PGDx elio™ diversiPhi A0840101
GUDID 00192721001241
PERSONAL GENOME DIAGNOSTICS INC.
Methotrexate therapeutic drug monitoring IVD, kit, chemiluminescent immunoassay| Primary Device ID | 00192721001241 |
| NIH Device Record Key | 451b1812-a96e-4c5c-9bc2-9b02a56cbc9b |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | PGDx elio™ diversiPhi |
| Version Model Number | A0840101 |
| Catalog Number | A0840101 |
| Company DUNS | 963427120 |
| Company Name | PERSONAL GENOME DIAGNOSTICS INC. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | true |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00192721001241 [Primary] |
FDA Product Code
| PFT | Reagents For Molecular Diagnostic Test Systems |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2020-05-19 |
| Device Publish Date | 2020-05-11 |
Devices Manufactured by PERSONAL GENOME DIAGNOSTICS INC.
| 00192721001449 - PGDx elio™ server | 2021-08-09 PGDx elio™ server (128C) |
| 00192721001456 - PGDx elio™ server | 2021-08-09 PGDx elio™ server (reconditioned 128C) |
| 00192721001432 - PGDx elio™ server | 2020-07-16 PGDx elio™ server (reconditioned) |
| 00192721000756 - PGDx elio™ server | 2020-05-19 |
| 00192721000763 - PGDx elio™ platform | 2020-05-19 |
| 00192721000770 - PGDx elio™ tissue complete | 2020-05-19 PGDx elio™ tissue complete software |
| 00192721001241 - PGDx elio™ diversiPhi | 2020-05-19 |
| 00192721001241 - PGDx elio™ diversiPhi | 2020-05-19 |
| 00192721001418 - PGDx elio™ tissue complete | 2020-05-19 PGDx elio™ tissue complete reagent kit |
Trademark Results [PGDx elio]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
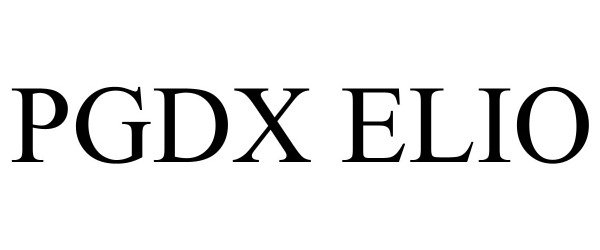 PGDX ELIO 98029210 not registered Live/Pending |
Personal Genome Diagnostics Inc. 2023-06-06 |
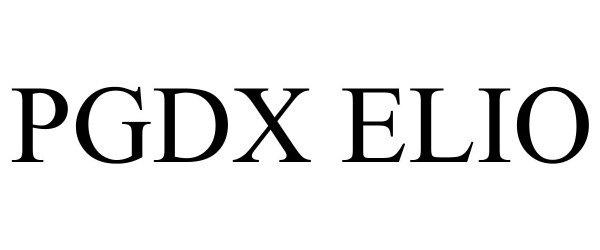 PGDX ELIO 98029209 not registered Live/Pending |
Personal Genome Diagnostics Inc. 2023-06-06 |
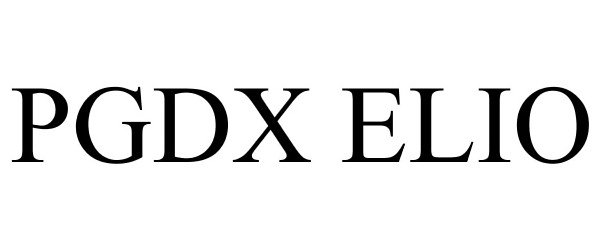 PGDX ELIO 87950371 not registered Live/Pending |
Personal Genome Diagnostics Inc. 2018-06-06 |
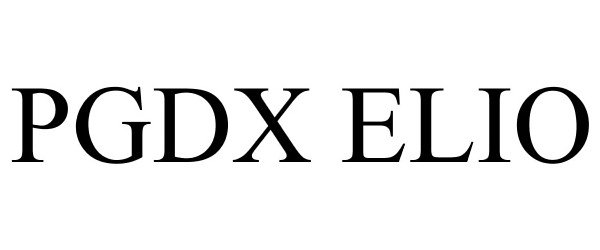 PGDX ELIO 87925167 not registered Live/Pending |
Personal Genome Diagnostics Inc. 2018-05-17 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.