/2014 1 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC: 51013-111-24 1 in 1 CARTON 08/22/2014 2 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 08/22/2014 Labeler - PuraCap Pharmaceutical LLC (962106329) Establishment Name Address ID/FEI Business Operations Humanwell PuraCap Pharmaceutical (Wuhan) Co., Ltd. 421293287 analysis(51013-111) , manufacture(51013-111)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.