NDC 17856-0114
SSKI
Potassium Iodide
SSKI is a Oral Solution in the Human Prescription Drug category. It is labeled and distributed by Atlantic Biologicals Corp.. The primary component is Potassium Iodide.
| Product ID | 17856-0114_5fa7e81f-17ab-46e6-a7ff-c697f34552c4 |
| NDC | 17856-0114 |
| Product Type | Human Prescription Drug |
| Proprietary Name | SSKI |
| Generic Name | Potassium Iodide |
| Dosage Form | Solution |
| Route of Administration | ORAL |
| Marketing Start Date | 2017-11-13 |
| Marketing Category | UNAPPROVED DRUG OTHER / UNAPPROVED DRUG OTHER |
| Labeler Name | ATLANTIC BIOLOGICALS CORP. |
| Substance Name | POTASSIUM IODIDE |
| Active Ingredient Strength | 1 g/mL |
| NDC Exclude Flag | N |
| Listing Certified Through | 2022-12-31 |
Packaging
NDC 17856-0114-1
120 SYRINGE in 1 BOX, UNIT-DOSE (17856-0114-1) > .1 mL in 1 SYRINGE
| Marketing Start Date | 2021-02-17 |
| NDC Exclude Flag | N |
| Sample Package? | N |
Drug Details
NDC Crossover Matching brand name "SSKI" or generic name "Potassium Iodide"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0245-0003 | SSKI | Potassium Iodide |
| 17856-0114 | SSKI | Potassium Iodide |
| 71740-112 | SSKI | Potassium Iodide |
| 51803-001 | IOSAT | Potassium Iodide |
| 51803-002 | IOSAT | Potassium Iodide |
| 0220-2798 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2799 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2800 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2801 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2802 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2803 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2804 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2805 | Kali iodatum | POTASSIUM IODIDE |
| 0220-2887 | Kali iodatum | POTASSIUM IODIDE |
| 10191-1857 | KALI IODATUM | POTASSIUM IODIDE |
| 68428-453 | Kali iodatum | POTASSIUM IODIDE |
| 71919-383 | Kali iodatum | POTASSIUM IODIDE |
| 43406-0094 | KALI IODATUM 1X | Potassium Iodide |
| 0178-0314 | POTASSIUM IODIDE | POTASSIUM IODIDE |
| 16477-601 | Potassium Iodide | Potassium Iodide |
| 75834-280 | Potassium Iodide | Potassium Iodide |
| 50633-910 | ThyroSafe | Potassium Iodide |
| 66983-350 | ThyroSafe | Potassium Iodide |
| 0256-0210 | ThyroShield | Potassium Iodide |
| 59465-210 | ThyroShield | Potassium Iodide |
Trademark Results [SSKI]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
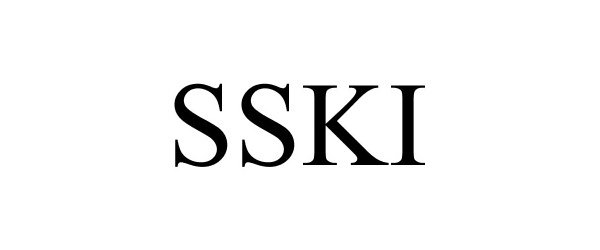 SSKI 77978562 3753309 Live/Registered |
Briareos, Inc. 2007-07-01 |
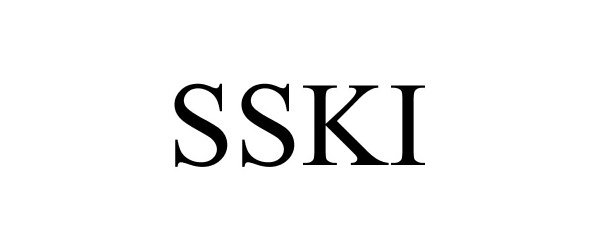 SSKI 77219915 not registered Dead/Abandoned |
Briareos, Inc. 2007-07-01 |
 SSKI 73165702 1142198 Live/Registered |
Upsher-Smith Laboratories, Inc. 1978-04-10 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.